Chapter 35 Immediate Load and Restoration in Implant Dentistry: Rationale and Treatment
Predictable formation of a direct bone-implant interface is a consistent treatment goal in implant dentistry. The two-stage surgical protocol established by Brånemark et al.1 to accomplish osseointegration consisted of several prerequisites, including the following: countersinking the implant below the crestal bone, obtaining and maintaining a soft tissue covering over the implant for 3 to 6 months, and maintaining a nonloaded implant environment for 3 to 6 months. The primary reasons cited for the submerged, countersunk surgical approach to implant placement were to reduce and minimize the risk of bacterial infection, to prevent apical migration of the oral epithelium along the body of the implant, and to reduce and minimize the risk of early implant loading during bone remodeling. After this procedure, a second-stage surgery is necessary to uncover these implants and place a prosthetic abutment. A high degree of long-term, clinical rigid fixation has been observed with this protocol in completely and partially edentulous patients.2,3
During the last 15 years, several authors have reported that root form implants may osseointegrate, even though they reside above the bone and through the soft tissue during early bone remodeling.4–6 This surgical approach has been called a one-stage or nonsubmerged implant procedure and eliminates the second-stage implant uncovery surgery. As a result, the tissue discomfort and healing associated with second-stage surgery are eliminated for the patient. The dentist also eliminates the surgical time for uncovery and suture removal. In addition, the soft tissue is already mature before fabrication of the final prosthesis.
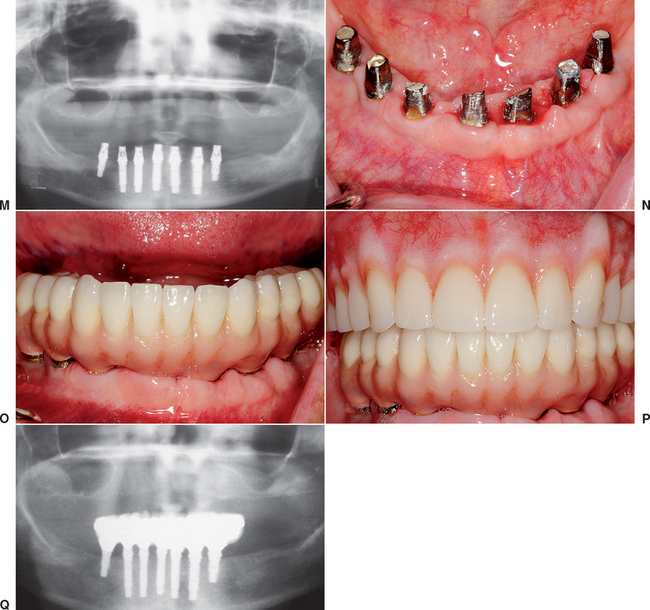
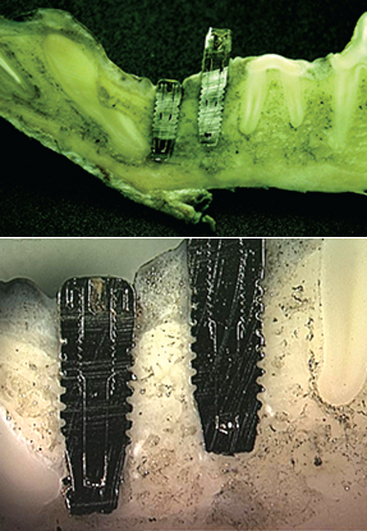
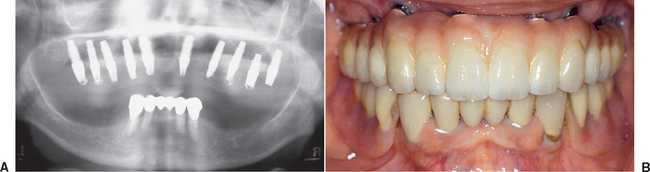
Immediate loading of a dental implant not only includes a nonsubmerged, one-stage surgery but also actually loads the implant with a provisional restoration at the same appointment or shortly thereafter. Immediate loading was the initial protocol suggested with dental implants. These implants yielded a wide range of clinical survival.7–11 On occasion, a direct bone interface could be developed and maintained for more than 20 years12 (Figure 35-1).
TERMINOLOGY
Immediate restoration of dental implants not only includes a nonsubmerged first-stage surgery but also implies the occlusal surfaces and implants are loaded with a provisional or definitive restoration. Discussions have involved whether a restoration must be delivered at the time of surgery for this description. Because the restoration is not truly loaded immediately after implant insertion, regardless of the technique, an agreement should be established as to what guidelines and language may be acceptable to a majority of the profession. Misch et al.13 suggested a terminology for immediate restoration and/or occlusal loading. The immediate occlusal-loading protocol is an implant-supported temporary or definitive restoration in occlusal contact within 2 weeks of the implant insertion. Early occlusal loading refers to an implant-supported restoration in occlusion between 2 weeks and 3 months after implant placement and may use the time period in parentheses (i.e., early [5-week] occlusal loading). Delayed or staged occlusal loading refers to an implant prosthesis with an occlusal load after more than 3 months after implant insertion. The delayed occlusal-loading approach may use either a two-stage surgical procedure that covers the implants with soft tissue or a one-stage approach that exposes a portion of the implant at the initial surgery. Nonfunctional immediate restoration describes an implant prosthesis with no direct occlusal load within 2 weeks of implant insertion and is primarily considered in partially edentulous patients. Nonfunctional early restoration describes a restoration in a partially edentulous patient delivered between 2 weeks and 3 months after the implant insertion.
COMPLETELY EDENTULOUS PATIENTS
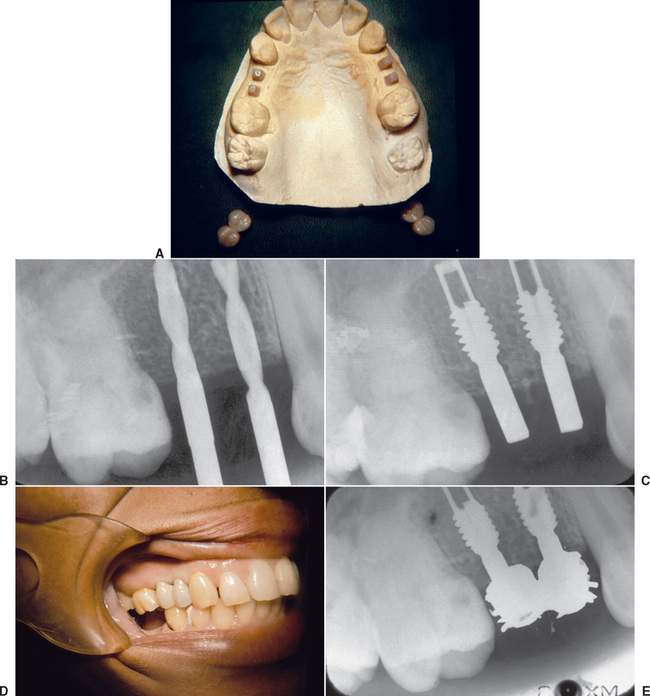
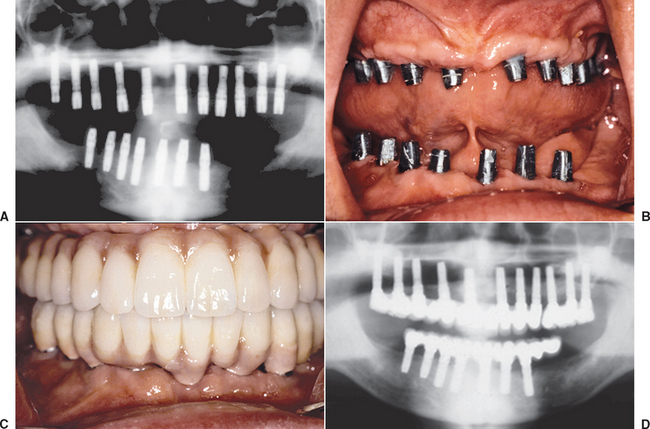
Early studies in immediate loading with root form implants, with a primary goal of a direct bone-implant contact, have been proposed and have shown encouraging results.14 In general, two different approaches have emerged. The first approach involves placing several more implants than the usual treatment plan for a conventional healing period (Figure 35-2, A). Selected implants around the arch (three or more) then are loaded immediately with a transitional prosthesis (Figure 35-2, B). Enough implants are left submerged for a regular healing period to allow delivery of a fixed prosthesis, even if all immediately loaded implants fail (Figure 35-2, C). If any of the implants survive, then they also are used in the final restoration. This protocol was first published by Schnitman et al.15 in 1990, using 28 screw-shaped implants in 10 mandibles to support a fixed transitional prosthesis. Schnitman et al.16 suggested that this procedure be used only in the completely edentulous mandible, where moderate to abundant bone was present posterior and anterior to the mental foramens. Using this approach, 100% of the submerged unloaded implant survived, whereas three immediately loaded implants failed before 6 months and one implant failed 18 months after surgery (84% survival) over 9 years.
In 1997, Tarnow et al.17 reported on immediate loading with a fixed prosthesis, using threaded implants in 10 consecutive completely edentulous cases over 5 years. Sixty-six of 69 implants were integrated in six mandibular and four maxillary completely edentulous arches (96% survival) using a total of 10 to 13 implants in each arch for the final prosthesis. Like Schnitman et al.,15,16 Tarnow et al. did not immediately load all the implants for the transitional prosthesis.
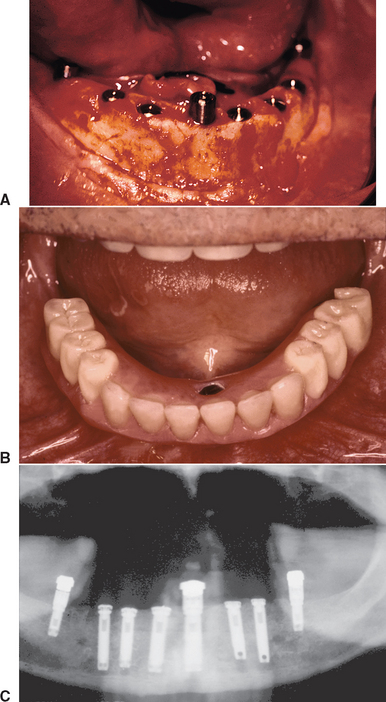
The other protocol for immediate occlusal loading of dental implants initially loads all of the implants inserted. The implants are splinted together, which decreases the stresses on all the developing interfaces and increases the stability, retention, and strength of the transitional prosthesis during the initial healing phase (Figure 35-3). Often additional implants also are used with this technique compared with the traditional healing method.18–22
Rationale for Implant Immediate Loading
Surgical Trauma
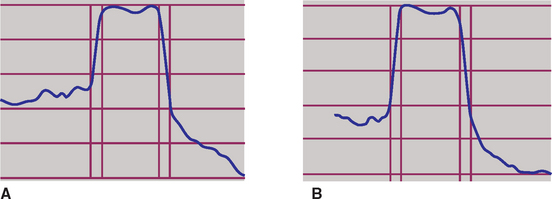
The immediate implant-loading concept challenges the conventional healing time of 3 to 6 months of no loading before the restoration of the implant. Often the risks of this procedure are perceived to be during the first week after the implant insertion surgery. In reality, the bone interface is stronger on the day of implant placement compared with 3 months later23 (Figure 35-4).
The surgical process of the implant osteotomy preparation and implant insertion cause a regional acceleratory phenomenon of bone repair around the implant interface.24 As a result of the surgical placement, organized, mineralized lamellar bone in the preparation site becomes unorganized, less-mineralized, woven bone of repair next to the implant.25 The implant-bone interface is weakest and most at risk of overload at 3 to 6 weeks after surgical insertion because the surgical trauma causes bone remodeling at the interface that is least mineralized and unorganized during this time frame. A clinical report by Buchs et al.26 found immediately loaded implant failure occurred primarily between 3 and 5 weeks after implant insertion from mobility without infection. At 4 months the bone is still only 60% mineralized, organized lamellar bone.27 However, this has proved to be sufficient in most bone types and clinical situations for two-stage healing and delayed implant loading.
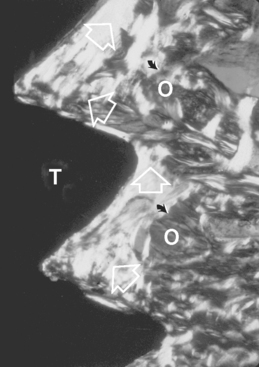
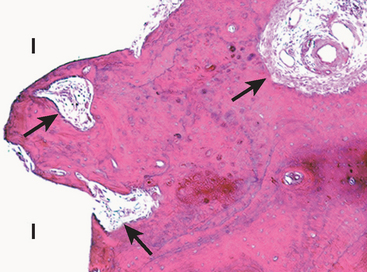
One method to decrease the risk of immediate occlusal overload is to decrease the surgical trauma and amount of initial bone remodeling at implant placement. Roberts et al.28 reported a devital zone of bone for 1 mm or more around the implant as a result of the surgical trauma (Figure 35-5). Causes of trauma include thermal injury and microfracture of bone during implant placement. Excessive surgical trauma and thermal injury may lead to osteonecrosis and result in fibrous encapsulation around the implant.29 Eriksson and Albrektsson30 have reported bone cell death at temperatures as low as 40° C. Sharawy et al.31 have shown that the amount of heat generated in the bone next to the implant drills depends on the drill design and speed. The temperature next to the drill ranged from 38° C to more than 41° C, from a baseline of 37° C, and required 34 to 58 seconds to return to baseline. In this report, the two implant drill systems with internal-cooled drills cut at a higher temperature than the two systems tested with external irrigation. Other factors related to heat generated to bone include the amount of bone prepared, drill sharpness, depth of the osteotomy, and variation in cortical thickness.32,33
A self-tapping implant causes greater bone remodeling (woven bone) around the implant during initial healing compared with a bone tap and implant placement technique.34 The implant should be nonmobile on insertion, but excess strain from additional torque also may increase the risk of microdamage at the interface. The dentist may believe a protocol for immediate load is to tighten the implant within the bone to 45 to 60 N-cm. Although this concept helps ensure that the implant has rigid fixation and is in good-quality bone, the additional torque used to secure or evaluate fixation of an implant in bone actually may increase the strain at the interface and therefore increase the amount of remodeling, which decreases the strength of the bone-implant interface. Therefore it is prudent to minimize factors related to thermal injury and surgical trauma when considering immediate load to the implant interface.
Bone-Loading Trauma
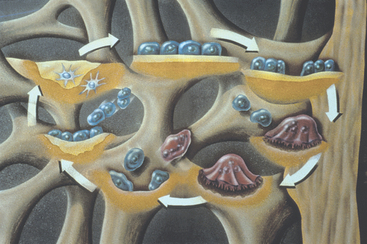
Cortical and trabecular bone may be modified by modeling or remodeling.25 Remodeling, or bone turnover, permits the repair of bone after trauma or allows the bone to respond to its local mechanical environment. The bone most often is lamellar but may become woven bone during the repair or remodeling process, so the bone may respond more rapidly to the current situation. Lamellar bone and woven bone are the primary bone tissue types found around a dental implant. Lamellar bone is organized, highly mineralized, is the strongest bone type, has the highest modulus of elasticity, and is called load-bearing bone. By comparison, woven bone is unorganized, less mineralized, weaker, and more flexible (lower modulus of elasticity). Woven bone may form at a rate of 60 μm per day, whereas lamellar bone forms at a rate of 1 to 5 μm per day.27 The classic two-stage surgical approach to implant dentistry permitted the surgical repair of the implant to be separated from the early loading response by 3 to 6 months. Therefore the majority of the woven bone that formed to repair the initial surgical trauma was replaced with lamellar bone. Lamellar bone is stronger and able to respond to the mechanical environment of occlusal loading. Therefore a rationale for immediate loading is not only to reduce the risk of fibrous tissue formation (which results in clinical failure) but also to minimize woven bone formation and promote lamellar bone maturation to sustain occlusal load. The woven bone of surgical trauma has been called repair bone, and the woven bone formed from the mechanical response may be called reactive woven bone.35 Remodeling also is called bone turnover and not only repairs damaged bone but also allows the implant interface to adapt to its biomechanical situation (Figure 35-6). The interface-remodeling rate is the period of time for bone at the implant interface to be replaced with new bone. Once the bone is loaded by the implant prosthesis, the interface begins to remodel again. However, this time the trigger for this process is strain, rather than the trauma of implant placement. Strain is defined as the change in length of a material divided by the original length and is measured as the percentage of change. When the surgical trauma is too great or the mechanical situation is too severe, fibrous tissue may form rather than bone. Fibrous tissue at an implant interface may result with clinical mobility rather than rigid fixation.
Histologic Evaluation: Short Term
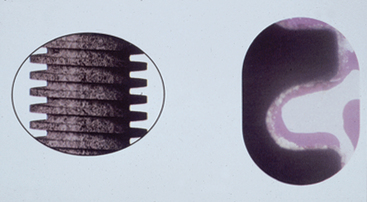
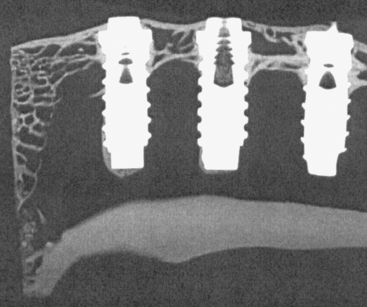
General agreement is that excess stresses to an implant interface may cause overload and implant failure. However, immediate loading of an implant does not necessarily result in excessive stresses. The initial histologic response of bone at the implant interface has been evaluated on immediately loaded implants. A direct bone-implant contact with favorable bone quality around the implants is reported. For example, Romanos et al.36 demonstrated no statistical difference between immediate- and delayed-loaded implants. Sharawy37 evaluated the immediate- versus delayed-healing interface of 20 BioHorizons dental implants in five adult beagle dogs (Figure 35-7). All implants were inserted into premolar grafted bone defect sites. The implants were paired, so one half the implants were submerged and the adjacent implants received an abutment and were subjected to immediate function for 4 weeks. The implants then were evaluated with histometric analyses of plastic embedded calcified sections. No statistically significant difference (p > 0.05) was found in the bone-implant contact ratios between the submerged and loaded implants (Figure 35-8). Similarly, the volume fractions of the interface bone were not significantly different. The bone next to the implants appeared mature and showed evidence of remodeling.
Suzuki et al.38 performed a clinical and histological evaluation of immediate-loaded posterior implants in nonhuman primates. After loading 10 implants for 90 days, they were compared with 5 control implants with no loading. The bone-implant contact percent (BIC) ranged from 50.3% to 64.1%, with an average of 56.3% for the controls. The immediate-loaded group had one implant failure, seven implants with an average of 67.6% BIC, and two implants with 43.2% and 45.6% BIC, respectively. Therefore the study demonstrated immediate-loaded implants may have a higher BIC than nonloaded implants, most likely a response to the strain conditions in the bone. However, three implants had less BIC or failure when compared with controls. Although benefits exist related to immediate loading, it appears some risks are involved in the procedure.
Testori et al.39 reported on the histologic interface of two implants in human beings that were immediately loaded after 4 months. The bone contact ranged from 78% to 85%, with no epithelial migration. Therefore immediately loading an implant interface apparently does not necessarily place the interface at increased risk of fibrous tissue formation.
Histologic Evaluation: Long Term
Piatelli et al.40 evaluated bone reactions and the bone and titanium interface in early loaded implants in monkeys compared with unloaded implants in the same arch several months after immediate loading. No statistically significant differences were detected in the BIC after 8 months. However, loaded implants had less marrow spaces and more compact bone. A later study by the same group demonstrated greater bone contact in immediately loaded implants at 9 months.41 No fibrous tissue was found at the interface. After 15 months, unloaded and immediately loaded implants were compared, and loaded implants exhibited greater (almost twice) direct bone contact at the interface. In particular, early loaded screws demonstrated thicker lamellar and cortical bone than unloaded implants.42 This finding suggests that early occlusal loading may enhance bone remodeling and further increase bone density.
Randow et al.43 evaluated the bone interface in a human patient after 18 months in an immediate-loading situation. They noted a direct bone-implant interface. Ledermann44 confirmed these results in a 95-year-old patient who had an immediately loaded, bar-connected overdenture in function for 12 years (Figure 35-9). Thus a long-lasting direct bone-implant contact relationship appears possible.
Immediate Occlusal Loading: Factors That Decrease Risks
Bone Microstrain
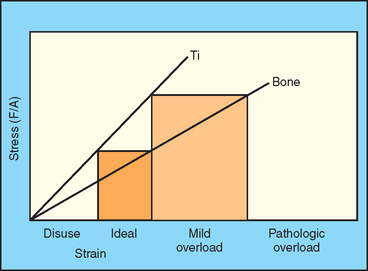
Loaded bone changes its shape. This change may be measured as strain. Microstrain conditions 100 times less than the ultimate strength of bone may trigger a cellular response. Frost45 has developed a microstrain language for bone based on its biological response at different microstrain levels (Figure 35-10). Bone fractures at 10,000 to 20,000 microstrain (μ ) units (1% to 2% strain). However, at levels of 20% to 40% of this value, bone already starts to disappear or form fibrous tissue and is called the pathologic overload zone. The ideal microstrain for bone is called the physiologic or adapted zone. The remodeling rate of the bone in the jaws of a dentate canine or human being that is in the physiologic zone is about 40% each year.46 At these levels of strain, the bone is allowed to remodel and remain an organized, mineralized lamellar bone. This is called the ideal load-bearing zone for an implant interface. The mild overload zone corresponds to an intermediate level of microstrain between the ideal load-bearing zone and pathologic overload. In this strain region, bone begins a healing process to repair microfractures, which are often caused by fatigue. Histologically, the bone in this range is called reactive woven bone. Rather than the surgical trauma causing this accelerated bone repair, the microstrain causes the trauma from overload. In either condition, the bone is less mineralized, less organized, weaker, and has a lower modulus of elasticity.
) units (1% to 2% strain). However, at levels of 20% to 40% of this value, bone already starts to disappear or form fibrous tissue and is called the pathologic overload zone. The ideal microstrain for bone is called the physiologic or adapted zone. The remodeling rate of the bone in the jaws of a dentate canine or human being that is in the physiologic zone is about 40% each year.46 At these levels of strain, the bone is allowed to remodel and remain an organized, mineralized lamellar bone. This is called the ideal load-bearing zone for an implant interface. The mild overload zone corresponds to an intermediate level of microstrain between the ideal load-bearing zone and pathologic overload. In this strain region, bone begins a healing process to repair microfractures, which are often caused by fatigue. Histologically, the bone in this range is called reactive woven bone. Rather than the surgical trauma causing this accelerated bone repair, the microstrain causes the trauma from overload. In either condition, the bone is less mineralized, less organized, weaker, and has a lower modulus of elasticity.
When strain is placed on the horizontal axis and stress is positioned on the vertical axis, the relationship between these two mechanical indexes results in the flexibility or modulus of elasticity of a material. Therefore the modulus conveys the amount of deformation in a material (strain) for a given load (stress) level. The lower the stress applied to the bone (force divided by the functional surface area that receives the load), the lower the microstrain in the bone (Figure 35-11). Therefore one method to decrease microstrain and the remodeling rate in bone is to provide conditions that increase functional surface area to the implant-bone interface.47 The surface area of load may be increased in a number of ways: implant number, implant size, implant design, and implant body surface conditions. The force to the prosthesis also is related to the strain and may be altered in magnitude, duration, direction, or type. Methods that affect the amount of force include patient conditions, implant position, and direction of occlusal load.
Increased Surface Area
Implant Number
The dentist may increase the functional surface area of occlusal load at an implant interface by increasing implant number.48 Therefore rather than three to five implants to support a fixed restoration, use of additional implants when immediate loading is planned is more prudent. Immediate-loading reports in the literature with the lowest percentage survival correspond to fewer implants loaded.15,16,49 Alternatively, a clinical system has been developed specifically for immediate loading with a definitive prosthesis using only three mandibular implants. A 3-year study demonstrated 98% implant survival rates.50 However, if even one implant fails in this approach, then the patient treatment may require extended time for bone grafting and implant reinsertion before the fabrication of another fixed restoration.
When 10 to 13 implants were inserted and splinted together per arch, implant survival may be greater than 97%.19–22 The increased number of implants also increases the retention of the restoration and reduces the number of pontics (Figure 35-12, A). The increased retention minimizes the occurrence of partially unretained restorations during healing, which can overload the implants still supporting the restoration. The decrease in pontics may decrease the risk of fracture of the transitional prosthesis, which also may be a source of overload to the remaining implants supporting the prosthesis (Figure 35-12, B). More implants typically are used in the maxilla (eight to 12) compared with the mandible (five to nine) (Figure 35-13). This approach helps compensate for the less dense bone and increased directions of force often found in the upper arch.
Implant Size
The dentist also may increase the surface area of implant support by the size of the implant. The length of the implant in most systems increases in increments of 2 to 4 mm. Each 3-mm increase in length can improve surface area support by more than 20%.51 The benefit of increased length is not found at the crestal bone interface but rather in initial stability of the bone-implant interface. Most of the stresses to an implant-bone interface are concentrated at the crestal bone, so the increased implant length does little to decrease the stress that occurs at the transosteal region around the implant.52 Therefore length is not an effective method to decrease stress because it does not address the problem in the functional surface area region of the bone-implant interface. However, because the implant is loaded before the establishment of a histologic interface, implant length is more relevant for immediate-load applications, especially in softer bone types. The additional implant length also may permit the implant to engage the opposing cortical plate, which further increases implant initial stability.
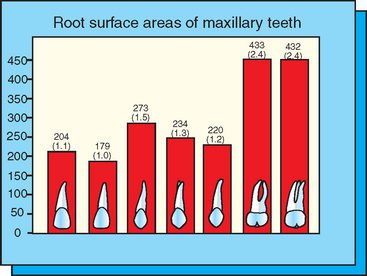
The functional surface area of each implant support system is related primarily to the width and the design of the implant. Wider root form implants provide a greater area of bone contact than narrow implants (of similar design). The crest of the ridge is where the occlusal stresses are greatest. As a result, width is more important than length of implant (once a minimum length has been obtained for initial fixation). Bone augmentation in width may be indicated to increase implant diameter when forces are greater, as in cases of moderate to severe parafunction. The major increase in tooth size occurs in the molar regions for natural teeth, where root surface area doubles compared with the rest of the dentition (Figure 35-14). Therefore implant diameter often is increased in the molar region. When a larger-diameter implant is not possible without additional augmentation surgery, more implants may be inserted (i.e., two for each molar), which also is a method to double the overall surface area in the posterior region.
Implant Body Design
The implant body design should be more specific for immediate loading because the bone has not had time to grow into recesses or undercuts in the design or attach to a surface condition before the application of occlusal load. For example, a press-fit implant with a cylinder design does not have bone integration the day of implant placement (Figure 35-15). An implant body with a series of horizontal plates that is tapped or pressed into place does not have bone present between the plates the day of surgical placement. Macrospheres on an implant surface do not have bone present within or around the porous surfaces of the implant the day of implant placement. In general, press-fit implants exhibit decreased initial stability and have an inherent design flaw for immediate-load applications. The surface condition of the implant, although vital for a cylinder implant, is less relevant during immediate-load protocols.
For a threaded implant, bone is present in the depth of the threads from the day of insertion. Therefore the functional surface area is greater during the immediate-load format. The number of the threads also affects the amount of area available to resist the forces during immediate loading. The greater the number of threads, the greater the functional surface area at the time of immediate load (Figure 35-16). Some threaded implants have a 1.2-mm distance between the threads (e.g., ITI dental implants), whereas others have a 0.4-mm distance (e.g., BioHorizons dental implants). The smaller the distance between the threads, the greater the thread number and corresponding surface area.52
The thread depth varies in implant design. The greater the thread depth, the greater the functional surface area for immediate-load application. The thread depth of some threaded implants is 0.2 mm, whereas the thread depth of other implant designs may reach 0.42 mm.52 Therefore one threaded implant may have more than two times the overall functional surface area compared with other implants of similar length and width (Figure 35-17).
The functional surface area of an implant body may affect the remodeling rate of bone during loading. A macrosphere implant with reduced surface area may have twice the remodeling rate of a typical threaded implant design. A square-threaded implant design, with deeper threads in greater number, is reported to have a tenfold reduction in remodeling rate under similar loading conditions and approximates 50% per year. The higher the remodeling rate, the weaker the bone interface. The teeth have a bone-remodeling rate of 40% per year, which maintains lamellar bone at the interface.47
The thread geometry also may affect the strength of early osseointegration and the bone-implant interface. Steigenga53 placed 72 implants into 12 rabbits and reverse-torque tested the unloaded implants after 12 weeks (Figure 35-18). One third of the implants had a V-thread, one third had a reverse buttress shape, and one third had a square thread. The number and depth of threads were the same, as were the width and length of each implant. The V and reverse buttress thread geometry yielded similar values for reverse-torque and BIC values. The square thread demonstrated statistically significantly higher values for both of these evaluations.
Implant thread design may affect the bone turnover rate (remodeling rate) during occlusal load conditions. For a V-shaped thread design, a tenfold greater shear force is applied to bone compared with a square thread shape.52 Bone is strongest to compression and weakest to shear loading.54 Compressive forces decrease the microstrain to bone compared with shear forces. Therefore the thread shape and implant design may decrease the early risks of immediate loading while the bone is repairing the surgical trauma.
A few clinical trials have compared immediate loading with different implant thread designs and tapered-implant bodies in the completely edentulous patient. The short-term clinical reports indicate a high success rate, regardless of implant design. As a result, overall shape and thread geometry apparently may not be the most important aspects for immediate occlusal load survival. Implant number, implant position, and patient factors most likely are more relevant components of success. Future studies in this area certainly are needed.
Implant Surface Conditions
Implant surface conditions may affect the rate of bone contact, lamellar bone formation, and the percentage of bone contact. The surface condition that allows bone formation in greatest percentage, higher BIC with higher mineralization rate, and fastest lamellar bone formation would be of benefit in immediate loading. These factors have been noted in delayed- and immediate-loading environments with hydroxyapatite (HA) coatings.55 HA also has been shown to decrease the remodeling rate during occlusal loading.56 Therefore if the bone is not of ideal density (D4) for immediate loading, then HA may decrease the risk of overload. However, Sullivan et al.57 found that peak insertion torque and resonance frequency at initial implant insertion were related to implant design rather than surface condition in type 2 and 3 bone. Sirota et al.58 compared various calcium phosphate (CaPO4)-coated implants to noncoated titanium plasma-sprayed (TPS) implants in immediate-load applications and found a high BIC after 30 days of functional loading in both groups when in good-quality bone. Evans et al.59 agree that HA-coated and noncoated screws have similar bone contact using a two-stage surgical approach in the mandible. The coating or surface condition of the implant is most beneficial during the initial healing and early loading conditions.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses