Chapter 3
Nonopioid analgesics, salicylates, NSAIDs, and corticosteroids for chronic pain
3.1 Introduction
While pain during dental restorative, endodontic, and surgical therapy is usually adequately controlled by local anesthesia, postoperative pain control usually requires anti-inflammatory and/or analgesic medications. These same medications are also used for pain and inflammation control with various chronic orofacial pain (OFP) disorders (e.g., osteoarthritis involving the temporomandibular joint [TMJ], trigeminal neuropathic pain disorders, and oral ulcerative disease or mucositis). The rationale for selection of analgesic and anti-inflammatory medications and the protocol for their use postoperatively will be quite different than when used to manage chronic-OFP patients. Using too much medication in either situation will lead to side effects such as gastritis, drowsiness, nausea, and vomiting (from opioids) and using too little medication causes suffering. If acute pain is not adequately suppressed during the immediate postoperative period, in some susceptible patients this may contribute to the conversion of nociceptive pain into neuropathic pain.1 Moreover, if a short-acting analgesic is used to suppress episodically occurring headache pain, this may lead to transformation of the headache into a chronic pain condition.2 The mechanisms underlying these conversions are discussed in detail in Chapters 15 and 17. While systemic corticosteriod use is covered here, the use of corticosteroids as an injectable agent for TMJ pain or for severe oral ulcerative lesions and inflammation is covered in Chapter 18. Topical use of corticosteriods for local mucosal tissue inflammation and ulcerations is covered in Chapter 12. Disease-modifying arthritic drugs (DMARDs), which also alter inflammation, are also discussed in Chapter 18. The primary focus of this chapter is twofold. First we review the use of 14 different medications (summarized in Table 3.1): nonopioid analgesics (acetaminophen, tramadol), salicylates (aspirin, diflunisal), nonselective nonsteroidal anti-inflammatory medications (ibuprofen, naproxen, ketoprofen, meclofenamate sodium, piroxicam, diclofenac, and nabumetone), and cyclooxygenase 2 (COX-2) selective nonsteroidal anti-inflammatory medications (celecoxib, meloxicam, and etodolac). At the end of the chapter we review two commonly used systemic corticosteroids (prednisone and methylprednisolone) as they are used for various chronic pain situations.
Table 3.1 Dosing information for common oral NOAs, salicylates, NSAIDs, and corticosteriods in the United States
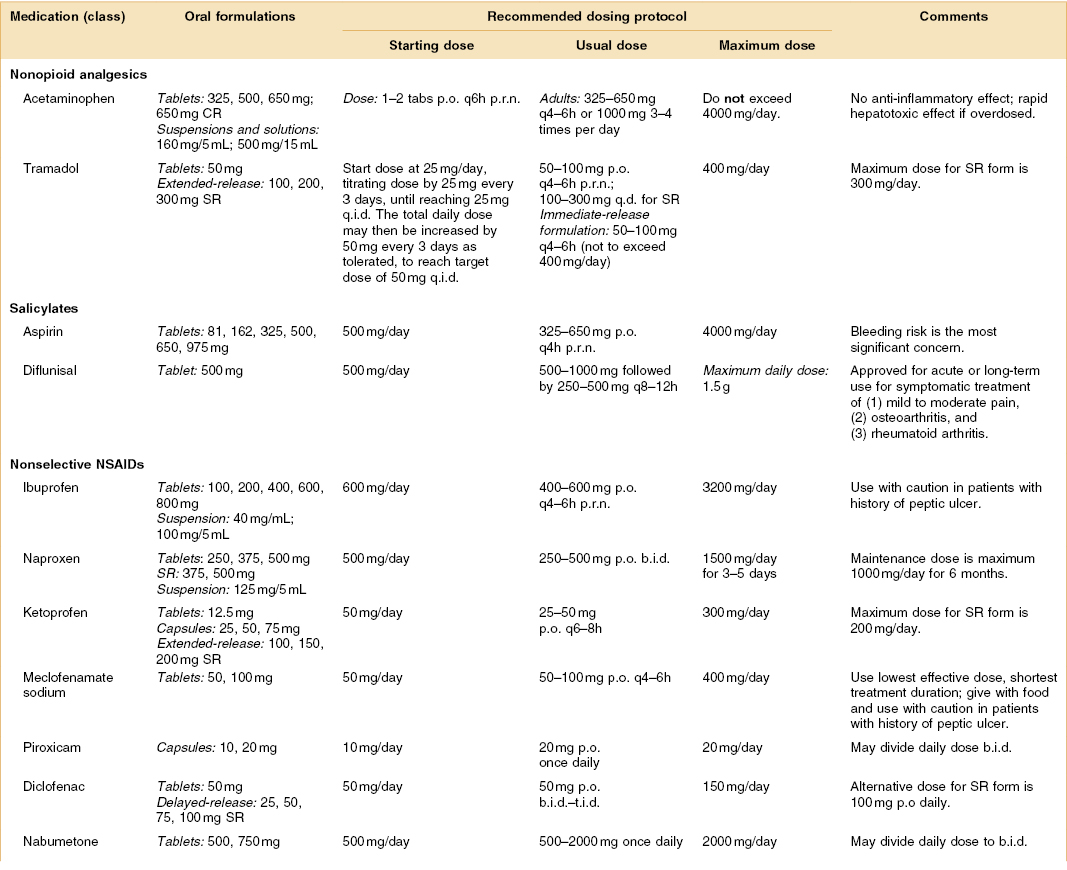

Dosing abbreviations: b.i.d., bis in die (a Latin phrase meaning “twice daily”); p.o., per os (a Latin phrase meaning “by mouth”); p.r.n., pro re nata (a Latin phrase commonly used in medicine to mean “as needed” or “as the situation arises”); q.i.d., quater in die (a Latin phrase meaning “four times daily”); q.d., quaque die (a Latin phrase meaning “every day”); qxh, every x hours (from quaque hora, a Latin phrase meaning “every hour”); t.i.d., ter in die (a Latin phrase meaning “three times daily”).
COX-2, cyclooxygenase-2; CR, controlled release; NOA, nonopioid analgesic; NSAIDs, nonsteroidal anti-inflammatory drugs; SR, sustained release.
3.2 Nonopioid Analgesics
Not unexpectedly, the World Health Organization recommends that, for most conditions where pain is the primary problem, nonopioid analgesics (NOAs) are the first-choice medications.3 In addition, and slightly less expected, is that the American College of Rheumatology (ACR) guidelines emphasize acetaminophen should be the first-line treatment for osteoarthritis of the hip and knee.4–6 Two drugs that are classified as nonopioid analgesics are acetaminophen and tramadol. Some authors would include nonsteroidal anti-inflammatory drugs (NSAIDs) in this grouping since they are nonopioid also, but in this chapter, we limit discussion of the NOAs to acetaminophen and tramadol.
3.2.A Acetaminophen
Indications
Acetaminophen is used for headaches, musculoskeletal pain, and almost all acute disease pains (e.g., pulpitis) and as a postoperative analgesic. This drug has a fast onset and short half-life and therefore also has value in a patient with episodic headaches, acute temporomandibular osteoarthritis pain, pain due to acute internal derangement, and any chronic pain that is undergoing an acute flare-up. Acetaminophen is also commonly prescribed for cancer pain management, and pain specialists often undertake combination therapy with multiple analgesics, including adjuvant pain analgesics (e.g., amitriptyline), during the treatment of severe, refractory pain.
Dosage
Acetaminophen has a rapid onset of action and a relatively short half-life. Acetaminophen comes in 325-, 500-, and 650-mg tablets and it is common to take one tablet every 4–6 hours; however, patients should be very careful not to exceed 4000 mg/day.
Adverse Effects
The biggest concern with the use of acetaminophen is liver toxicity. It is contraindicated in any patient with a pre-existing liver disease. Doses higher than 4 g daily may cause serious, irreversible hepatic toxicity, which can be fatal in some patients. Damage to the liver is not due to the drug itself but to a toxic metabolite (N-acetyl-p-benzoquinone imine) that is produced by cytochrome P450 enzymes in the liver. Under normal circumstances this metabolite is detoxified by conjugating with glutathione in a phase 2 reaction; however, when a patient takes too much acetaminophen, a large amount of toxic metabolite is generated that overwhelms the detoxification process and leads to rapid and devastating hepatotoxicity.
Efficacy for Chronic Pain
There are three chronic pain conditions (osteoarthritis, chronic musculoskeletal pain, and episodic headaches) for which acetaminophen might be recommended as a first-line therapy. The recent evidence is reviewed next for each condition. There is little or no evidence that acetaminophen is efficacious as a primary treatment for neuropathic chronic pain states, but it is often used combined with an opioid analgesic in neuropathic pain. Information on the efficacy of combined acetaminophen and opioids for chronic pain is provided in Chapter 4.
Osteoarthritis
As mentioned earlier, the ACR recommends that acetaminophen should be a primary treatment for pain associated with osteoarthritis. A 2006 study examined the safety of acetaminophen in adult patients with mild to moderate osteoarthritis in a large multicenter randomized controlled 6-month-long study.7 The dose used in the study was high (4 g/day) and it was administered for up to 12 months to 290 adult patients with osteoarthritis pain. This acetaminophen group was compared with 291 adult patients with osteoarthritis pain who were randomized to receive naproxen 750 mg/day. All subjects had liver and renal function assessments performed, as well as periodic physical examination. Both groups had a substantial dropout problem but no patient in either treatment group experienced hepatic failure, hepatic dysfunction, renal failure, or elevated serum creatinine levels. Two adverse events considered to be drug related and reported by more than 1% of patients were seen more frequently in the naproxen group than in the acetaminophen group: constipation (9.9% vs. 3.1%) and peripheral edema (3.9% vs. 1.0%). No adverse event reported in the acetaminophen group was considered both serious and related to study medication, but one subject in the naproxen group did develop gastrointestinal (GI) bleeding. The authors concluded that, with treatment under physician supervision, acetaminophen was found to be generally well tolerated for the treatment of osteoarthritis pain of the hip or knee for periods of up to 12 months. In contrast, a 2006 Cochrane database meta-analysis8 on the efficacy of acetaminophen for osteoarthritis reviewed 15 randomized controlled studies (5986 subjects) that compared acetaminophen with either placebo or an NSAID. In the placebo-controlled studies, the authors acknowledged that acetaminophen was superior to placebo but the magnitude of the effect was considered of questionable clinical significance since the relative percentage improvement from baseline was 5% or 4 points on a 0-to-100 scale and the calculated number needed to treat (NNT) ranged from 4 to 16, which is quite poor. For the studies that compared acetaminophen and NSAIDs, the authors reported that acetaminophen was less effective overall than NSAIDs in terms of reducing pain and improving functional status. Moreover, the authors noted that no significant difference was found overall between the safety of acetaminophen and NSAIDs, although patients taking traditional NSAIDs were more likely to experience an adverse GI event. This analysis concluded that the data suggests that NSAIDs are superior to acetaminophen for improving knee and hip pain in people with osteoarthritis.
Chronic Musculoskeletal Pain
Although acetaminophen is not considered a highly successful therapy for osteoarthritis, myofascial pain, or fibromyalgia and most experts consider it inadequate for this purpose, it has substantial merit in the elderly with chronic musculoskeletal pain. In the elderly, frequently NSAIDs and even opioids are contraindicated and the side effects of anticonvulsant medications are intolerable; therefore acetaminophen is elevated to a primary pain control medication. A 2009 study examined the efficacy of acetaminophen for pain control in elderly chronic musculoskeletal pain patients with dementia.9 The study enrolled community-dwelling elderly patients diagnosed with dementia, and it used a within-subjects repeated-measures A-B-A-B prospective design. The the patients were provided with a 1.3-g controlled-release (CR) formulation of acetaminophen three times a day (t.i.d.; every-8-hour dosing). The study used behavioral measures of pain and the authors concluded that, during both treatment phases, pain behaviors decreased in both frequency and duration relative to the control and baseline phases.
Episodic Headaches
Third, acetaminophen is used frequently for control of pain related to episodic headaches such as migraine and tension-type headache (see Chapter 15).10 However, the NSAIDs and nonopioid analgesics have an inherent risk when used frequently for headache control. A 2004 study examined the concept that chronic use of analgesics to manage a frequent episodic headache can cause medication overuse headache disorder.11 The study collected data from 114 consecutive patients (96 women and 18 men, with a mean age of 54.2 years) diagnosed with a chronic daily headache due to overuse of medications. All patients in this group had been referred for inpatient detoxification of their analgesic medications. The authors determined that, of these patients, 71% had an initial headache diagnosis of migraine without aura and, of these, 38.6% were overusing simple analgesics, which included both NSAIDs and acetaminophen.
3.2.B Tramadol
Indications
The other NOA we discuss is tramadol, which has value as a chronic pain medication but is rarely considered effective as a rapid-acting analgesic for acute pain. The enigma of tramadol is that it is categorized as a nonopioid analgesic yet it binds (weakly) to an opioid receptor. It is marketed as an analgesic without scheduling under the US Controlled Substances Act, even though recent literature has suggested classic opioid withdrawal occurs with discontinuation or dose reduction and there are increasing reports of abuse and dependence.12 Tramadol exhibits a combination of serotonin and norepinephrine reuptake inhibition (similar to what is seen in the tricyclic antidepressant drugs [TCAs]) and it is a weak μ-opioid agonist.13 The manufacturer thus claims that tramadol can reduce chronic pain by affecting both the ascending and descending pain pathways.
Dosage
The common titration method for prescribing tramadol is usually 1 tablet every 4 days to full therapeutic levels (or minimum of 50% pain relief). A typical maintenance dosage for fibromyalgia patients is 300–400 mg/day in three to four divided doses, concomitant with acetaminophen at 2–3 g/day in divided doses. Commonly for chronic pain, therapy begins with just one tablet at bedtime for 1–2 weeks since this usually reduces the side effects and allows progressive increase of the dosage after this.
Adverse Effects
Tramadol has the same set of side effects seen in TCAs (e.g., nausea and dizziness) and these side effects can be limiting at first in approximately 20% of patients. In addition, a review of idiopathic seizures in 11,383 patients showed that tramadol is not associated with a higher risk of seizure activity when compared with other analgesics.14
Efficacy for Chronic Pain
Like acetaminophen, tramadol is used for several types of chronic pain, including osteoarthritis and chronic musculoskeletal pain. It is not typically recommended for episodic headaches since it is usually too slow to achieve reasonable pain suppression. It does have a place in the control of neuropathic pain and other non-neoplastic chronic pain disorders.
Osteoarthritis
Painful degenerative joint disease of the knee responded to tramadol therapy in a two-phase trial of 129 patients with significantly improved pain intensity and pain relief scores compared with the placebo group.15 Another osteoarthritis two-phase trial showed that patients given tramadol can significantly reduce their intake of naproxen without compromising pain relief.16
Chronic Musculoskeletal Pain
Three controlled studies have evaluated the efficacy of tramadol in fibromyalgia. The first small study used a double-blind crossover design to compare single-dose intravenous tramadol 100 mg with placebo in 12 patients with fibromyalgia.17 The authors reported that fibromyalgia patients receiving tramadol experienced a 20.6% reduction in pain compared with an increase of 19.8% of pain in the placebo group. In a two-phase study in 2000, tramadol was again shown to reduce the impact of pain in fibromyalgia patients.18 There was an initial 3-week, open-label phase of tramadol 50–400 mg/day followed by a 6-week double-blind phase in which only patients who tolerated tramadol and perceived benefit were enrolled. The results showed that more patients on tramadol than placebo tolerated the drug and achieved adequate pain relief in the double-blind phase. In 2003 a third study found that tramadol, in combination with acetaminophen, provided a substantial additive effect for pain reduction in fibromyalgia.19 The randomized, controlled, double-blind trial (RCBT) examined the efficacy of tramadol (37.5 mg) combined with acetaminophen (325 mg) in 315 patients with fibromyalgia and found that patients taking tramadol and acetaminophen (4±1.8 tablets per day) were significantly more likely than placebo-treated subjects to continue treatment and experience an improvement in pain and physical function. Treatment-related adverse events were reported by significantly more patients in the tramadol/acetaminophen group (75.6%) than the placebo group (55.8%).
Neuropathic and Other Nonmalignant Chronic Pain Disorders
Two studies examined tramadol in painful diabetic polyneuropathy20 and painful polyneuropathy of different etiologies.21 Both studies found tramadol was superior to placebo and exhibited an NNT of 3.1 and 4.3, respectively. In addition to its relief of ongoing pain, it reduced touch-evoked pain and experimentally induced mechanical hyperalgesia. Finally, a comparison of tramadol and morphine in 25 patients with severe chronic pancreatitis pain showed that tramadol interferes significantly less with GI function.22
3.3 Nonsteroidal Anti-Inflammatory Medications
In this category are the salicylates and the various nonsteroidal anti-inflammatory drugs (NSAIDs). The NSAIDs are usually subcategorized into those that nonselectively inhibit and those that selectively inhibit cyclooxygenase (COX) enzyme 2. Nonselective or nonspecific COX inhibition means the NSAID exhibits inhibitory effects on both COX-1 and COX-2 enzymes, while the selective COX inhibition action usually means that the NSAID inhibits COX-2 enzyme only. We discuss salicylates first and then describe the various NSAIDs. The latter are the mainstay of therapy for the management of acute dental disease (e.g., pulpal abscess) and postoperative-related dental pain that occurs following surgical and endodontic procedures. When used as directed, nonprescription (i.e., over-the-counter) dosing regimens for ibuprofen (200 mg four times per day [q.i.d.]), ketoprofen (75 mg t.i.d), or naproxen sodium (220 mg two times per day [b.i.d.]) are both safe and effective for most patients across a wide variety of dental pain conditions.23
3.3.A Salicylates
We review two medications in the salicylate category: aspirin and diflunisal. Salicylates have been used in medicine (as willow bark) as an analgesic since 1763. The active agent in willow bark, salicin, was eventually used to produce salicyclic acid in 1838 and this led to the production of a substance with known chemical purity and properties. Aspirin itself was introduced in 1899 and since then has been widely used for pain control and reduction of fever and swelling. The mechanism of action of salicylates has since been identified—the inhibition of prostaglandin synthesis, which is critical to blocking the initial oxygenation of arachidonic acid by cyclooxygenase enzyme.
Aspirin
Indications
Aspirin is a weak acid that is well absorbed from the GI tract when taken orally. Its ability to dissociate favors absorption from the stomach, but it is principally absorbed from the small intestine because of the greater surface area. Acetylsalicylic acid, or aspirin, is rapidly metabolized to salicylic acid by plasma and gastric esterases. Salicylate, an active form of aspirin, is widely distributed in the body, is metabolized mainly in the liver by conjugation, and is excreted in the urine mostly as salicyluric acid. Although the efficacy of aspirin has been accepted for several generations based on over 100 years of clinical use, it is only in the past 20 years that controlled studies have documented its efficacy for dental pain. Through its inhibition of prostaglandin synthesis, aspirin has also been shown to affect platelet function. This may result in prolonged bleeding time when aspirin is used postsurgically. Aspirin should not be given to patients with liver disease, hypothrombinemia, hemophilia, or vitamin K deficiency. It should also be avoided in patients who are taking anticoagulant drugs. Allergic reactions to aspirin are uncommon but are more frequently seen in persons with asthma, nasal polyps, or a history of an allergic reaction to other aspirin-like drugs (including the NSAIDs). Aspirin interactions with insulin or oral hypoglycemic agents may result in a greater hypoglycemic effect; an alternative nonopioid analgesic should be considered in patients taking one of these agents. The relationship between plasma levels and therapeutic effect is not direct and no fixed dose, schedule, or dosage form will provide the desired result in all patients.
Dosage
The maximum recommended dose is 650 mg every 4 hours (3900 mg/day) or 500–1000 mg every 4 hours, up to a maximum of 4000 mg/day.
Adverse Effects
Aspirin has several side effects that are frequently the reasons for using other nonopioids in its place, and it is not suggested for prolonged use in a chronic pain population. The most commonly reported side effects are epigastric distress, nausea, ulceration, and, less frequently, vomiting. Aspirin-induced GI injury results from two known mechanisms. Local irritation of the mucosal lining allows diffusion of acid into the mucosa, with subsequent tissue damage. In addition, gastric prostaglandins that inhibit secretion of acid and promote secretion of cytoprotective mucus are inhibited by aspirin. For this reason, aspirin is contraindicated for patients with GI ulcers.
Efficacy for Chronic Pain
Aspirin is not logically used as a primary treatment in any of the chronic pain disorders (osteoarthritis, chronic musculoskeletal pain, or neuropathic pain) since gastric damage is so common with prolonged use. Unfortunately, frequent-headache patients do consume aspirin almost daily. A 2009 study reported on the patterns of medication use among those with chronic daily headache (n = 206) versus episodic headache (n = 507) in the general population.24 Questions about analgesic use revealed that chronic-daily-headache (CDH) sufferers were more likely to use over-the-counter and caffeine-containing products, triptans, opioid compounds, and prescription pain medications. However, based on the data the authors reported that aspirin and ibuprofen were negatively associated with CDH (OR = 0.5 and 0.7) but opioids were positively associated with CDH (OR = 2.3). These data suggest that, with CDH, aspirin was not an effective medication for severe frequent headaches and the salicylates do not contribute greatly to the transformation of episodic to chronic daily headache.
Diflunisal
Indications
Diflunisal is a salicylic acid derivative [5-(2,4-difluorophenyl)salicylic acid] that is more effective than aspirin as an analgesic.
Dosage
The recommended diflunisal dosage for most people with mild to moderate pain is 1000 mg, followed by a dose of 500 mg every 12 hours. Some people may need to take diflunisal (Dolobid) every 8 hours in order to achieve adequate pain relief.
Adverse Effects
Diflunisal has fewer GI and hematologic adverse effects than aspirin but, nevertheless, gastritis is the main complication of prolonged use.
Efficacy for Chronic Pain
There is very little information about the efficacy of diflunisal as a pain control agent in chronic pain conditions. No studies were available on its use in neuropathic pain or CDH, but an open trial with diflunisal (500 mg b.i.d.) in 766 outpatients with chronic back pain was published.25 These patients (mean age 41 years) had a variety of back pain disorders; outcomes were pain at rest and during exercise, the patient’s evaluation of the efficacy of the treatment, and the need for any supportive treatment. All side effects were recorded and those of the drug therapy were registered. In all diagnostic groups the relief of pain both at rest and during exercise was greater in patients receiving diflunisal than in the controls who received no drug therapy. The authors reported that taking the medication diminished the need for supportive physical therapy and the frequency of side effects was 8.6%, with 3% of the patients stopping the medication as a result.
3.4 Nonsteroidal Anti-Inflammatory Drugs
The wealth of data from clinical trials using NSAIDs makes them one of the most well-studied drug classes for acute inflammatory pain in ambulatory patients. The acute postoperative sequelae of dental procedures include other signs of inflammation due to tissue injury, most prominently edema. While synthetic analogs of endogenous corticosteroids are used extensively to control the sequelae of both acute and chronic inflammation, their use postoperatively is tempered by their ability to suppress the immune system thereby increasing the risk of infection. NSAIDs have a more selective mechanism of action than glucocorticoids and a more favorable side-effect profile, suggesting that drugs of this class may inhibit inflammation without the risks of corticosteroid administration. When considering the use of NSAIDs for prolonged use in chronic nonmalignant pain and cancer pain, there are several issues to consider.26 For example, in patients with a history of peptic ulcer disease, advanced age (>60 years of age), and female gender, concurrent corticosteroid therapy should be considered before NSAID administration to prevent upper GI tract bleeding and perforation. When NSAIDs are administered in a peptic ulcer risk group, proton pump inhibitors are usually added to the therapeutic mix to try to prevent GI side effects induced by NSAIDs. NSAIDs should be prescribed with caution in patients having compromised fluid status, interstitial nephritis, concomitant administration of other nephrotoxic drugs, and renally excreted chemotherapy in order to prevent renal toxicities. A meta-analysis of 16 controlled studies suggests that users of NSAIDs have a threefold greater risk of developing serious adverse GI events than nonusers and that this risk is greater for those over 60 years of age.27 NSAIDs alter kidney blood flow by interfering with the synthesis of prostaglandins in the kidney that are involved in the autoregulation of blood flow and glomerular filtration.28 The inhibitory effects of NSAIDs on kidney prostaglandin production lead to acute, reversible kidney failure in 0.5–1% of patients who take NSAIDs chronically.29 The most significant kidney-related side effect of NSAIDs is hemodynamically mediated acute kidney failure, which occurs in persons with pre-existing reduced kidney blood perfusion. A retrospective analysis of patients with end-stage kidney disease requiring hemodialysis demonstrated an association between chronic NSAID use (more than 5000 pills over a lifetime) and a ninefold increased risk of end-stage kidney disease.30 Finally, the efficacy of systemic NSAIDs has been examined in several Cochrane reviews of various mixed but chronic musculoskeletal pain conditions.31 These reviews have generally concluded that systemic NSAIDs are not effective as monotherapy for chronic pain. However, the toxicity associated with chronic high-dose NSAID administration is well documented, suggesting the need to carefully weigh the benefit-to-risk relationship for each therapeutic indication. The clinical pharmacology of NSAIDs is based in large part on studies performed in the oral surgery model.32 With regard to NSAIDs for chronic orofacial pain, the data is mixed. A review article that examined the primary literature suggests that daily use of nonopioid analgesics offers benefit for chronic orofacial pain.33 In contrast, the results of a placebo-controlled study suggest that NSAIDs are ineffective for chronic myogenous orofacial pain.34 This study examined the analgesic effects of ibuprofen, 2400 mg/day for 4 weeks, and found it could not be separated from placebo in a group of patients with chronic orofacial pain characterized as myogenic in origin.
3.4.A Nonselective Cyclooxygenase Inhibitory Medications
Research into the pathophysiology of inflammatory pain led to recognition that there are at least several forms of the cyclooxygenase enzyme responsible for the formation of products of the arachidonic acid cascade. One form, characterized as COX-1, is responsible for the normal homeostatic functions of prostaglandins in the GI tract that maintain GI mucosa integrity, initiate platelet aggregation, and regulate renal blood flow. The other form, COX-2, was initially thought to be induced only during inflammation and to contribute to the pain, edema, and tissue destruction associated with acute inflammation, rheumatoid arthritis, and osteoarthritis. Most of the NSAIDs are nonselective inhibitors of the COX enzymes (see, e.g., Table 3.1). This inhibition makes them slightly less likely to cause gastric disease than the salicylates, but gastric disease is still the main contraindication and the most frequent adverse event to occur with long-term use of NSAIDs. Next we review nine of the common nonspecific COX inhibitory NSAIDs used for pain and inflammatory suppression.
Ibuprofen
Indications
Ibuprofen, a propionic acid derivative, is the prototype of the NSAID class of analgesics and was first introduced into clinical practice in the United States in 1974. It is particularly useful for conditions in which aspirin or acetaminophen does not result in adequate pain re/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


