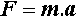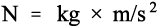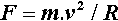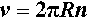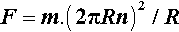Casting
The process of the casting of a fluid material into a mould clearly depends on the viscosity of that fluid. But if the demands include the accurate reproduction of the details of the mould, and overall dimensional accuracy, additional factors must be taken into account.
In the case of gypsum and investment products, the flow itself is affected by the special rheological properties of slurries, while conforming closely to the mould or pattern requires that the surfaces be wetted. Wetting is not possible with molten metals on ceramics and extra measures must be taken to obtain a good result.
In essence, accurate metal casting depends on supplying just sufficient casting pressure. This may be obtained hydrostatically, assisted by centrifugation, pressurized air, or the use of vacuum. However, excessive casting pressure is detrimental. Obtaining the correct balance depends on the understanding and fine control of the various factors.
A variety of defects can arise in the casting of metal, in part from the risks associated with the challenging conditions of high temperature and high speed. Faulty castings are not just a waste of time and money, but may also be detrimental to the life of the device if it is pressed into service. This is a treatment failure.
Casting is the final critical stage in the construction of complex metal prosthetic and restorative dental devices. A variety of factors interact with each other to affect the outcome, and fine control of the process requires a proper understanding.
Casting is the process of pouring a fluid material into a mould where, by some process (physical or chemical), that material hardens to produce a replica of an original pattern. The mould necessarily is a ‘negative’ or reversed three-dimensional image of the shape of the pattern. In the course of restoring tooth form for function, or in the provision of a larger scale prosthesis, both the reproduction of the shape of tissues for working models as well as the creation of the final functional device rely on casting in one sense or another. In the long chain of steps leading to the fabrication of a final device, the casting process itself is at least as important as the materials that are used for the attainment of dimensional accuracy and completeness of structure.
We are drawing a distinction here between systems in which flow is spontaneous (or, at least, occurring at a rate great enough to be useful) and those that require some assistance to flow in the form of a raised stress. Examples of the latter are commonplace in dentistry: all cements, denture base acrylic dough, most impression materials such as silicone and polysulphide rubbers, amalgam, filled resin restorative materials, waxes … the list goes on. Making such a material conform to the shape of some surface is described as moulding. A little thought will show that the distinction is rather artificial, there being no sharp, true demarcation but rather a continuum again. It is more a case of the timescale on which the desired effect operates that determines whether, and how much, force is to be used. Indeed, all of these processes involve flow, and flow is time-dependent.
§1 Models and Investments
Dental plaster and artificial stone models and dies are prepared by casting a slurry of hemihydrate and water into the impression material mould. Several factors are of direct importance in controlling the accuracy of the replica produced. Firstly, the slurry must be sufficiently fluid that it may flow into all of the fine details of the mould completely. This is the purpose of the mixing water and the inclusion in it of a proportion which we called the dilution allowance (2§2), the particle shape and size distribution playing a part in controlling the viscosity. Coupled with this is the effect of the surface tension of the slurry: it must not be great enough to cause rounding of sharp corners by preventing penetration. Ordinarily no special steps are taken to reduce this as, in such aqueous mixtures, the surface tension is apparently low enough for few problems to arise. But in this context the wetting of the mould material by the slurry is the primary consideration, and so the penetration coefficient (10§2.4) becomes relevant here (as in fissure sealants) when fine detail is required. Aqueous gel impression materials, agar and alginate, naturally offer least difficulty but some silicone materials, being hydrophobic, can cause problems. Accordingly, polymers have been designed to be ‘hydrophilic’, meaning wetted by water, to be used in these systems with a lower risk of trapping air bubbles in critical places.
•1.1 Vibration
Even so, and with all impression materials, care must be taken to avoid the entrapment of air bubbles in fine detail or narrow spaces. To this end vibration is routinely employed to aid the displacement of bubbles, which must be expected to be present as a matter of course. Firstly, this is because mixing by hand cannot avoid the inclusion of bubbles in the mix, and mixing under reduced pressure ultimately only reduces the size of those bubbles. Secondly, in pouring viscous media it is very difficult to avoid creating bubbles, whether due to trapping air against a wall or by ‘folding’ into the liquid.
Vibration both improves the flow of the slurry and eliminates bubbles from within its bulk that were incorporated at the mixing stage. This depends on the phenomenon of shear-thinning (4§7.9): the solid particles are prevented from mechanically interlocking with each other by the vibration supplying the activation energy for relative movement. The same phenomenon can be observed in dry powders. Tilting a container of dry powder such as plaster of Paris may require a rather large angle before the powder moves (and then it collapses unhelpfully dramatically). However, tapping gently permits more controlled and gradual flow. This effect is used to good advantage when weighing powders accurately from a spatula: tapping (or a vibrating spatula) may help dispensing small increments with greater control. Even if a mixture is already fluid enough to flow spontaneously, vibration will reduce the viscosity. But bear in mind that, for gypsum products and the like, we wish to work as close to the minimum mixing liquid as we can in order to maximize strength. Shear-thinning can allow us to get closer to that ideal.
It does not require bubbles to be trapped at a critical surface for them to be detrimental. Even if a bubble were to be entirely surrounded by slurry, if it were to end up sufficiently close to the surface of the set material the thin wall may easily be broken in subsequent handling, causing a deficiency of possible later concern, such as at a cusp or fitting surface. In any case, the setting rate of the gypsum product, or whatever it is being cast, must not be so great as to compromise the above considerations.
•1.2 Investing
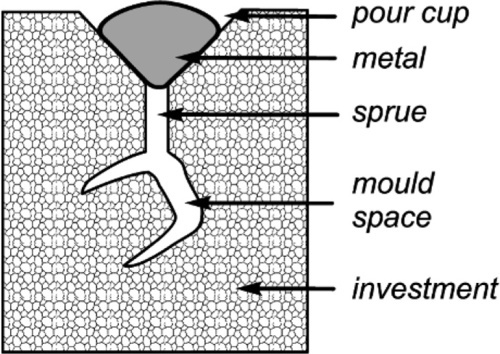
The production of an investment mould for metal casting is itself also a casting operation: the investment slurry is cast into the space between the wax pattern and the investment ring. Similar criteria as for pouring models must therefore apply for accuracy to be obtained. But, as the wetting of wax by water or watery mixtures is very poor, waxes necessarily having low energy surfaces, surface active agents (i.e. detergents or surfactants, 10§8.3) are generally applied to the surface of the pattern to avoid incomplete wetting and the trapping of air bubbles. The presence of sub-surface air bubbles is again of concern because the investment may break down under the impact of the molten metal (§3.8). Again, air may be eliminated by vibration, although care must be taken to avoid bubbles coming to rest underneath the pattern, so the proper orientation of the pattern on the sprue is crucial.
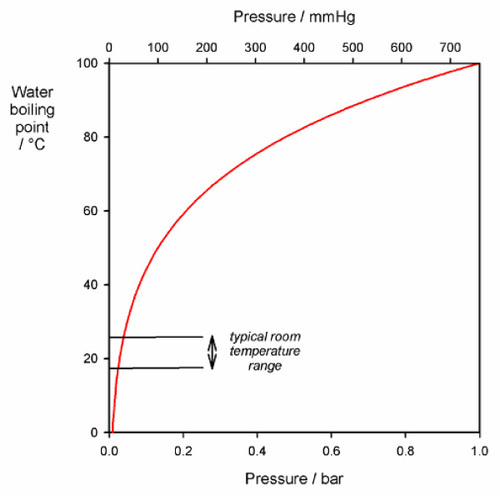
Even so, the application of vacuum (or, rather, reduced pressure) is a more reliable adjunct procedure. The reduction of pressure means that any bubbles that are present must expand (Boyle’s Law: PV = constant). This means that their total buoyancy, the force acting to lift the bubble through the slurry, is increased. The buoyancy is simply the weight (mass × gravitational acceleration) of the slurry displaced by the bubble (we can ignore the weight of the contained air). If we were to reduce the pressure to, say, 25 mm Hg, the volume, and therefore the buoyancy, of a bubble would be increased about 30 times (normal atmospheric pressure being about 760 mm Hg). Under such conditions bubbles tend to be expelled readily. Any bubbles remaining, or fragments left behind from the other escaping bubbles, will then collapse to about one-thirtieth of their volume (or about one-third their diameter) when atmospheric pressure is restored. Notice that the pressure could not be lowered much further at normal room temperatures without the water in the slurry boiling, since its vapour pressure is then about 25 mm Hg (Fig. 1.1). However, if this could be tolerated, it would have the effect of quickly flushing out the air, the vapour bubbles then quickly collapsing completely so that an essentially bubble-free casting would be obtained. The original air bubbles would form good nuclei for that boiling.
The use of a duplicating material such as agar, as for example is used to prepare the refractory model for a cast metal framework, involves pouring the fluid sol over the master model. This therefore is also a casting process. In this application, too, bubbles must be avoided. Although shear-thinning is not required, and wetting of the gypsum model by an aqueous sol should not be a problem if there is no stray wax on its surface, careless work could easily create problems of this kind.
§2 Metal Casting
The casting of molten metal at high temperatures into an investment mould has similar requirements as the above two operations as regards flow and detail reproduction, but these are made more critical because of the high speed and temperature involved. However, the casting of metal has its own special problems.
•2.1 Surface tension
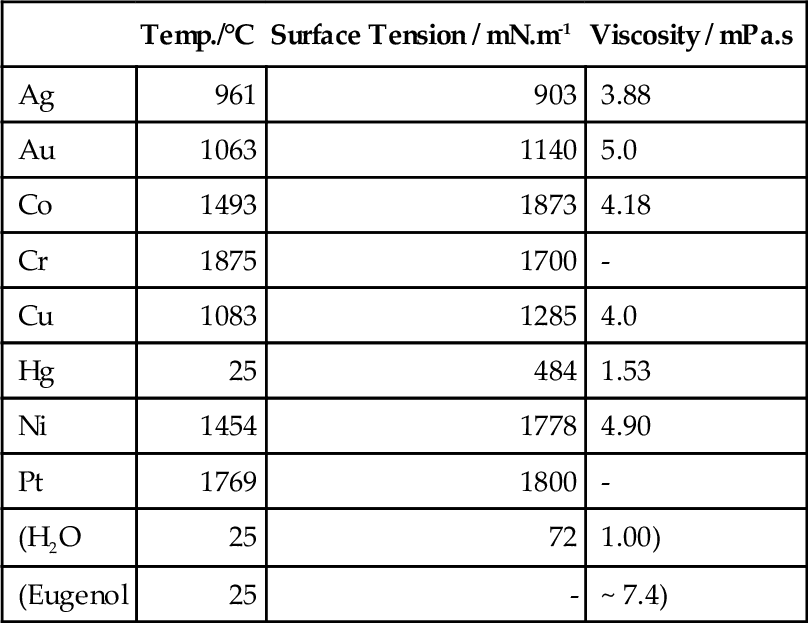
Clearly, the alloy must be at a temperature above the liquidus when it first enters the mould, otherwise solid will be present which could occlude detail at a surface or even block channels. An incomplete casting must then result. The viscosity of molten metals is usually quite low and so this presents no problems with flow as such, but the surface tension is certainly very high (Table 2.1) and wetting of the investment may be taken as impossible (contact angle θ = 180°). Most ceramics simply are not wetted by metals, cf. mercury on glass (the notable exception to this is cobalt on certain carbides, 21§3). This effect will prevent sharp, detailed castings unless there is sufficient pressure to force the liquid into corners and fine sections. For the scale of dental castings it is unlikely that this could be realized by simple pouring, relying on the hydrostatic pressure under a long sprue, even allowing for the high density of gold alloys, for example. Thus, some mechanical assistance must be provided for the purpose, i.e. with centrifugal, pressure or vacuum casting machines. We return to this point below.
•2.2 Porosity
Even so, and quite unlike pouring gypsum products and investment casts, the molten metal is admitted to the mould space through a rather narrow sprue. Although a large amount of air may in fact escape through the sprue, bubbling back against the metal flow, not all of it may do so because of the shape of the mould space. This would further retard or even curtail the completion of the casting. However, investments are prepared from a powder with a relatively large quantity of mixing water, certainly in excess of the amount required for the setting reaction. This excess, of course, leaves a very porous structure when it has been evaporated, and this in turn allows trapped air to escape. Obviously, such porosity must be connected for flow through the mass to occur (Fig. 2.1). Even then, for casting complicated shapes and large volumes, vents may be added to the larger structural elements of the pattern, remote from the sprue, to permit ready escape of a large volume of the trapped air (17§7).
For uniform spheres the maximum packing density corresponds to a volume fraction of about 74%. Irregular particles decrease this figure considerably and careful selection of the particle size distribution of the refractory silica is essential if a dense, and therefore strong, structure is to be obtained. But, clearly, care must be taken not to go too far and impede the escape of the air through that structure. The pore size must be small enough to put a satisfactory upper limit on the surface roughness of the resultant casting. But even though the viscosity of air is low, because of the high speed necessary to get the metal into the mould without premature freezing, this factor is critical. The failure of the air to escape creates ‘back-pressure’ defects, bubbles trapped between investment and metal.
•2.3 Oxidation
Air has another connotation in metals casting: it is an oxidizing agent. The problems with this are two-fold: there will be a resultant change in alloy composition because the elements vary in their tendency to oxidize and the rate of their oxidation. Excessive amounts of oxides on the melt (known as dross) may possibly get carried over with the metal and be included in the structure, thus weakening it (these oxides, again, are not wetted by the metal); although under normal conditions most, if not all, should remain in the crucible. Even then, the failure to wet would usually mean that they would be ejected from the melt and only lie on the surface of the casting (cf. oxides on amalgam alloy, 15§1).
When a flame is used to melt the metal, reducing conditions are employed (see §5). Such conditions may be attempted within the investment to prevent oxides forming on the surface of the casting so as to reduce the work required for cleaning it up after divesting, but also to allow a smoother surface to the cleaned work. Powdered graphite or copper has been used in this way, dispersed in the investment powder, i.e. as easily oxidizable material which, by using up oxygen already present and as it continues to diffuse in, reduces the exposure of the metal. However, copper would dissolve in gold alloys, changing their composition, especially at the surface (which could make them more prone to tarnishing), while graphite is prone to being burnt out along with the wax residues and could not be used at high temperatures because of reaction with calcium sulphate (17§3.4). An alternative has been the use of a graphite crucible, but this may not be used for alloys that dissolve carbon because of the embrittlement that the extra carbide formed would cause. In principle, any reducing agent that does not react with the alloy could be acceptable. Thus, while boron and aluminium have been suggested for gold alloys,[1] Al has appreciable solid solubility in Ag, Au and Cu (and thus presumably in their alloys), and AuB2 may form under such conditions; the options are limited (cf. 19§1.14).
The speed of heating obtained with induction casting machines greatly reduces the problem of air oxidation and such measures become less critical, but the price of such machines is likely to mean the continued use of flames in many countries for a long time yet. The more obvious procedure of doing the casting under an inert atmosphere (such as argon) is even more costly because of the specialized equipment required. However, it must be used for titanium because of its reactivity (28§1).
•2.4 Cooling
The need to ‘burn out’ an investment mould before casting has already been discussed from the point of view of removing the pattern materials (16§4) and achieving mould expansion by heating (Chap. 17) (see also §4). There is, however, a third requirement: avoidance of premature chilling. Although molten metal velocities in the mould space are known to be of the order of 0.5 ~ 1 ms-1, the metal probably then takes only about 0.1 ~ 0.2 s to cool to the liquidus. In simple designs, such as crowns and inlays, this may not present a problem, but for large frameworks and mesh it is a much more serious matter. The relationships involved are complex and not fully worked out yet, but several factors are of obvious importance. Firstly, the ‘temperature excess’,1 how far above the liquidus that the metal has been heated, controls how much heat is available to be lost before solidification commences. Secondly, the temperature of the investment then establishes the temperature gradient and thus the rate of loss of heat. The specific heat capacities of metal and investment, and the conductivity of the latter, are also involved but not amenable to control in practice. Clearly, it is not just a matter of simply raising both metal and investment temperatures. Mould space expansion, investment breakdown (also in contact with the hot metal), and alloy component oxidation and volatilization also need to be considered as critical issues in casting accuracy and resultant properties. Part of the problem, though, is the driving force for the hot metal into the mould. It is this we now consider.
§3 Physics of Casting
The near-total exclusion of air (or other gas) by casting in a vacuum would of course avoid both interference from effects due to porosity and also from oxidation, but this approach cannot of itself assist in obtaining complete and sharp castings as surface tension remains unaffected: some extra assistance is required. Centrifugal casting machines are commonly used to provide such assistance and the effect may be analysed in the following way.
•3.1 Rotation
Newton’s Second Law of Motion states that
the rate of change of momentum is proportional to the applied force, and takes place in the direction in which the force acts.
After integration, this leads to the expression
< ?xml:namespace prefix = "mml" ns = "http://www.w3.org/1998/Math/MathML" />
where F is the force, m the mass, and a the acceleration. In terms of the units this is
which in fact provides the definition of the newton as the SI unit of force. For a body rotating uniformly about a fixed point, as for example a ball on a string, the equivalent of equation 3.1 is
where R is the radius of the path of the centre of gravity of the (small) body and v is its velocity. What this means is that, despite the rotation being uniform, there is still an acceleration of v2/R acting, and this is outwards, away from the centre of rotation (hence ‘centrifugal’) (Fig. 3.1). This is due to the body being constrained to follow the circular path by the force F. The velocity that is to be measured is taken along the tangent to the path of rotation (which in fact shows that the body would move further away from the centre of rotation if it were not constrained) and so may be expressed by:
where n is the number of revolutions per second (i.e. the units of n are s-1). Hence, substituting from 3.4 into 3.3, we have:
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses