Alternative and specialized imaging modalities
Introduction
An array of medical imaging modalities has been developed in recent years and these continue to be developed at a phenomenal rate. Totally new imaging techniques have been introduced, while the resolution and image quality of existing systems are continually being refined and improved. Research and development have focused on manipulating and altering all three of the basic requirements for image production – the patient, the image-generating equipment (to find alternatives to ionizing radiation) and the image receptor. Digital image receptors (solid-state and photostimulable phosphor plates) are now used routinely (see Ch. 4). More and more sophisticated computer software is being developed to manipulate the image itself, once it has been captured. Many of these imaging modalities are playing an increasingly important role in dentistry. As a result, clinicians need to be aware of them and their application in the head and neck region. The main specialized imaging modalities include:
This chapter provides a summary of these modalities and their main applications in the head and neck region.
Contrast studies
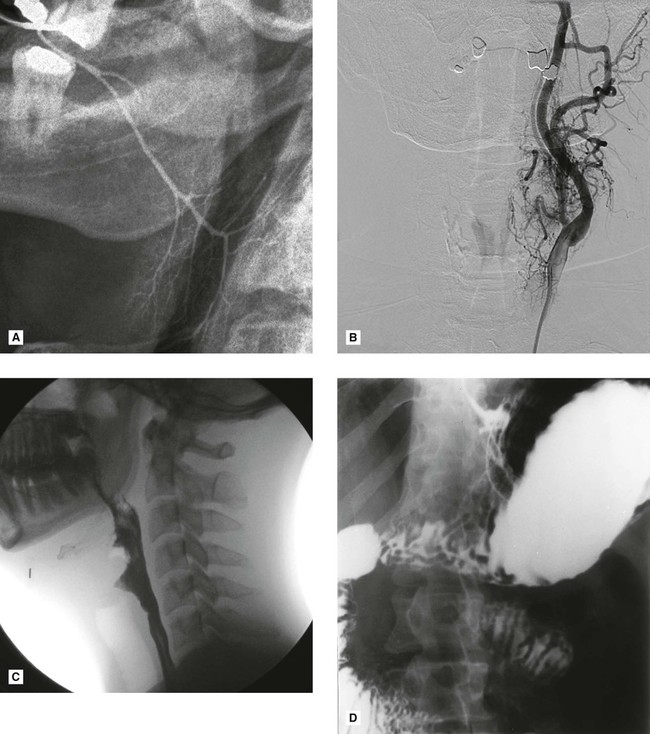
These investigations use contrast media, radiopaque substances that have been developed to alter artificially the density of different parts of the patient, so altering subject contrast – the difference in the X-ray beam transmitted through different parts of the patient’s tissues (see Ch. 17). Thus, by altering the patient, certain organs, structures and tissues, invisible using conventional means, can be seen (see Fig. 18.1). Contrast studies, and the tissues imaged, include:
• Sialography – salivary glands
• Lymphography – lymph nodes and vessels
Main contrast studies used in the head and neck
• Computed tomography – to provide general enhancement (see later)
• Barium swallow – to image the oesophagus
• Angiography – aqueous iodine-based contrast media introduced into selected blood vessels, e.g., the carotids (common, internal or external) or the vertebral arteries.
The procedure usually entails introducing a catheter into a femoral artery followed by selective catheterization of the carotid or vertebral arteries, as required, using fluoroscopic control. Once the catheter is sited correctly, the contrast medium is injected and radiographs of the appropriate area taken (see Fig. 18.1B).
Main indications for angiography in the head and neck
• To show the vascular anatomy and feeder vessels associated with haemangiomas
• To show the vascular anatomy of arteriovenous malformations
• Investigation of suspected subarachnoid haemorrhage resulting from an aneurysm in the Circle of Willis
• Investigation of transient ischaemic attacks possibly caused by emboli from atheromatous plaques in the carotid arteries.
Radioisotope imaging
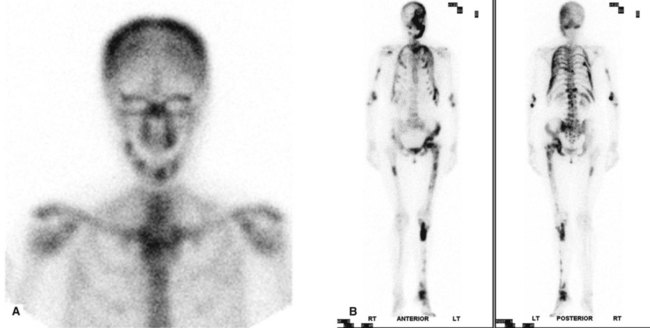
Radioisotope imaging relies upon altering the patient by making the tissues radioactive and the patient becoming the source of ionizing radiation. This is done by injecting certain radioactive compounds into the patient that have an affinity for particular tissues – so-called target tissues. The radioactive compounds become concentrated in the target tissue and their radiation emissions are then detected and imaged, usually using a stationary gamma camera (see Fig. 18.2). This investigation allows the function and/or the structure of the target tissue to be examined under both static and dynamic conditions.
Radioisotopes and radioactivity
Radioisotopes, as defined in Chapter 2, are isotopes with unstable nuclei that undergo radioactive disintegration. This disintegration is often accompanied by the emission of radioactive particles or radiation. The important emissions include:
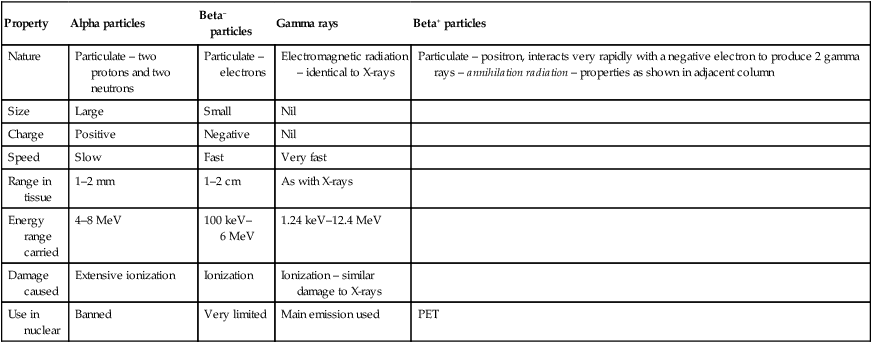
The main properties and characteristics of these emissions are summarized in Table 18.1.
Table 18.1
Summary of the main properties and characteristics of radioactive emissions
| Property | Alpha particles | Beta– particles | Gamma rays | Beta+ particles |
| Nature | Particulate – two protons and two neutrons | Particulate – electrons | Electromagnetic radiation – identical to X-rays | Particulate – positron, interacts very rapidly with a negative electron to produce 2 gamma rays – annihilation radiation – properties as shown in adjacent column |
| Size | Large | Small | Nil | |
| Charge | Positive | Negative | Nil | |
| Speed | Slow | Fast | Very fast | |
| Range in tissue | 1–2 mm | 1–2 cm | As with X-rays | |
| Energy range carried | 4–8 MeV | 100 keV–6 MeV | 1.24 keV–12.4 MeV | |
| Damage caused | Extensive ionization | Ionization | Ionization – similar damage to X-rays | |
| Use in nuclear | Banned | Very limited | Main emission used | PET |
Radioisotopes used in conventional nuclear medicine
• Technetium (99mTc) – salivary glands, thyroid, bone, blood, liver, lung and heart
99mTc is the most commonly used radioisotope. Its main properties include:
• Single 141 keV gamma emissions which are ideal for imaging purposes
• A short half-life of 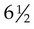 hours which ensures a minimal radiation dose
hours which ensures a minimal radiation dose
• It is readily attached to a variety of different substances that are concentrated in different organs, e.g.:
• It can be used on its own in its ionic form (pertechnetate 99mTcO4–), since this is taken up selectively by the thyroid and salivary glands
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses