The orofacial pain–endo interface
Definition of pain
Pain is defined as an unpleasant sensory and emotional experience associated with actual or potential tissue damage. Pain has a strong motivational input to behaviour, yet the link between tissue damage, pain and behaviour is not always proportionate or direct. Pain from an accidental burn, such as a hot iron, elicits sharp, transient pain causing a withdrawal reflex; this is acute pain, which is protective in its behavioural drive. A stimulus that is potentially damaging to tissues is considered noxious. The endodontic equivalent of acute pain is that of dentinal, pulpal or periapical origin. Pain that lasts for a few days or a few weeks can follow musculoskeletal or sports injuries; this is prolonged pain, which can also be protective in its behavioural drive. In a proportion of cases, the prolonged pain becomes persistent or chronic, lasting beyond the period of injury and its healing; this no longer has a protective value but it may continue to affect behaviour. The endodontic equivalent is neurogenic pain or pain from other structures that appears to arise from teeth and its associated structures. Although there are relatively few conditions to diagnose for the endodontist, their overlapping presentation and innate subtleties complicate straightforward identification (see Chapter 4). The problem is similar and even more complicated when considering the overlapping presentation of pain from other orofacial structures.
Orofacial pain prevalence
It is difficult to determine accurately the true prevalence and incidence of pain, both in general and in the orofacial region. Nevertheless, it is estimated that a quarter of the adult population have suffered from orofacial pain (MacFarlane et al., 2001). In children, toothache is the most common orofacial pain, 12% experiencing an episode before the age of 5 and 32% by the age of 12. Meanwhile, two recent UK studies of adults showed 12–19% of the population reported orofacial pain within the previous month (Drangsholt & LeResche, 2009).
The prevalence of chronic pain is reputed to be 35.5% in the general population (Raferty et al., 2011). In another large scale survey of 15 European countries, 19% of adults reported suffering from chronic pain, which seriously affected the quality of their lives (Breivik et al., 2006). Furthermore, the overall incidence of chronic orofacial pain has been given as 38.7 per 100 000 per year (Koopman et al., 2009). In referred endodontic patients, about 12% may have some form of chronic pain, either as a result of endodontic treatment or because of initial misdiagnosis (Polycarpou et al., 2005).
Neurophysiological basis of orofacial pain
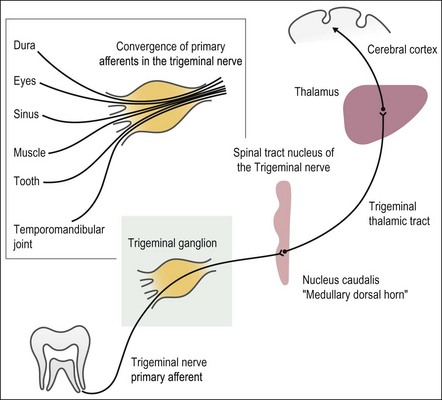
The sensory transmission of orofacial pain is conveyed predominantly by Aδ and C fibres of the trigeminal nerve, which project to the nucleus caudalis in the medulla. The latter is sometimes referred to as the medullary dorsal horn as it is the functional equivalent of the spinal dorsal horn. Pain input is modulated at this site in the brain stem before fibres then project via the spinothalamic tract to the thalamus and thence to the cerebral cortex. The sensory-discriminative component of pain and activation of the endogenous analgesic system are believed to originate within the cortex. A schematic diagram illustrating the trigeminal pain pathway is presented in Figure 16.1.
Classification of orofacial pain
Orofacial pain (OFP) is defined as pain whose origin is below the orbito-meatal line, above the neck and anterior to the ears, including pain within the mouth (Zakrzewska & Hamlyn, 1999).
Numerous classification systems based on the structures, symptoms or treatment (Table 16.1) have been proposed for the orofacial region, the more detailed assigning individuals to multiple categories, which ideally incorporate both the physical component and the psychological impact of pain. The number of classifications reflects the fact that the discipline is still developing and there is still uncertainty about aetiopathogenesis, in turn reflected in a rich field of research.
Table 16.1
Origin of some classification systems
Some of the classification systems available for orofacial pain
RDC/TMD (Research Diagnostic Criteria for Temporomandibular Disorders) 1992
IASP (International Association for the Study of Pain) 1994
AAOP (American Academy of Orofacial Pain) 2008
ICHD (International Classification of Headache Disorders) 2nd edn 2004
Diagnosis of orofacial pain
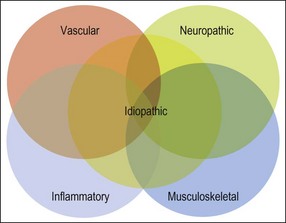
A careful history is obtained to determine the characteristics of the orofacial pain, as well as any history of other generalized chronic pain conditions. Information on co-morbid anxiety, depression, sleep problems, social and family history should all be recorded. Pre-consultation questionnaires may be useful when managing chronic pain to assess the behavioural impact of pain and in establishing the patient’s goals and expectations. Clinical examination, together with special tests if required, is used to confirm diagnosis. The differential diagnoses of orofacial pain constitute a vast list but a broad assessment is essential to exclude rare presentations of intra- or extracranial malignant neoplasia, severe infective inflammatory conditions before considering the more commonly encountered presentations of pain. The main causes of chronic orofacial pain are then often classified into inflammatory, vascular, musculoskeletal, neuropathic and idiopathic (Fig. 16.2).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses