Management of tooth resorption
Aetiology and pathogenesis
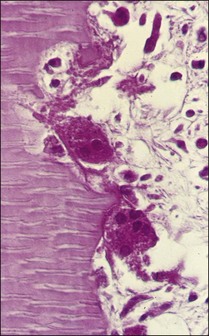
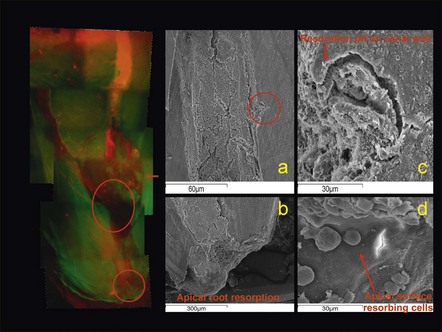
The cells responsible for resorption of dental hard tissues are multinucleated giant cells classified as clasts (Fig. 11.1). These cells attach themselves to the mineralized tissues by the cell membrane adjacent to the part of the cytoplasm called the “clear zone”. Resorption takes place under the highly folded cell membrane, known as the ruffled border (Fig. 11.2) in this zone. The membrane releases a cocktail of enzymes capable of dissolving both organic and inorganic components of hard tissues. Osteoclasts are involved in resorption of bone and dentinoclasts resorb dentine and cementum. The latter are smaller, have fewer nuclei and their clear zone is minimal or non-existent.
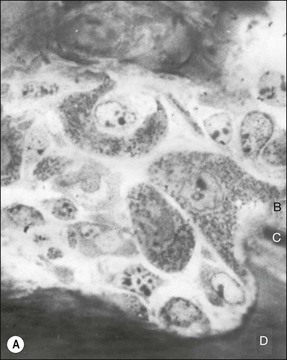
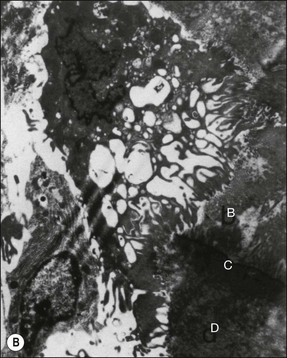
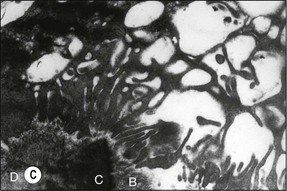
Fig. 11.2 (a) Light micrograph of an osteoclast-like cell in close apposition to dentine, cementum and alveolar bone (×100); (b) electron micrograph of the osteooclast-like cell in (a) with its ruffled border along dentine, cementum, and alveolar bone (×5000); (c) electron micrograph showing the ruffled border of the osteoclast-like cell in (a) along dentine, cementum, and alveolar bone (×19 000). B = alveolar bone, C = cementum, D = dentine
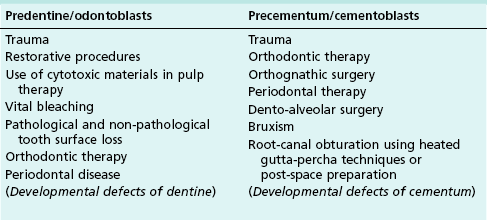
For dental resorption to occur, there must be damage to the non-mineralized components (predentine and periodontal ligaments) and interference with the normal protection afforded by blast cells (odontoblasts, cementoblasts and fibroblasts), which allows colonization of the denuded surfaces by dentinoclasts. The causes of such damage are listed in Table 11.1. The concomitant acute inflammation during injury activates the dentinoclasts and initiates resorption. However, this process may be transient in the absence of a perpetuating or persistent stimulus for the dentinoclasts, allowing repair by hard tissue deposition to occur. Resorption becomes progressive if it is sustained by chronic inflammation, whether in the pulp or periodontium. The causes of persistent chronic inflammation are listed in Table 11.2.
1. Transient external resorption
2. Progressive external resorption without persistent inflammation of periodontal tissue (replacement resorption)
3. Progressive external resorption associated with persistent localized inflammation of periodontal tissue
i. Sustained by root-canal infection
ii. Sustained by “pressure effect”
4. Progressive external resorption associated with identified systemic disease process
5. Progressive external resorption associated with no obvious local or systemic disease process (idiopathic).
Internal resorption
This type of resorption has a prevalence of 0.1–1.6% in permanent teeth. It is initiated by damage to or loss of the predentine and odontoblast layer (see Table 11.1) and sustained by pulpal inflammation (see Table 11.2). Dental trauma, overheating and dehydration during restorative procedures, use of cytotoxic restorative materials or dressing materials in pulpotomies, and orthodontic tooth movement are factors that may injure the protective predentine and odontoblast cells.
Transient internal resorption
In the absence of persistent pulpal inflammation, initial resorption of dentine from the pulpal aspect is self-limiting with no clinical consequence (Fig. 11.3). It has been detected, for example, in boxers sustaining repetitive injury to anterior teeth.
Progressive internal resorption
Clinically, the early stages of internal resorption are usually asymptomatic. A pink spot may be present on the crown if resorption takes place in the pulp chamber (Fig. 11.4). The pink discoloration associated with internal resorption is caused by the highly vascularized inflammatory tissue undermining the coronal enamel. Vital pulp tissue is required for internal resorption to progress, therefore, such teeth will give positive responses to pulp tests. However, a negative response could not rule out the progression of resorption because only the pulp tissue coronal to the lesion may be necrotic. Progression of inflammation fuelling the resorption may result in total necrosis of the pulp and, therefore, cessation of internal resorption. The necrotic pulp may then become infected and symptomatic, discoloured and give negative responses to pulp tests (Fig. 11.5).
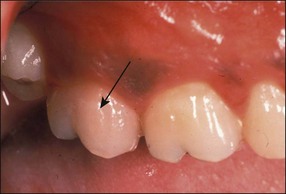
Fig. 11.4 Internal resorption affecting the pulp chamber of the maxillary second molar, the crown exhibited a pinkish discoloration (arrowed)
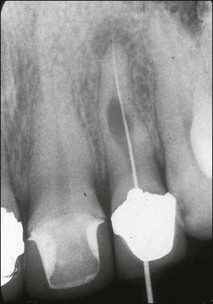
Fig. 11.5 Internal resorption affecting the maxillary lateral incisor, the pulp became infected and apical periodontitis developed
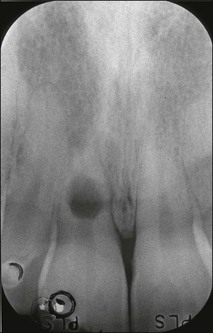
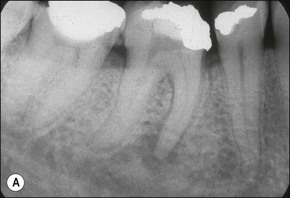
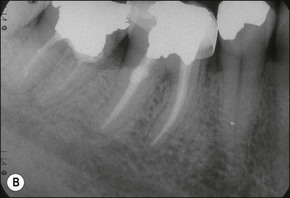
Radiographically, the appearance of internal resorption has been classically described as a well-circumscribed, symmetrical, oval or circular shaped radiolucency continuous with the root canal (Fig. 11.6). The radiolucency has a uniform density and the pulp chamber or canal cannot be followed through the lesion. The position of the radiolucency remains the same in mesial or distal angled radiographic views. However, a radiopaque mass resembling calcification may be present within the lesion or in the canal space apical to the internal resorption. A periradicular radiolucency may be associated with perforation of the root or total necrosis of the pulp in advanced cases (Fig. 11.7). This may pose difficulty in differentiation from external inflammatory resorption associated with root-canal infection (Fig. 11.8). Occasionally, internal resorptive lesions remain undetected and may only appear incidentally on a post-obturation radiograph (Fig. 11.9).
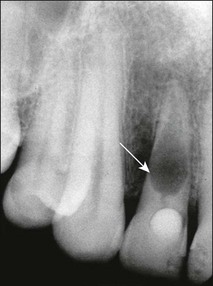
Fig. 11.7 The right maxillary lateral incisor associated with extensive internal resorption and associated periradicular radiolucency where it breached the distal surface (arrowed)
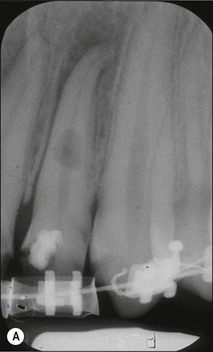
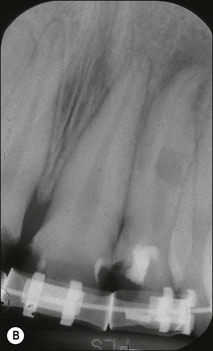
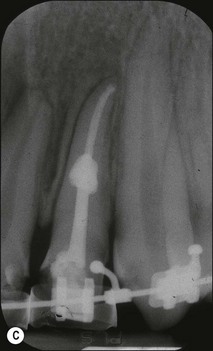
Fig. 11.8 (a–c) Internal resorption affecting the left maxillary incisor with similar presentation as external resorption
It may be difficult to control haemorrhage and to remove completely the severely inflamed tissue from the resorptive defect. Therefore, debridement should be carried out using copious amounts of sodium hypochlorite irrigant. In addition to its antibacterial properties, sodium hypochlorite also possesses tissue-dissolving ability. Use of higher concentration (5%) of sodium hypochlorite in combination with ultrasonic agitation has been recommended and may enhance debridement. The canal should be checked for perforation by exploring with a curved file attached to an apex locator. Care should be taken to prevent extrusion of sodium hypochlorite irrigant through any perforation. An extensive lesion with calcification may render canal location and complete debridement of the resorptive defect impossible (Figs 11.4, 11.10). Calcium hydroxide dressing should be used between appointments to prevent recolonization by bacteria and to facilitate removal of any residual inflamed tissue within the irregular resorptive defect. Long-term dressing with calcium hydroxide is indicated to induce hard tissue repair if a perforation is present (see Figs 11.7, 11.11). It may be prudent to delay root-canal obturation until complete bone healing has occurred to ensure minimum extrusion of filling material.
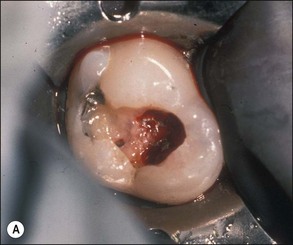
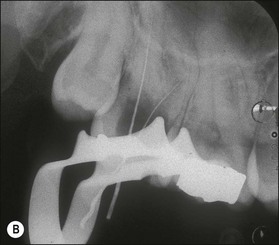
Fig. 11.10 (a,b) Extensive internal resorption defect with calcification of the tooth in Figure 11.4 rendered complete debridement impossible
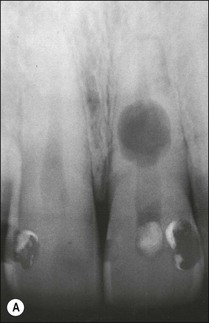
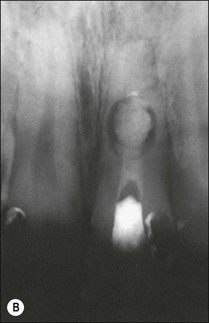
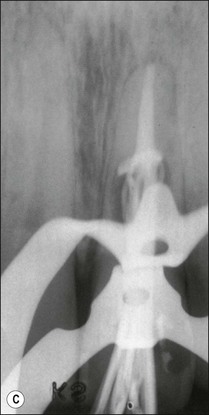
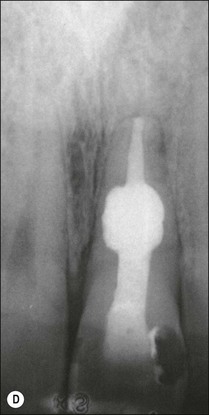
Fig. 11.11 (a) Internal resorption in a maxillary central incisor; (b) calcium hydroxide dressing filling the defect incompletely and inadequately; (c) apical part of the root canal obturated with laterally compacted gutta-percha and sealer; (d) the resorption cavity and coronal part of the canal obturated with warm gutta-percha and sealer
Complete obturation of the defect may be achieved by using a thermoplasticized root-filling technique, such as injection of molten gutta-percha using an Obtura II system (see Chapter 8) after filling the canal apical to the defect using lateral or vertical compaction technique (Fig. 11.11).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses