Radiation protection
16.1 Ionising radiation and its effects
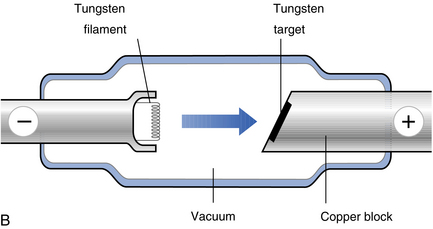
X-rays are produced by an electrical process in an X-ray tube (Fig. 16.1). Electrons, released from a heated tungsten filament, are accelerated in a vacuum by the application of a high voltage (typically 50–75 kV (kilovolts)) and strike a positively charged target. The sudden halt of the electrons releases energy, mainly as heat but also as X-rays. The X-rays are a mixture of photons of different energies, but low-energy (dose-producing) photons predominate.
The X-rays are filtered, usually using aluminium, to remove the low-energy photons (Fig. 16.2). The photons are then shaped into an appropriately sized beam by collimation using steel diaphragms or cylinders (Fig. 16.3).
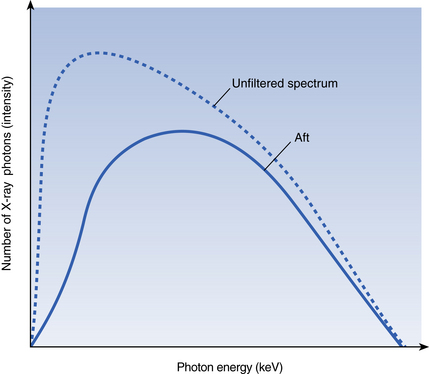
Fig. 16.2 The effect of filtration on the X-ray beam. Low-energy photons predominate in the unfiltered spectrum.
Filtration removes proportionately more of these ‘weak X-rays’, resulting in a filtered beam with a higher mean energy at the expense of a loss of intensity.
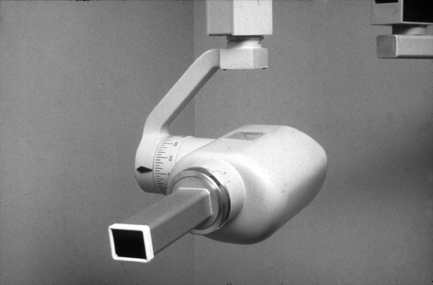
Fig. 16.3 An X-ray set with rectangular collimation.
This beam restriction method results in a dose reduction to patients of about 65% compared with conventional 6-cm-diameter round beams.
Interaction with matter
Three possible interactions can occur at the atomic level when X-rays interact with matter:
Somatic and genetic effects of X-rays
Doses and risks in dental radiography
‘Radiation dose’ is a measure of the energy imparted by X-ray exposure and its biological effect. At a simple level, we can measure the energy imparted per unit mass (joules/kg), but using established methods we can calculate ‘effective dose’. This is a ‘whole body equivalent’ value which can be directly related to stochastic effects. Doses and risks vary enormously according to the type of equipment used, so it is hard to give firm figures, but recent estimations of risk (and radiation dose) are given in Table 16.1.
Table 16.1
Estimates of dose and risk in dental radiography
| Technique | Effective dose (microsieverts) | Risk of cancer (per million) |
| Intraoral (bitewing, periapical) | <2 | <0.1 |
| Panoramic | 3–24 | 0.1–1.3 |
| Lateral cephalogram | <6 | <0.2 |
| CBCT (‘dento-alveolar’) | 11–674 (median = 61) | 0.6–37 (median = 3.4) |
| CBCT (‘craniofacial’) | 30–1073 (median = 87) | 1.6–59 (median = 4.7) |
| Computed tomography (as used for dental implant planning) | 280–1410 | 15–77 |
16.2 Radiation protection
Protection of patients
In dental radiography, protection of patients is achieved by three main means:
Justification
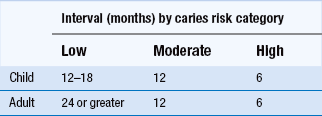
Selection of bitewing radiographs: The nearest we come to ‘routine’ radiography in dentistry is with the bitewing radiograph. For dentate patients who are new to the practice (and partially dentate patients where films can be supported in the mouth), most authorities agree that a posterior bitewing examination is justified. Thereafter, the intervals between bitewing examinations should be determined by assessment of caries risk. Current UK guidelines are shown in Table 16.2, although other guidelines with slightly different intervals are available and can be found internationally. Obviously bitewing frequency for an individual patient may change if the patient changes caries risk category.
If the dentist feels that a radiographic examination is of help in assessment of bone loss in periodontal disease, then bitewing radiographs will provide the necessary information in the premolar and molar regions, providing geometrically accurate images. Where periodontal probing depths exceed 5 mm, then vertical bitewing radiographs are appropriate.
Selection of periapical radiographs: Periapical radiographs are indicated in the following situations:
• When dictated by localised symptoms/signs (pain, swelling and tenderness of a tooth).
• Prior to the extraction of erupted third molars, retained roots, lone-standing upper molars or where there is reasonable clinical suspicion that problems may arise. The fashion of routine pre-extraction radiographs has arisen in the absence of any scientific evidence of benefit.
• Prior to preparation of a tooth for a crown or bridge retainer.
• In endodontics, where basic guidelines suggest radiograph(s) at the following stages:
Selection of panoramic radiographs: In terms of image quality, panoramic radiography is inferior to good intraoral radiographs. Consequently, for most dental diagnostic uses, it is a ‘second best’ imaging technique. Possible situations where it may be useful include:
• where a bony lesion or unerupted tooth is of a size or position that precludes its complete demonstration on intraoral radiographs
• in orthodontic assessment when clinically indicated (no ‘screening’)
• preoperative assessment of third molars, unless other adequate radiographs are available
• when mandibular fracture is suspected.
• as part of implant dentistry planning, unless other adequate radiographs are available
Selection of CBCT examinations: Cone beam computed tomography (CBCT) is a relatively new technology and its capabilities are changing as equipment is refined. Despite this, one general principle can be stated: that CBCT should only be used when the question for which imaging is required cannot be answered adequately by lower-dose conventional (traditional) radiography. This view reflects the status quo in which radiation doses and economic costs associated with CBCT are usually higher than with conventional radiography.
Dose limitation
Patient doses in dental radiography can be minimised by considering the following:
• Operating potential (kilovoltage): for intraoral radiography, a minimum of 50 kV is set and 65–70 kV is recommended. This is often fixed on dental intraoral X-ray sets but is usually used to control exposures on panoramic X-ray equipment.
• Tube current-exposure time product (mAs). The current (mA) is often fixed on dental intraoral X-ray sets, while the exposure time(s) is often fixed for panoramic and CBCT machines.
• AC/DC generation of X-rays: ‘DC’ (constant potential) generators lead to fewer low-energy (dose-producing) X-ray photons.
• Filtration: aluminium filters absorb low-energy X-ray photons.
• Collimation: on intraoral X-ray sets, the beam can be restricted to a rectangle of 4 cm by 3 cm, leading to a substantial dose reduction over the conventional 6-cm-diameter round beam; all new equipment should be fitted with rectangular collimation and it should be retrofitted on older equipment; on panoramic machines, selective field size collimation facilities may be available. For CBCT equipment, there should be a choice of fields of view and examinations must use the smallest that is compatible with the clinical situation if this provides less radiation dose to the patient.
• Image receptor speed: for intraoral radiography, digital systems may offer some reduction in mAs (and hence dose) compared with film. Where film is used, E- or F-speed films should be used. For digital panoramic equipment, this factor is out of the control of the operator, but for film-based panoramic radiography, a rare-earth screen/film combination should be used (a combination of ISO speed 400 or better is used). For CBCT equipment, the choice of image receptor is out of the control of the operator, although this will influence radiation dose.
• Lead shielding of patients: the only requirement to use a lead apron is when a pregnant woman is being examined using a technique involving a beam that would pass through the fetus. Thyroid shielding can be used if that organ lies in the primary beam of X-rays.
Quality assurance
A poor-quality image means that the patient receives reduced, or no, benefit from the risk of the X-ray examination. Even in the best hands, radiographs may be produced that are ‘rejects’. A quality standard of no greater than 10% of radiographs (5% for CBCT images) being non-diagnostic has been set for general dental practice. Good quality of radiographs can be addressed by attention to all of the criteria listed in Table 16.3.
Table 16.3
Methods of assuring good-quality radiographs
| Area | Improving methods |
| Radiographic technique | Use of film-holding/beam-aiming devices for intraoral radiography Careful positioning for panoramic radiography Careful selection and instruction of patients |
| X-ray set | Regular maintenance and servicing, as recommended by the manufacturer Triennia/> |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses