15
Primary and Secondary Hemostasis: Normal Mechanisms, Disease States, and Coagulation Tests: Assessment, Analysis, and Associated Dental Management Guidelines
PRIMARY AND SECONDARY HEMOSTASIS: OVERVIEW, FACTS, AND ASSOCIATED DISEASE STATES
Hemostasis Introduction
Coagulation is now determined to be a complex interaction of procoagulation factors, anticoagulation factors, and the fibrinolytic system. The normal clotting process goes through four phases to achieve optimal clot formation, clot limitation, and clot dissolution. Primary hemostasis and secondary hemostasis relates to clot formation; antithrombin processes in addition to proteins C and S relate to clot limitation; and fibrinolysis relates to clot dissolution.
Thus, to understand bleeding disorders, one needs to have a good understanding of the following:
The Elements of Hemostasis
The elements of hemostasis are:
Physiology of Hemostasis
Primary and secondary hemostasis has to function optimally for a patient to have a negative bleeding history. Additionally, there needs to be a physiologic balance between bleeding and clotting mechanisms. The immediate type of bleeding occurs when there are problems associated with the elements involved with primary hemostasis. The delayed type of bleeding occurs when there are problems associated with the elements involved with secondary hemostasis.
Elements Associated with Primary Hemostasis
The elements associated with primary hemostasis include:
Elements Associated with Secondary Hemostasis
Good secondary hemostasis is dependent on appropriate and adequate Clotting Factor interactions leading to formation of the fibrin clot.
Primary Hemostasis: Detailed Discussion
Primary hemostasis is a procoagulation clot forming process associated with the initiation and formation of the platelet plug. When injury occurs, within seconds to minutes the blood vessels at the injured site constrict and attract the circulating platelets (out of the circulation), promoting the adhesions of these circulating platelets to the subendothelium at the site of injury. The platelets aggregate in large number and link with one another to form the platelet plug. Von Willebrand’s Factor (vWF) enhances the sticking together, or cohesiveness, of the platelets. The surface phospholipids of platelets provide the surface for factor complexes and for the propagation of clot formation. Thus, platelets and vWF protein cooperate to form a loose plug in the lacerated blood vessel. This is primary hemostasis. Primary hemostasis defect is associated with excessive oozing at the time of surgery and oozing that continues beyond 24 hours postoperatively.
Factor VIII gene produces two transcripts of factor VIII. Transcript 1 (VIII-vWF) is the FVIII associated with von Willebrand factor and it helps in platelet aggregation (primary hemostasis). Transcript 2 consists primarily of the phospholipid-binding domain of Factor VIIIc. vWF helps stabilize and transport Factor VIIIc, which participates in the clotting cascade. Thus, von Willebrand Factor (vWF) plays a very important role in both primary and secondary hemostasis.
As you will see in the secondary hemostasis discussion, activated platelets express their cell surface phosphatidylserine (P-serine) that promotes the conversion of Factor II (prothrombin) to thrombin via Factors Xa, Va, and calcium. This is the starting phase of a chain of events that leads to Thrombin generation and the final conversion of fibrinogen to fibrin. Fibrin is stabilized Factor XIII, and fibrinolysis is responsible for degradation of fibrin through a complex mechanism of pro-activators and anti-activators that regulates the generation of plasmin.
Drugs affecting primary hemostasis:
- Aspirin
- NSAIDS
- Aspirin plus dipyridamole (Aggrenox)
- Adenosine diphosphate inhibitors: clopidogrel (Plavix)
- Ticlopidine (Ticlid)
Note that an immediate type of bleeding can also occur from increased vascular fragility due to chronic corticosteroid therapy. Chronic steroid use causes thinning of the vascular connective tissue lining, resulting in increased fragility of the small blood vessels. The bleeding time (BT) and platelet counts are normal and not affected with chronic corticosteroid use. During dentistry these patients respond well to local pressure and local hemostats.
Secondary Hemostasis: Detailed Discussion
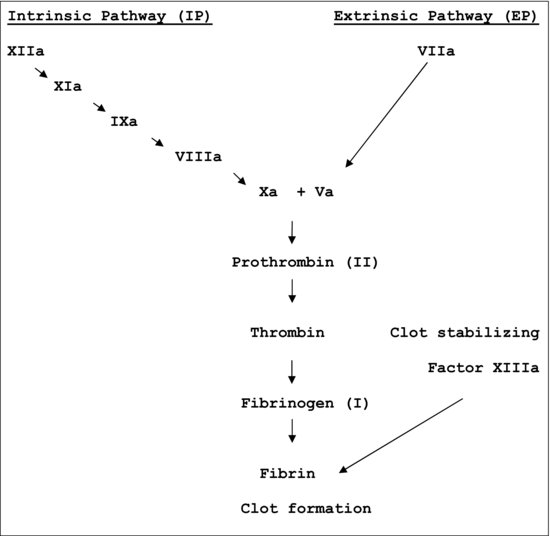
Secondary hemostasis is also a procoagulation clot forming process and it is associated with the propagation of the clotting process via the intrinsic and extrinsic coagulation cascades. All Clotting Factors, with the exception of Factor VIII, are manufactured in the liver. Factor VIII and vWF are manufactured in the endothelial cells of the blood vessels. Factors II, VII, IX, and X are vitamin K–dependent clotting factors, and they participate in the intrinsic and extrinsic clotting cascades. The clotting factors are present in the circulation in the inactive form. Normal coagulation system is a Y-shaped pathway with separate intrinsic and extrinsic component initiators. Factor XII and Factor VIIa/tissue Factors are initiators of the intrinsic and extrinsic pathways respectively, and both then lead to a common pathway of Factor Xa/Factor Va. The intrinsic and extrinsic clotting cascades demonstrate the sequential order in which the clotting factors activate to promote secondary hemostasis. Tissue factor released from the injured vasculature immediately activates Factor VII in the extrinsic clotting pathway and exposure to collagen from the ruptured vessel wall slowly activates Factor XII in the intrinsic pathway. The chain reaction that follows in the intrinsic and extrinsic clotting cascades (Figure 15.1< ?anchor c15-fig-0001 ?>) results in the formation of the fibrin clot that gets deposited on the platelet plug mesh (from primary hemostasis), and, together, they seal the injured site. The clot starts to stabilize in minutes to less than two hours. This is secondary hemostasis. Patients experiencing problems with secondary hemostasis present with deep-tissue bleeding 4–10days postoperatively. Anticoagulants/blood thinners, heparin or warfarin (Coumadin), affect secondary hemostasis.
Figure 15.1 The clotting cascade.
Clot Limitation
Antithrombotic control mechanisms plus proteins C and S are anticoagulation, clot-limiting processes. They bring about cessation of the clotting process.
Clot Dissolution
Fibrinolysis is an anticoagulation clot dissolution process. Fibrinolysis is mediated by the enzyme plasmin, which remodels or dissolves the clot once the bleeding stops. The clot breaks down hours after the initial clot has formed. Fibrinogen and plasmin balance is crucial for optimal hemostasis.
Clotting Factor Facts
Optimal vitamin K absorption from the gut is needed for the manufacture of Factors II, VII, IX, and X in the liver. Low levels of these factors prolong the PT/INR and the PTT. Liver disease, particularly decompensated alcoholic cirrhosis, is the leading cause of Clotting Factor deficiency. It is important to note that not all patients with cirrhosis will have a prolonged PT/INR. Clotting Factor activity has to drop by more than 75% to cause prolongation of the PT/INR.
Note that platelets are also produced in the liver, and thrombocytopenia can occur in the presence of cirrhosis. Always determine the PT/INR and the CBC with platelets prior to probing a cirrhotic patient. Chronic small bowel disease affects vitamin K absorption. Thus, Factors II, VII, IX, and X can be low in the presence of significant vitamin K deficiency due to chronic small bowel disease. The deficiency can be corrected by injecting 10mg vitamin K daily, IV for three days. Vitamin K IV promotes reversal in 6–12 hours. Oral vitamin K will be ineffective in the presence of chronic malabsorption.
The liver normally modifies the vitamin K–dependent clotting Factors (II, VII, IX, and X) for proper physiologic function, and this function may be deranged in patients with vitamin K deficiency associated with cirrhosis. Vitamin K deficiency occurs commonly in end-stage liver disease (ESLD) because of poor nutrition, malabsorption of fat-soluble vitamins, biliary tract obstruction, bile salt deficiency, bile salt secretory failure, and with the use of broad-spectrum antibiotics. Additionally, cirrhotic patients develop an acquired unresponsiveness to vitamin K, which is reflected by increased levels of hypocarboxylated vitamin K–dependent clotting factors. Vitamin K deficiency is more likely in primary cholestatic diseases such as primary biliary cirrhosis and primary sclerosing cholangitis.
Providing vitamin K by oral, subcutaneous, or intravenous administration can correct the decline. The IV route is preferred because of edema and possible gut malabsorption in seve/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


