Odontogenic Cysts and Tumors*
Orthokeratinized Odontogenic Cyst
Nevoid Basal Cell Carcinoma Syndrome
Gingival (Alveolar) Cyst of the Newborn
Carcinoma Arising in Odontogenic Cysts
TUMORS OF ODONTOGENIC EPITHELIUM
Malignant Ameloblastoma and Ameloblastic Carcinoma
Clear Cell Odontogenic Carcinoma
Calcifying Epithelial Odontogenic Tumor
TUMORS OF ODONTOGENIC ECTOMESENCHYME
Peripheral Odontogenic Fibroma
Odontogenic Cysts
With rare exceptions, epithelium-lined cysts in bone are seen only in the jaws. Other than a few cysts that may result from the inclusion of epithelium along embryonic lines of fusion, most jaw cysts are lined by epithelium that is derived from odontogenic epithelium. These are referred to as odontogenic cysts. (Nonodontogenic jaw cysts are discussed in Chapter 1.)
Odontogenic cysts are subclassified as developmental or inflammatory in origin. The inciting factors that initiate the formation of developmental cysts are unknown, but these lesions do not appear to be the result of an inflammatory reaction. Inflammatory cysts are the result of inflammation. Box 15-1 presents categories of odontogenic cysts modified from the 2005 World Health Organization (WHO) classification. (The periapical cyst is discussed in Chapter 3.)
DENTIGEROUS CYST (FOLLICULAR CYST)
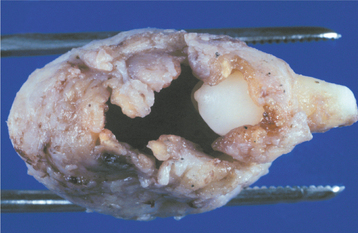
The dentigerous cyst is defined as a cyst that originates by the separation of the follicle from around the crown of an unerupted tooth. This is the most common type of developmental odontogenic cyst, making up about 20% of all epithelium-lined cysts of the jaws. The dentigerous cyst encloses the crown of an unerupted tooth and is attached to the tooth at the cemento-enamel junction (Fig. 15-1). The pathogenesis of this cyst is uncertain, but apparently it develops by accumulation of fluid between the reduced enamel epithelium and the tooth crown.
CLINICAL AND RADIOGRAPHIC FEATURES: Although dentigerous cysts may occur in association with any unerupted tooth, most often they involve mandibular third molars. Other relatively frequent sites include maxillary canines, maxillary third molars, and mandibular second premolars. Dentigerous cysts rarely involve unerupted deciduous teeth. Occasionally, they are associated with supernumerary teeth or odontomas.
Although dentigerous cysts may be encountered in patients across a wide age range, they are discovered most frequently in patients between 10 and 30 years of age. There is a slight male predilection, and the prevalence is higher for whites than for blacks. Small dentigerous cysts are usually completely asymptomatic and are discovered only on a routine radiographic examination or when films are taken to determine the reason for the failure of a tooth to erupt. Dentigerous cysts can grow to a considerable size, and large cysts may be associated with a painless expansion of the bone in the involved area. Extensive lesions may result in facial asymmetry. Large dentigerous cysts are uncommon, and most lesions that are considered to be large dentigerous cysts on radiographic examination will prove to be odontogenic keratocysts or ameloblastomas. Dentigerous cysts may become infected and be associated with pain and swelling. Such infections may arise in a dentigerous cyst that is associated with a partially erupted tooth or by extension from a peri-apical or periodontal lesion that affects an adjacent tooth.
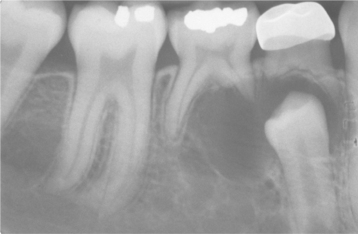
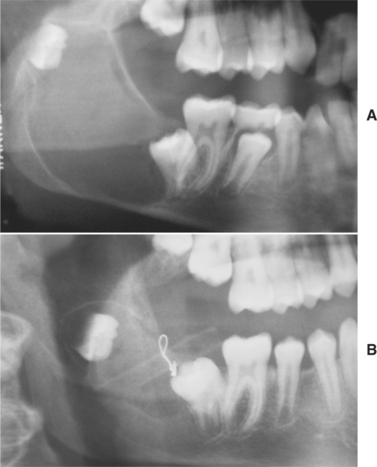
The cyst-to-crown relationship shows several radiographic variations. In the central variety, which is the most common, the cyst surrounds the crown of the tooth and the crown projects into the cyst (Fig. 15-2). The lateral variety is usually associated with mesioangular impacted mandibular third molars that are partially erupted. The cyst grows laterally along the root surface and partially surrounds the crown (Fig. 15-3). In the circumferential variant, the cyst surrounds the crown and extends for some distance along the root so that a significant portion of the root appears to lie within the cyst (Fig. 15-4). Rarely, a third molar may be displaced to the lower border of the mandible or higher up into the ascending ramus. Maxillary anterior teeth may be displaced into the floor of the nose, and other maxillary teeth may be moved through the maxillary sinus to the floor of the orbit. Dentigerous cysts may displace the involved tooth for a considerable distance. Root resorption of adjacent erupted teeth can occur.
Radiographic distinction between a small dentigerous cyst and an enlarged follicle about the crown of an unerupted tooth is difficult and may be largely an academic exercise (Fig. 15-5). For the lesion to be considered a dentigerous cyst, some investigators believe that the radiolucent space surrounding the tooth crown should be at least 3 to 4 mm in diameter. Radiographic findings are not diagnostic for a dentigerous cyst, however, because odontogenic keratocysts, unilocular ameloblastomas, and many other odontogenic and nonodontogenic tumors may have radiographic features that are essentially identical to those of a dentigerous cyst.
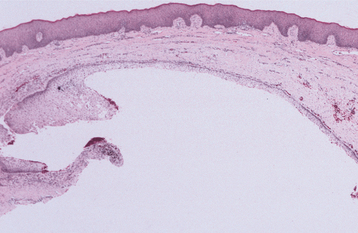
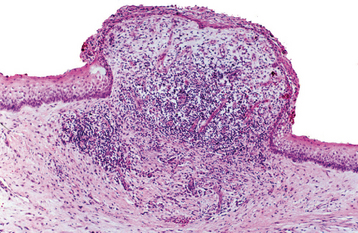
HISTOPATHOLOGIC FEATURES: The histopathologic features of dentigerous cysts vary, depending on whether the cyst is inflamed or not inflamed. In the noninflamed dentigerous cyst, the fibrous connective tissue wall is loosely arranged and contains considerable glycosaminoglycan ground substance. Small islands or cords of inactive-appearing odontogenic epithelial rests may be present in the fibrous wall. Occasionally these rests may be numerous, and at times pathologists who are not familiar with oral lesions have misinterpreted this finding as ameloblastoma. The epithelial lining consists of two to four layers of flattened nonkeratinizing cells, and the epithelium and connective tissue interface is flat (Fig. 15-6).
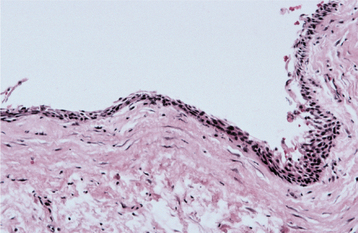
Fig. 15-6 Dentigerous cyst. This noninflamed dentigerous cyst shows a thin, nonkeratinized epithelial lining.
In the fairly common inflamed dentigerous cyst, the fibrous wall is more collagenized, with a variable infiltration of chronic inflammatory cells. The epithelial lining may show varying amounts of hyperplasia with the development of rete ridges and more definite squamous features (Fig. 15-7). A keratinized surface is sometimes seen, but these changes must be differentiated from those observed in the odontogenic keratocyst. Focal areas of mucous cells may be found in the epithelial lining of dentigerous cysts (Fig. 15-8). Rarely, ciliated columnar cells are present. Small nests of sebaceous cells rarely may be noted within the fibrous cyst wall. These mucous, ciliated, and sebaceous elements are believed to represent the multipotentiality of the odontogenic epithelial lining in a dentigerous cyst.
Gross examination of the wall of a dentigerous cyst may reveal one or several areas of nodular thickening on the luminal surface. These areas must be examined microscopically to rule out the presence of early neoplastic change.
TREATMENT AND PROGNOSIS: The usual treatment for a dentigerous cyst is careful enucleation of the cyst together with removal of the unerupted tooth. If eruption of the involved tooth is considered feasible, then the tooth may be left in place after partial removal of the cyst wall. Patients may need orthodontic treatment to assist eruption. Large dentigerous cysts also may be treated by marsupialization. This permits decompression of the cyst, with a resulting reduction in the size of the bone defect. The cyst can then be excised at a later date, with a less extensive surgical procedure.
The prognosis for most dentigerous cysts is excellent, and recurrence seldom is noted after complete removal of the cyst. However, several potential complications must be considered. Much has been written about the possibility that the lining of a dentigerous cyst might undergo neoplastic transformation to an ameloblastoma. Although undoubtedly this can occur, the frequency of such neoplastic transformation is low. Rarely, a squamous cell carcinoma may arise in the lining of a dentigerous cyst (see page 700). It is likely that some intraosseous mucoepidermoid carcinomas (see page 490) develop from mucous cells in the lining of a dentigerous cyst.
ERUPTION CYST (ERUPTION HEMATOMA)
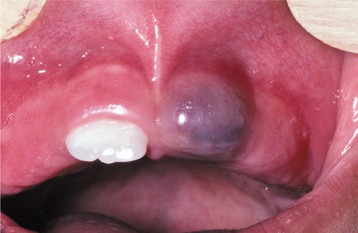
CLINICAL FEATURES: The eruption cyst appears as a soft, often translucent swelling in the gingival mucosa overlying the crown of an erupting deciduous or permanent tooth. Most examples are seen in children younger than age 10. Although the cyst may occur with any erupting tooth, the lesion is most commonly associated with the deciduous mandibular central incisors, the first permanent molars, and the deciduous maxillary incisors. Surface trauma may result in a considerable amount of blood in the cystic fluid, which imparts a blue to purple-brown color. Such lesions sometimes are referred to as eruption hematomas (Fig. 15-9).
HISTOPATHOLOGIC FEATURES: Intact eruption cysts seldom are submitted to the oral and maxillofacial pathology laboratory, and most examples consist of the excised roof of the cyst, which has been removed to facilitate tooth eruption. These show surface oral epithelium on the superior aspect. The underlying lamina propria shows a variable inflammatory cell infiltrate. The deep portion of the specimen, which represents the roof of the cyst, shows a thin layer of nonkeratinizing squamous epithelium (Fig. 15-10).
PRIMORDIAL CYST
Whether there is a primordial cyst that is not microscopically an odontogenic keratocyst is still unsettled. Many believe that all primordial cysts are odontogenic keratocysts, although some recognize the existence of a primordial cyst that does not have the histopathologic features of the odontogenic keratocyst. If such a lesion exists, then it must be exceedingly rare. Reference to this lesion is almost nonexistent in the current literature, and no reported series include a significant number of cases. In the authors’ experience, a cyst clinically considered to represent a primordial cyst, in the older meaning of the term, almost always is an odontogenic keratocyst after microscopic study (Fig. 15-11).
ODONTOGENIC KERATOCYST
Although there are wide variations in the reported frequency of odontogenic keratocysts compared with that of other types of odontogenic cysts, several studies that include large series of cysts indicate that odontogenic keratocysts make up 3% to 11% of all odontogenic cysts.
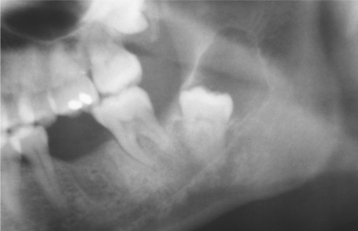
CLINICAL AND RADIOGRAPHIC FEATURES: Odontogenic keratocysts may be found in patients who range in age from infancy to old age, but about 60% of all cases are diagnosed in people between 10 and 40 years of age. There is a slight male predilection. The mandible is involved in 60% to 80% of cases, with a marked tendency to involve the posterior body and ascending ramus (Fig. 15-12).
Odontogenic keratocysts tend to grow in an anteroposterior direction within the medullary cavity of the bone without causing obvious bone expansion. This feature may be useful in differential clinical and radiographic diagnosis because dentigerous and radicular cysts of comparable size are usually associated with bony expansion. Multiple odontogenic keratocysts may be present, and such patients should be evaluated for other manifestations of the nevoid basal cell carcinoma (Gorlin) syndrome (see page 688).
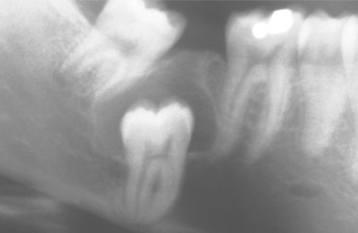
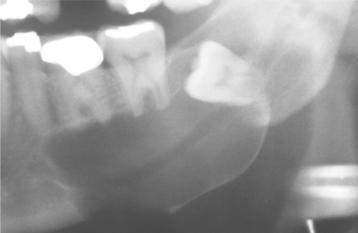
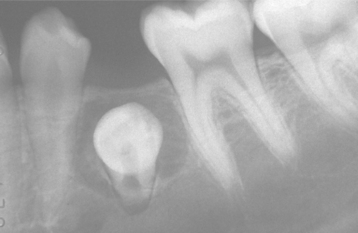
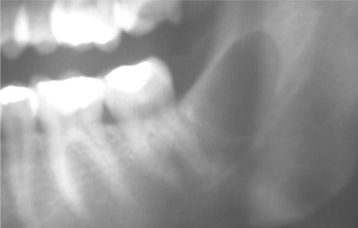
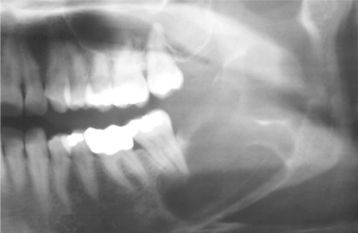
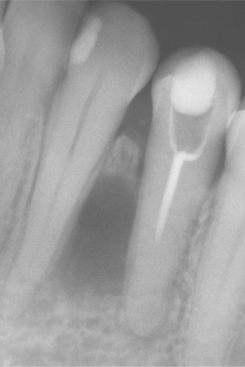
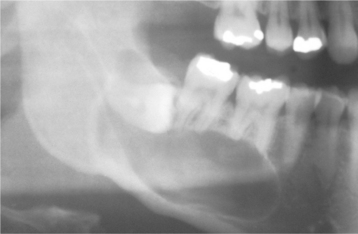
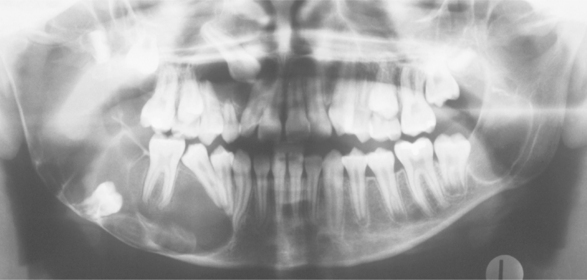
Odontogenic keratocysts demonstrate a well-defined radiolucent area with smooth and often corticated margins. Large lesions, particularly in the posterior body and ascending ramus of the mandible, may appear multilocular (Fig. 15-13). An unerupted tooth is involved in the lesion in 25% to 40% of cases; in such instances, the radiographic features suggest the diagnosis of dentigerous cyst (Figs. 15-14 and 15-15). In these cases, the cyst has presumably arisen from dental lamina rests near an unerupted tooth and has grown to envelop the unerupted tooth. Resorption of the roots of erupted teeth adjacent to odontogenic keratocysts is less common than that noted with dentigerous and radicular cysts.
The diagnosis of odontogenic keratocyst is based on the histopathologic features. The radiographic findings, although often highly suggestive, are not diagnostic. The radiographic findings in an odontogenic keratocyst may simulate those of a dentigerous cyst, a radicular cyst, a residual cyst, a lateral periodontal cyst (Fig. 15-16), or the so-called globulomaxillary cyst (which is no longer considered to be a true entity). Odontogenic keratocysts of the anterior midline maxillary region can mimic nasopalatine duct cysts. For unknown reasons, this particular subset of keratocyst usually occurs in older individuals with a mean age of nearly 70 years. Rare examples of peripheral odontogenic keratocysts within the gingival soft tissues have been reported.
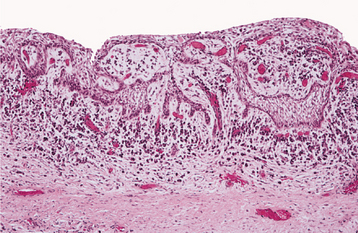
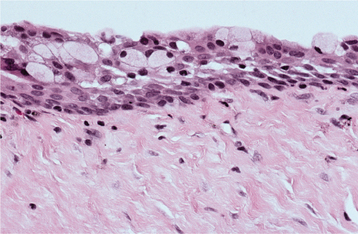
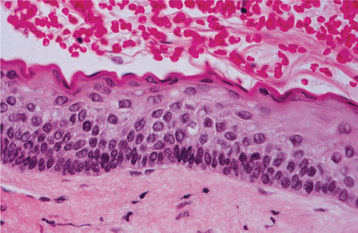
HISTOPATHOLOGIC FEATURES: The odontogenic keratocyst typically shows a thin, friable wall, which is often difficult to enucleate from the bone in one piece. The cystic lumen may contain a clear liquid that is similar to a transudate of serum, or it may be filled with a cheesy material that, on microscopic examination, consists of keratinaceous debris. Microscopically, the thin fibrous wall is essentially devoid of any inflammatory infiltrate. The epithelial lining is composed of a uniform layer of stratified squamous epithelium, usually six to eight cells in thickness. The epithelium and connective tissue interface is usually flat, and rete ridge formation is inconspicuous. Detachment of portions of the cyst-lining epithelium from the fibrous wall is commonly observed. The luminal surface shows flattened parakeratotic epithelial cells, which exhibit a wavy or corrugated appearance (Fig. 15-17). The basal epithelial layer is composed of a palisaded layer of cuboidal or columnar epithelial cells, which are often hyperchromatic. Small satellite cysts, cords, or islands of odontogenic epithelium may be seen within the fibrous wall. These structures have been present in 7% to 26% of cases in various reported series. In rare instances, cartilage has been observed in the wall of an odontogenic keratocyst.
In the presence of inflammatory changes, the typical features of the odontogenic keratocyst may be altered. The parakeratinized luminal surface may disappear, and the epithelium may proliferate to form rete ridges with the loss of the characteristic palisaded basal layer (Fig. 15-18). When these changes involve most of the cyst lining, the diagnosis of odontogenic keratocyst cannot be confirmed unless other sections show the typical features described earlier.
TREATMENT AND PROGNOSIS: Although the presence of an odontogenic keratocyst may be suspected on clinical or radiographic grounds, histopathologic confirmation is required for the diagnosis. Consequently, most odontogenic keratocysts are treated similarly to other odontogenic cysts, that is, by enucleation and curettage. Complete removal of the cyst in one piece is often difficult because of the thin, friable nature of the cyst wall. In contrast to other odontogenic cysts, odontogenic keratocysts often tend to recur after treatment. Whether this is due to fragments of the original cyst that were not removed at the time of the operation or a “new” cyst that has developed from dental lamina rests in the general area of the original cyst cannot be determined with certainty.
Many surgeons recommend peripheral ostectomy of the bony cavity with a bone bur to reduce the frequency of recurrence. Others advocate chemical cauterization of the bony cavity with Carnoy’s solution after cyst removal. Intraluminal injection of Carnoy’s solution also has been used to free the cyst from the bony wall, thereby allowing easier removal with a lower recurrence rate. After cystotomy and incisional biopsy, some surgeons have treated large odontogenic keratocysts by insertion of a polyethylene drainage tube to allow decompression and subsequent re-duction in size of the cystic cavity (Fig. 15-19). Such decompression treatment results in thickening of the cyst lining, allowing easier removal with an apparently lower recurrence rate.
Other than the tendency for recurrences, the overall prognosis for most odontogenic keratocysts is good. Occasionally, a locally aggressive odontogenic keratocyst cannot be controlled without local resection and bone grafting. In extremely rare instances, keratocysts have been seen to extend up into the skull base region. A few examples of carcinoma arising in an odontogenic keratocyst have been reported, but the propensity for an odontogenic keratocyst to undergo malignant alteration is no greater and is possibly less than that for other types of odontogenic cysts. Patients with odontogenic keratocysts should be evaluated for manifestations of the nevoid basal cell carcinoma syndrome (see page 688), particularly if the patient is in the first or second decade of life or if multiple keratocysts are identified.
ORTHOKERATINIZED ODONTOGENIC CYST
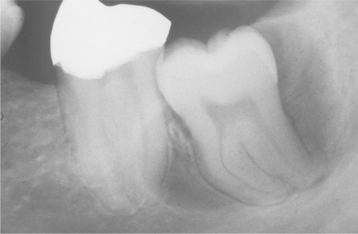
CLINICAL AND RADIOGRAPHIC FEATURES: Orthokeratinized odontogenic cysts occur predominantly in young adults and show a 2:1 male-to-female ratio. The lesion occurs twice as frequently in the mandible than the maxilla, with a tendency to involve the posterior areas of the jaws. They have no clinical or radiographic features that differentiate them from other inflammatory or developmental odontogenic cysts. The lesion usually appears as a unilocular radiolucency, but occasional examples have been multilocular. About two thirds of orthokeratinized odontogenic cysts are encountered in a lesion that appears clinically and radiographically to represent a dentigerous cyst; they most often involve an unerupted mandibular third molar tooth (Figs. 15-20 and 15-21). The size can vary from less than 1 cm to large lesions greater than 7 cm in diameter.
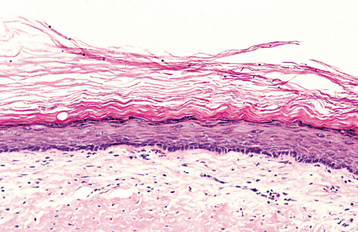
HISTOPATHOLOGIC FEATURES: The cyst lining is composed of stratified squamous epithelium, which shows an orthokeratotic surface of varying thickness. Keratohyaline granules may be prominent in the superficial epithelial layer subjacent to the orthokeratin. The epithelial lining may be relatively thin, and a prominent palisaded basal layer, characteristic of the odontogenic keratocyst, is not present (Fig. 15-22).
TREATMENT AND PROGNOSIS: Enucleation with curettage is the usual treatment for orthokeratinized odontogenic cysts. Recurrence has rarely been noted, and the reported frequency is around 2%, which is in marked contrast with the 30% or higher recurrence rate associated with odontogenic keratocysts. It has been suggested that cysts with an orthokeratinized surface may be at slightly greater risk for malignant transformation, but evidence for this is scant. Orthokeratinized odontogenic cysts have not been associated with nevoid basal cell carcinoma syndrome.
NEVOID BASAL CELL CARCINOMA SYNDROME (GORLIN SYNDROME)
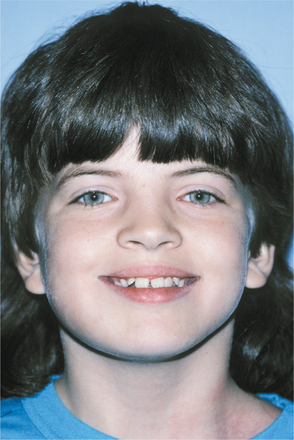
CLINICAL AND RADIOGRAPHIC FEATURES: There is great variability in the expressivity of nevoid basal cell carcinoma syndrome, and no single component is present in all patients. The most common and significant features are summarized in Box 15-2. The patient often has a characteristic facies, with frontal and temporoparietal bossing, which results in an increased cranial circumference (>60 cm in adults). The eyes may appear widely separated, and many patients have true mild ocular hypertelorism. Mild mandibular prognathism is also commonly present (Fig. 15-23).
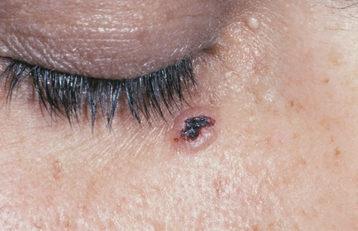
Basal cell carcinomas of the skin are a major component of the syndrome. Even though the microscopic appearance of the syndromic basal cell carcinomas is identical to that of nonsyndromic basal cell carcinoma, the syndromic lesions generally have a much less aggressive biologic behavior. The basal cell carcinomas usually begin to appear at puberty or in the second and third decades of life, although they can develop in young children. The tumors may vary from flesh-colored papules to ulcerating plaques. They often appear on skin that is not exposed to sunlight, but they are most commonly located in the midface area (Fig. 15-24). The number of skin tumors may vary from only a few to many hundreds. Blacks with the syndrome tend to develop basal cell carcinomas less frequently than whites (40% versus 90%), and they have fewer of these lesions, probably because of protective skin pigmentation.
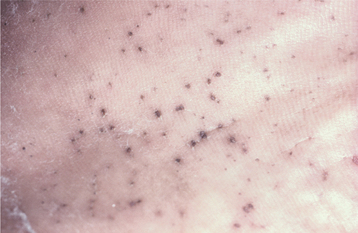
Palmar and plantar pits are present in about 65% to 80% of patients (Fig. 15-25). These punctate lesions represent a localized retardation of the maturation of basal epithelial cells, resulting in a focally depressed area as the result of a markedly thinned keratin layer. Basal cell carcinomas rarely may develop at the base of the pits.
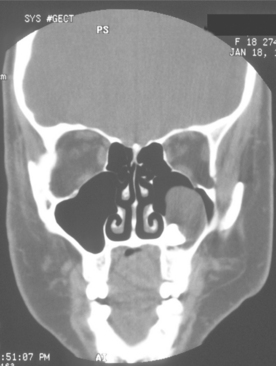
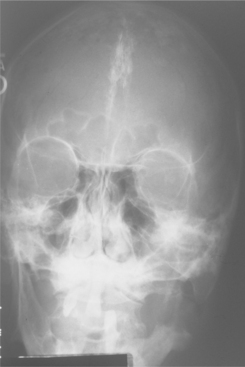
Skeletal anomalies are present in 60% to 75% of patients with this syndrome. The most common anomaly is a bifid rib or splayed ribs (Fig. 15-26). This anomaly may involve several ribs and may be bilateral. Kyphoscoliosis has been observed in about 30% to 40% of patients, and a number of other anomalies, such as spina bifida occulta and shortened metacarpals, seem to occur with unusual frequency. A distinctive lamellar calcification of the falx cerebri, noted on an anteroposterior skull radiograph or computed tomography (CT) image, is a common finding and is present in most affected patients (Fig. 15-27).
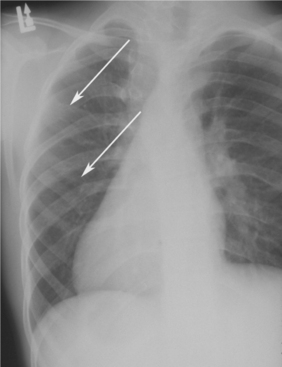
Fig. 15-26 Nevoid basal cell carcinoma syndrome. Chest film showing presence of bifid ribs (arrows).
Jaw cysts are one of the most constant features of the syndrome and are present in at least 75% of the patients. The cysts are odontogenic keratocysts, although there are some differences between the cysts in patients with nevoid basal cell carcinoma syndrome and in those with isolated keratocysts. The cysts are frequently multiple; some patients have had as many as ten separate cysts. The patient’s age when the first keratocyst is removed is significantly younger in those affected by this syndrome than in those with isolated keratocysts. For most patients with this syndrome, their first keratocyst is removed before age 19. About one third of patients with nevoid basal cell carcinoma syndrome have only a solitary cyst at the time of the initial presentation, but in most cases additional cysts will develop over periods ranging from 1 to 20 years.
Radiographically, the cysts in patients with nevoid basal cell carcinoma syndrome do not differ significantly from isolated keratocysts. The cysts in patients with this syndrome are often associated with the crowns of unerupted teeth; on radiographs they may mimic dentigerous cysts (Fig. 15-28).
HISTOPATHOLOGIC FEATURES: The cysts in the nevoid basal cell carcinoma syndrome histopathologically are invariably odontogenic keratocysts. The keratocysts in patients with this syndrome tend to have more satellite cysts, solid islands of epithelial proliferation, and odontogenic epithelial rests within the fibrous capsule than do isolated keratocysts (
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses