CHAPTER 13. Diseases of the oral mucosa: non-infective stomatitis
TRAUMATIC ULCERS → Summaries pp. 246, 247
Traumatic ulcers are usually caused by biting, denture trauma or chemical trauma and arise at trauma-prone sites such as lip, buccal mucosa or adjacent to a denture flange. They are tender, have a yellowish-grey floor of fibrin slough and red margins (Figs. 13.1 and 13.35). Inflammation, swelling and erythema are variable, depending on the cause and time since trauma. There is no induration unless the site is scarred from repeated episodes of trauma. If caused by the sharp edge of a broken-down tooth, they are usually on the tongue or buccal mucosa. Occasionally, a large ulcer is caused by biting after a dental local anaesthetic (see Fig. 33.1).
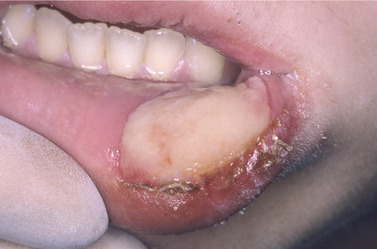 |
| Fig. 13.1
A large traumatic ulcer on the lower lip. Note the colour of the fibrin slough, distinct from the keratin of a white patch, and the well-defined epithelial margin with minimal inflammation.
|
Traumatic ulcers heal a few days after elimination of the cause. If they persist for more than 7–10 days, or there is any other cause for suspicion as to the cause, biopsy should be carried out.
LINGUAL PAPILLITIS
This condition, also known as transient lingual papillitis or fungiform papillitis, is common but patients rarely seek treatment. One or a cluster of fungiform papillae become slightly swollen, white and intensely painful to touch. The condition resolves spontaneously after a few days, sometimes after only one. The cause is unknown but trauma and certain foods are usually blamed. Biopsy does not aid diagnosis.
RECURRENT APHTHOUS STOMATITIS (RECURRENT APHTHAE) → Summaries pp. 221, 246
Recurrent aphthae constitute the most common oral mucosal disease and affect 10–25% of the population, but many cases are mild and no attention is sought for their relief.
Aetiology
The main factors thought to contribute are shown in Box 13.1.
Box 13.1
Possible aetiological factors for recurrent aphthae
• Genetic predisposition
• Exaggerated response to trauma
• Infections
• Immunological abnormalities
• Gastrointestinal disorders
• Haematological deficiencies
• Hormonal disturbances
• Stress
Genetic factors
There is some evidence for a genetic predisposition. The family history is sometimes positive and the disease appears to affect identical twins more frequently than non-identical. However, this probably applies to a minority. A variety of HLA associations have been reported, but no one haplotype seems to be consistently associated. In the possibly related Behçet’s disease (see below), the evidence for a genetic predisposition is much stronger.
Trauma
Some patients think that the ulcers result from trauma because the early symptoms simulate pricking of the mucosa by (for instance) a toothbrush bristle. Trauma may dictate the site of ulcers in patients who already have the disorder, but most aphthae are in relatively protected sites and the masticatory mucosa is generally spared.
Infections
There is no evidence that aphthae are directly due to any microbes and there is scanty evidence that cross-reacting antigens from streptococci or L-forms play any significant role. The hypothesis that there may be defective immunoregulation caused by herpes or other viruses is unproven.
Immunological abnormalities
Since the aetiology of recurrent aphthae is unknown, there has been a facile tendency to label them as ‘autoimmune’. A great variety of immunological abnormalities have been reported, but there have been almost as many contrary findings and no convincing theory of immunopathogenesis takes into account the clinical features. It is also possible that the immunological abnormalities are as much a consequence of the ulcers as the cause. Evidence of an association with atopic (IgE-mediated) disease is unconfirmed. Circulating antibodies to crude extracts of fetal oral mucosa have been reported, but their titre is unrelated to the severity of the disease and, in many patients, there are no significant changes in immunoglobulin levels. Antibody-dependent cytotoxic mechanisms have been postulated, but not convincingly demonstrated. The histological features of aphthae (see below) have also been invoked to support hypotheses that the disease is either an immune complex-mediated (type III) or a cell-mediated (type IV) reaction according to taste. However, others have failed to confirm the presence of circulating immune complexes and, in any case, the significance of such complexes, which are sometimes detectable in the absence of disease, is notoriously difficult to interpret. Depressed circulating helper/suppressor T-lymphocyte ratios have been reported, but others have found no difference between active and remittant phases of the disorder. Recurrent aphthae also lack virtually all features of and any association with typical autoimmune diseases (Ch. 23). They also fail to respond reliably to immunosuppressive drugs and become more severe in the immune deficiency state induced by HIV infection.
Gastrointestinal disease
Aphthae were previously known as ‘dyspeptic ulcers’, but are only rarely associated with gastrointestinal disease. Any association is usually because of a deficiency, particularly of vitamin B 12 or folate secondary to malabsorption. An association with coeliac disease (sometimes asymptomatic) has been found in approximately 5% of patients with aphthae, but a secondary haematinic deficiency, particularly folate deficiency, is probably the cause.
Haematological deficiencies
Deficiencies of vitamin B 12, folate, or iron have been reported in up to 20% of patients with aphthae. Such deficiencies are probably more frequent in patients whose aphthae start or worsen in middle age or later. In many such patients, the deficiency is latent, the haemoglobin is within normal limits and the main sign is micro- or macrocytosis of the red cells. In patients who thus prove to be vitamin B 12 or folate deficient, remedying the deficiency may bring rapid resolution of the ulcers.
Hormonal factors
In a few women, aphthae are associated with the stressful luteal phase of the menstrual cycle, but there is no strong evidence that hormone treatment is reliably effective.
‘Stress’
Some patients relate exacerbations of ulceration to times of stress and some studies have reported a correlation. However, stress is notoriously difficult to quantify and some studies have found no correlation.
HIV infection
Aphthous stomatitis is a recognised feature of HIV infection. Its frequency and severity are related to the degree of immune deficiency, as discussed later.
Non-smoking
It has long been established that recurrent aphthae are a disease, almost exclusively, of non-smokers. And this is one of the few consistent findings. Recurrent aphthae may also start when smoking is abandoned. The reasons are unclear but it is believed that smoking has a systemic protective action against this disease.
In brief, therefore, the aetiology of recurrent aphthae is unclear. There is no evidence that they are a form of autoimmune disease in any accepted sense and it is uncertain whether many of the reported immunological abnormalities are cause or effect. However, in a minority of patients there is a clear association with haematological deficiencies. The latter, in turn, may be secondary to small-intestine disease or other cause of malabsorption.
That speculation about the cause of recurrent aphthae has continued for at least half a century, the variety of current theories and the contradictory findings indicate how little is known.
Clinical features
Typical features of recurrent aphthae are summarised in Box 13.2.
Box 13.2
Typical features of recurrent aphthae
• Onset frequently in childhood but peak in adolescence or early adult life
• Attacks at variable but sometimes relatively regular intervals
• Most patients are otherwise healthy
• A few have haematological defects
• Most patients are non-smokers
• Usually self-limiting eventually
Females are not significantly more frequently affected than males. The frequency of ulceration typically reaches a peak in early adult life or a little later, then gradually wanes. Recurrent aphthae are rare in the elderly, particularly the edentulous. However, older persons may be affected if a haematological deficiency develops. The great majority of patients are clerical, semi-professional, or professional workers and are total non-smokers. Occasionally, aphthae start when smoking is given up.
The usual history is of painful ulcers recurring at intervals of approximately 3 to 4 weeks. Occasionally, they are continuously present. Unpredictable remissions of several months may be noted. Individual minor aphthae persist for 7–10 days then heal without scarring. Aphthae typically affect only the non-keratinised mucosa such as the buccal mucosa, sulcuses, or lateral borders of the tongue, but major aphthae can affect the masticatory mucosa. Ulcers are of three clinically distinguishable types (Box 13.3).
Box 13.3
Types of recurrent aphthae
Minor aphthae (Fig. 13.2)
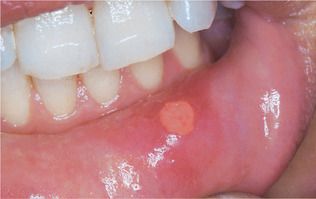 |
| Fig. 13.2
Aphthous stomatitis, minor form. A single, relatively large shallow ulcer in a typical site. There is a narrow band of periulcer erythema. These features are non-specific and the diagnosis must be made primarily on the basis of the history.
|
• The most common type
• Non-keratinised mucosa affected
• Ulcers are shallow, rounded, 5–7 mm across, with an erythematous margin and yellowish floor
• One or several ulcers may be present
Major aphthae (Fig. 13.3)
• Uncommon
• Ulcers frequently several centimetres across
• Sometimes mimic a malignant ulcer
• Ulcers persist for several months
• Masticatory mucosa such as the dorsum of the tongue or occasionally the gingivae may be involved
• Scarring may follow healing (Fig. 13.4)
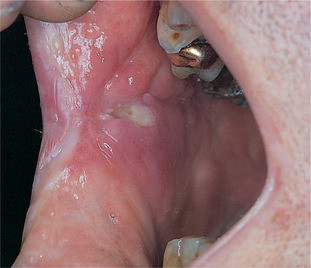 |
| Fig. 13.4
Recurrent aphthous stomatitis, major type. The same ulcer shown in Figure 13.3, but healing. The ulcer is much smaller but there is scarring and puckering of the surrounding mucosa.
|
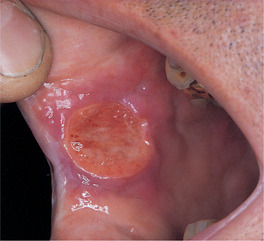 |
| Fig. 13.3
Aphthous stomatitis, major type. This large, deep ulcer with considerable surrounding erythema has been present for several weeks.
|
Herpetiform aphthae (Fig. 13.5)
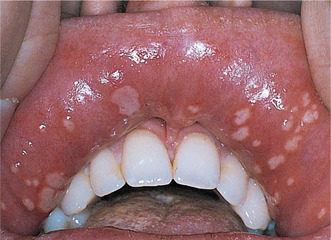 |
| Fig. 13.5
Recurrent aphthous stomatitis, herpetiform type. There are numerous small, rounded and pinpoint ulcers, some of which are coalescing. The surrounding mucosa is lightly erythematous and the overall picture is highly suggestive of viral infection, but the attacks are recurrent and no virus can be isolated.
|
• Uncommon
• Non-keratinised mucosa affected
• Ulcers are 1–2 mm across
• Dozens or hundreds may be present
• May coalesce to form irregular ulcers
• Widespread bright erythema round the ulcers
The pain of major aphthae can interfere with eating. Moreover, major aphthae are sometimes a feature of HIV disease and add to such patients’ burdens.
Pathology
There is alleged to be initial lymphocytic infiltration followed by destruction of the epithelium and infiltration of the tissues by neutrophils. Mononuclear cells may also surround blood vessels (perivascular cuffing). These changes are said to be consistent with either type III or IV reactions, but true vasculitis is not seen. Overall, the appearances are non-specific (Fig. 13.6).
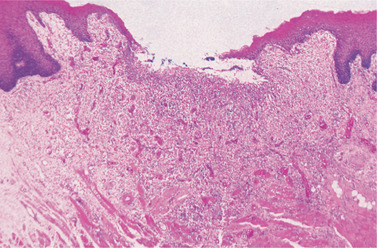 |
| Fig. 13.6
Recurrent aphtha. Section of an early ulcer showing the break in the epithelium, the inflammatory cells in the floor and the inflammatory changes more deeply where numerous dilated vessels can be seen.
|
Biopsy is of no value in the diagnosis except to exclude carcinoma in the case of major aphthae. Aphthae are not preceded by vesiculation and smears readily distinguish herpetiform aphthae from herpetic ulceration.
Diagnosis and management
The most important diagnostic feature is the history of recurrences of self-healing ulcers at fairly regular intervals (Table 13.1). The only other condition with this history is Behçet’s disease. Usually, increasing frequency of ulcers brings the patient to seek treatment. Otherwise, most patients appear well, but haematological investigation is particularly important in older patients. Routine blood indices are informative and usually the most important finding is an abnormal mean corpuscular volume (MCV). If macro- or microcytosis is present, further investigation is necessary to find and remedy the cause. Treatment of vitamin B 12 deficiency or folate deficiency is sometimes sufficient to control or abolish aphthae.
| Check for | Comments | |
|---|---|---|
| History |
• Recurrences
• Pattern? Minor, major or herpetiform type?
• Onset as child or teenager
• Family history
• Distribution only on non-keratinised mucosa
• Signs or symptoms of Behçet’s disease (ocular, genital, skin, joint lesions (Box 13.4)
|
The history is all-important |
| Examination |
• Discrete well-defined ulcers
• Scarring or soft palate involvement suggesting major aphthae
|
Exclude other diseases with specific, appearances, e.g. lichen planus or vesiculobullous disease |
| Special investigations |
• Anaemia, iron, red cell folate and vitamin B 12 status
• History of diarrhoea, constipation or blood in stools suggesting gastrointestinal disease, e.g. coeliac disease or malabsorption
|
Used to exclude underlying conditions, especially in patients with onset in later life |
Apart from the minority with underlying systemic disease, treatment is empirical and palliative only. Despite numerous clinical trials, no medication gives completely reliable relief. Patients should therefore be made to understand that the trouble may not be curable, but can usually be alleviated and usually resolves eventually of its own accord.
Corticosteroids
Some patients get relief from Corlan (hydrocortisone hemisuccinate 2.5 mg) pellets allowed to dissolve in the mouth three times a day. Corticosteroids are unlikely to hasten healing of existing ulcers, but probably reduce the painful inflammation. The most rational form of treatment is for patients to take these pellets continuously (whether or not ulcers are present) to enable the corticosteroid to act in the very early, asymptomatic stages. This regimen is only applicable to those who have frequent ulcers (at 2- or 3-week intervals or more frequently). This should be tried for 2 months then stopped for a month to assess any improvement and whether there is any deterioration without treatment.
Triamcinolone dental paste
This is a corticosteroid in a vehicle which sticks to the moist mucosa. When correctly applied the vehicle absorbs moisture and forms an adhesive gel which can remain in place for one or several hours, but it is difficult to apply a fragment of this paste to the ulcer and to get it to adhere firmly. It is only useful for patients with infrequent ulcers, for ulcers near the front of the mouth and for patients dextrous enough to be able to follow the instructions. This gel should form a protective layer over the ulcer to help make it comfortable. The corticosteroid is slowly released and has an anti-inflammatory action. Another alternative is the use of a corticosteroid asthma spray to deposit a potent corticosteroid over the ulcer. Topical corticosteroids used as described have no systemic effect.
Tetracycline mouth rinses
Trials in both Britain and the USA showed that tetracycline rinses significantly reduced both the frequency and severity of aphthae. For herpetiform aphthae particularly, the contents of a tetracycline capsule (250 mg) can be stirred in a little water and held in the mouth for 2–3 minutes, three times daily. Some patients like to use this mouth rinse regularly for 3 days each week if they have frequent ulcers. An antifungal drug may also need to be given to patients who are susceptible to superinfection by Candida albicans.
Chlorhexidine
A 0.2% solution has also been used as a mouth rinse for aphthae. Used three times daily after meals and held in the mouth for at least 1 minute, it has been claimed to reduce the duration and discomfort of aphthous stomatitis. Zinc sulphate or zinc chloride solutions may also have a slight beneficial effect.
Topical salicylate preparations
Salicylates have an anti-inflammatory action and also have local effects. Preparations of choline salicylate in a gel can be applied to aphthae. These preparations, which are available over the counter, sometimes appear to be helpful.
Possible treatments for recurrent aphthae are summarised in Appendix 13.1.
Treatment of major aphthae
Major aphthae, whether or not there is underlying disease such as HIV infection, may sometimes be so painful, persistent and resistant to conventional treatment as to be disabling. Reportedly effective treatments include azathioprine, cyclosporin, colchicine and dapsone, but thalidomide is probably most reliably effective. Their use may be justified for major aphthae even in otherwise healthy persons if they are disabled by the pain and difficulty of eating. However, thalidomide can cause severe adverse effects and is strongly teratogenic and, like the other drugs mentioned, can only be given under specialist supervision.
BEHÇET’S DISEASE → Summary p. 246
Behçet’s syndrome was originally defined as a triad of oral aphthae, genital ulceration and uveitis. However, it is a multisystem disorder with varied manifestations and now termed Behçet’s disease. It is rare in the UK and USA, but is particularly common in Turkey (Behçet was a Turk) and very common in Japan. It is said that a high prevalance of Behçet’s disease exists in races along the Silk Road1 but this is only a vague generalisation.
The aetiology is unknown but it has features, such as circulating immune complexes, suggesting immune complex disease, but any antigen remains unidentified, though there is some evidence for involvement of a virus, possibly herpes simplex. The immunological changes are similar to those found in aphthous stomatitis.
Patients are usually young adult males between 20 and 40 years old. Patients suffer one of four patterns of disease, namely:
1. Mucocutaneous (oral and genital ulceration)
2. Arthritic (joint involvement with or without mucocutaneous involvement)
3. Neurological (with or without other features) or
4. Ocular (with or without other features).
The racial distribution suggests a strong genetic component and this is confirmed by the fact that each of these presentations is linked to a different HLA tissue type. HLA-B12 and/or DR2 types, for example, are linked to mucocutaneous and arthritic disease. Unfortunately, these HLA associations are of no value in diagnosis, but HLA-B51 is useful as a predictor of ocular lesions, which can lead to blindness. There is also an association with HLA-B5 and HLA-B51(B5101). Diagnostic criteria are listed in Box 13.4.
Box 13.4
Diagnostic criteria for Behçet’s disease
Major criteria
• Recurrent oral aphthae
• Genital ulceration
• Eye lesions (recurrent hypopyon, iritis or iridocyclitis; chorioretinitis)
• Skin lesions (erythema nodosum, subcutaneous thrombophlebitis, hyperirritability)
Minor criteria
• Arthralgia or arthritis
• Gastrointestinal lesions
• Vascular lesions (mainly thrombotic) (Fig. 13.7)
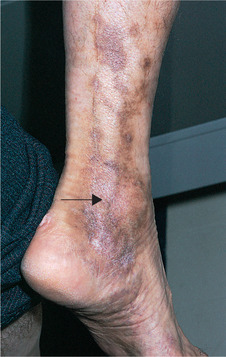 |
| Fig. 13.7
Thrombophlebitis in Behçet’s disease. Inflammation and pigmentation highlight the sites of veins (arrow) and their valves. This is only one of several possible skin manifestations.
|
• Central nervous system involvement
Oral aphthae are the most consistent feature and are not distinguishable from common aphthae. They are frequently the first manifestation. Behçet’s disease should therefore be considered in the differential diagnosis of aphthous stomatitis, particularly in patients in an at-risk racial group, and the medical history should be checked for the features shown in Box 13.4. The frequency of other manifestations is highly variable. As a result there are no absolute criteria or reliable tests for the diagnosis, but aphthous stomatitis in combination with any two of the other major features can be regarded as adequate.
Special tests are not helpful in diagnosis, apart from the pathergy test. The test is positive if there is an exaggerated response to a sterile needle puncture of the skin. However, the test must be interpreted by an experienced clinician and tends to be positive only in Mediterranean patients. Moreover, a positive pathergy test does not correlate with the presence of oral lesions or with the overall severity of the disease and is rarely positive in UK patients. It is also not entirely specific for Behçet’s disease.
The importance of making the diagnosis is indicated by the life-threatening nature of thrombosis and the risk of blindness or brain damage.
The main pathological finding is vasculitis and anti-endothelial cell (aEC) antibodies are reportedly found in 50% of those with active disease, but their role remains uncertain. The aetiology is largely speculative, but lesions may result from immune-complex mediated vasculities of small vessels which is said to be present in mucocutaneous and all types of lesion. However, microscopic evidence of vasculitis in the oral ulcers is open to question.
Management
Treatment is difficult and requires a multidisciplinary approach. The oral ulceration may be treated in the same way as the common form. When major aphthae are particularly severe or if there is frequent recurrence, colchicine may be helpful but thalidomide may be most effective particularly for HIV-associated aphthae.
NICORANDIL-INDUCED ORAL ULCERATION
The potassium channel activator Nicorandil, used for angina, causes ulcers very similar to major aphthae but without the recurrent pattern.
Ulcers appear within a year of starting the drug, often a few weeks, and are usually solitary on the lateral tongue, buccal mucosa, gingivae or fauces. They persist for several months unless the drug is withdrawn, when they heal within a few weeks depending on their size. Biopsy shows only non-specific inflammatory features and does not aid diagnosis other than to exclude other causes.
HIV-ASSOCIATED ORAL ULCERATION
Patients with HIV infection (Ch. 24) are susceptible to severe recurrent aphthae which are not otherwise distinguishable from common aphthae. With declining immune function, the ulcers become more frequent and severe and, by interfering with eating, may contribute to the patient’s deterioration. Biopsies should be taken to exclude opportunistic infections, otherwise the aetiology is unknown. Treatment with thalidomide is most frequently effective but, in some cases, cytomegalovirus can be seen in biopsies, and treatment with ganciclovir may be more appropriate.
Necrotising gingival ulceration may also be seen.
LICHEN PLANUS → Summaries pp. 232, 246, 247, 277, 278, 279
Lichen planus is a common chronic inflammatory disease of skin and mucous membranes. It mainly affects patients of middle age or over. Oral lesions have characteristic appearances and distribution.
Aetiology
In spite of histological changes which can be diagnostic, the aetiology of lichen planus remains problematical. The predominantly T-lymphocyte infiltrate suggests cell-mediated immunological damage to the epithelium and a plethora of immunological abnormalities has been reported. Though it has not been possible to demonstrate humoral or lymphocytotoxic mechanisms, the inflammatory infiltrate consists mainly of T-lymphocytes. Both CD4 and CD8 cells are present, but numbers of CD8 cells may rise with disease progression and they are more numerous in relation to the epithelium. Precise trigger mechanisms remain unclear, but lichen planus undoubtedly appears to be a T-lymphocyte mediated disorder.
Disease indistinguishable from lichen planus, induced by drugs (notably gold and anti-malarial agents), also suggests involvement of immunological mechanisms. Oral lichen planus is also a virtually invariable feature and an early sign of graft-versus-host disease (Ch. 23), but this does not clarify any immunological mechanisms.
In some countries, hepatitis C infection is thought to be contributory.
Clinical features
Typical features of lichen planus are summarised in Box 13.5.
Box 13.5
Oral lichen planus: typical features
• Females account for at least 65% of patients
• Patients usually over 40 years
• Untreated disease can persist for 10 or more years
• Lesions in combination or isolation, comprise:
Striae
Atrophic areas
Erosion
Plaques
• Common sites are:
Buccal mucosae
Dorsum of tongue
Gingivae (infrequently)
• Lesions usually bilateral and often symmetrical
• Cutaneous lesions only occasionally associated
• Usually, good response to corticosteroids
The lesions’ characteristic appearance and distribution should be taken into account in making the diagnosis.
Striae are most common and typically form sharply-defined snowy-white, lacy, starry, or annular patterns (Fig. 13.8). They may occasionally be interspersed with minute, white papules. Striae may not be palpable or may be firmer than the surrounding mucosa.
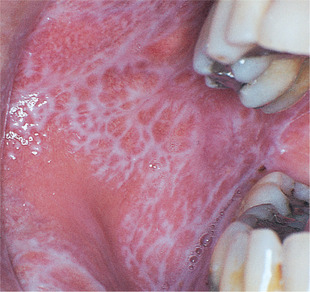 |
| Fig. 13.8
Lichen planus, striate pattern. This is the most common site and type of lesion, a lacy network of white striae on the buccal mucosa. The lesions are usually symmetrically distributed.
|
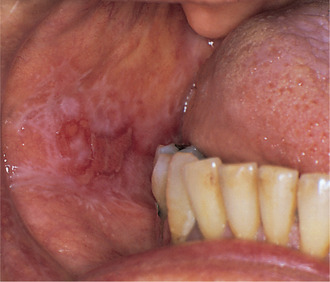
Atrophic areas are red areas of mucosal thinning (Fig. 13.9) and often combined with striae.
 |
| Fig. 13.9
Lichen planus, atrophic type. There are shallow irregular zones of erythema surrounded by poorly defined striae.
|
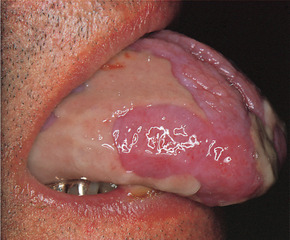
Erosions are shallow irregular areas of epithelial destruction. These also can be very persistent and may be covered by a smooth, slightly raised yellowish layer of fibrin (Fig. 13.10). The margins may be slightly depressed due to fibrosis and gradual healing at the periphery. Striae may radiate from the margins of these erosions.
 |
| Fig. 13.10
Severe erosive lichen planus. Thick plaques of fibrin cover extensive ulcers on the dorsum of the tongue in this case.
|
Plaques are occasionally seen in the early stages particularly on the dorsum of the tongue. Otherwise they may result from persistent disease and mainly affect the buccal mucosa.
Distribution
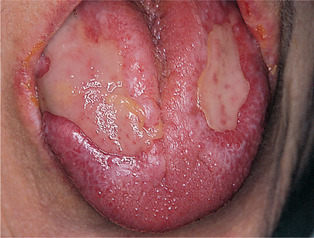
The buccal mucous membranes, particularly posteriorly, are by far the most frequently affected, but lesions may spread forward almost to the commissures. The next most common site is the tongue, either the edges or the lateral margins of the dorsum, or less frequently the centre of the dorsum. The lips and gingivae may also occasionally be affected, but the floor of the mouth and palate usually escape. Lesions are very often symmetrical, sometimes strikingly so (Fig. 13.11).
 |
| Fig. 13.11
Erosive lichen planus of the tongue. Two extensive areas of shallow ulceration have formed symmetrically on the tongue. Lingual involvement is seen usually in severe cases where the buccal mucosa and other sites are affected.
|
Symptoms
Striae alone may be asymptomatic and unnoticed by the patient, or cause roughness or slight stiffness of the mucosa. Atrophic lesions are sore and erosions usually cause more severe symptoms still and may make eating difficult.
Gingival lichen planus → Summary p. 279
The gingivae are occasionally the only site of lichen planus which needs to be distinguished from other forms of gingivitis. Lesions are usually atrophic so that the gingivae appear shiny, inflamed and smooth (‘desquamative gingivitis’) (Fig. 13.12). Striae are uncommon on the gingivae but sometimes present in other parts of the mouth. Only limited segments of the gingivae may be affected.
Soreness caused by atrophic lesions makes toothbrushing difficult. Plaque accumulation and associated inflammatory changes appear to aggravate lichen planus. The contribution of local irritation to lichen planus is suggested by disappearance of the lesions when the teeth are extracted. Lichen planus of the denture-bearing area is virtually unknown.
Skin lesions
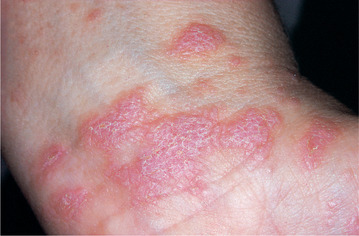
Lichen planus is a common skin disease, but skin lesions are uncommon in those who complain of oral symptoms. Skin lesions typically form purplish papules, 2–3 mm across with a glistening surface marked by minute fine striae and are usually itchy. Typical sites are the flexor surface of the forearms and especially the wrists (Fig. 13.13). Skin lesions help, but are not essential, to confirm the diagnosis of oral lichen planus.
 |
| Fig. 13.13
Dermal lichen planus. This, the flexor surface of the wrists, is a characteristic site. The lesions consist of confluent papules with a pattern of minute white striae on their surface.
|
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


