The State of the Evidence in Implant Prosthodontics
Gary R. Goldstein
New York University College of Dentistry, New York, New York, USA
Introduction
Okay, you have been placing and/or restoring implants for numerous years and are pleased with your clinical outcomes and your patient acceptance of this exciting treatment modality. You attend a lecture or read an article about a new product or technique that claims to have a higher insertion torque, less bone loss, etc.; so, how do you decide if you should switch? The rubrics are very simple, so, whether you read a paper in a peer‐reviewed journal, a non‐peer‐reviewed journal, or hear it in a lecture, the rules are the same for all three.
There is a multitude of information available to the clinician, some evidence‐based, some theory‐based, some compelling and, unfortunately, some useless. Evidence‐based dentistry (EBD) gives one the tools to evaluate the literature and scientific presentations. It constructs a hierarchy of evidence which allows the reader to put what they are reading, or hearing, into perspective. As we proceed on this short trail together, I want to state that there is no substitute for your own clinical experience and common sense, and hope that when you are done with this chapter you will understand why. I am not here to trash the literature, rather to propose that not all published works are equal.
Hierarchy of evidence
EBD is a relatively new phenomenon that was introduced in the 1990s. It evolved slowly due to misunderstandings and misrepresentations of what it is and what it means, and, despite a slow start, has picked up traction and is now an ADA Commission on Dental Accreditation (CODA) requirement, mandatory in dental education and the backbone of clinical research and practice. Journal editors and reviewers are well versed in the process and less likely to approve the methodologically flawed project for publication, putting more pressure on the researcher to pay heed to research design.
I could say, “Here is the hierarchy of evidence (Figure 1.1),” and save us, you the reader and me the author, a lot of time, but unfortunately things are not quite that simple. Routinely, if one is asked what the best evidence is, the response would be a meta‐analysis or systematic review and, not having that, a randomized controlled trial (RCT). What is also obvious from the figure is the categorization of animal and laboratory studies. While these present critical contributions to our basic knowledge and the background information needed to design clinical studies, they cannot and should not be utilized to make clinical decisions.
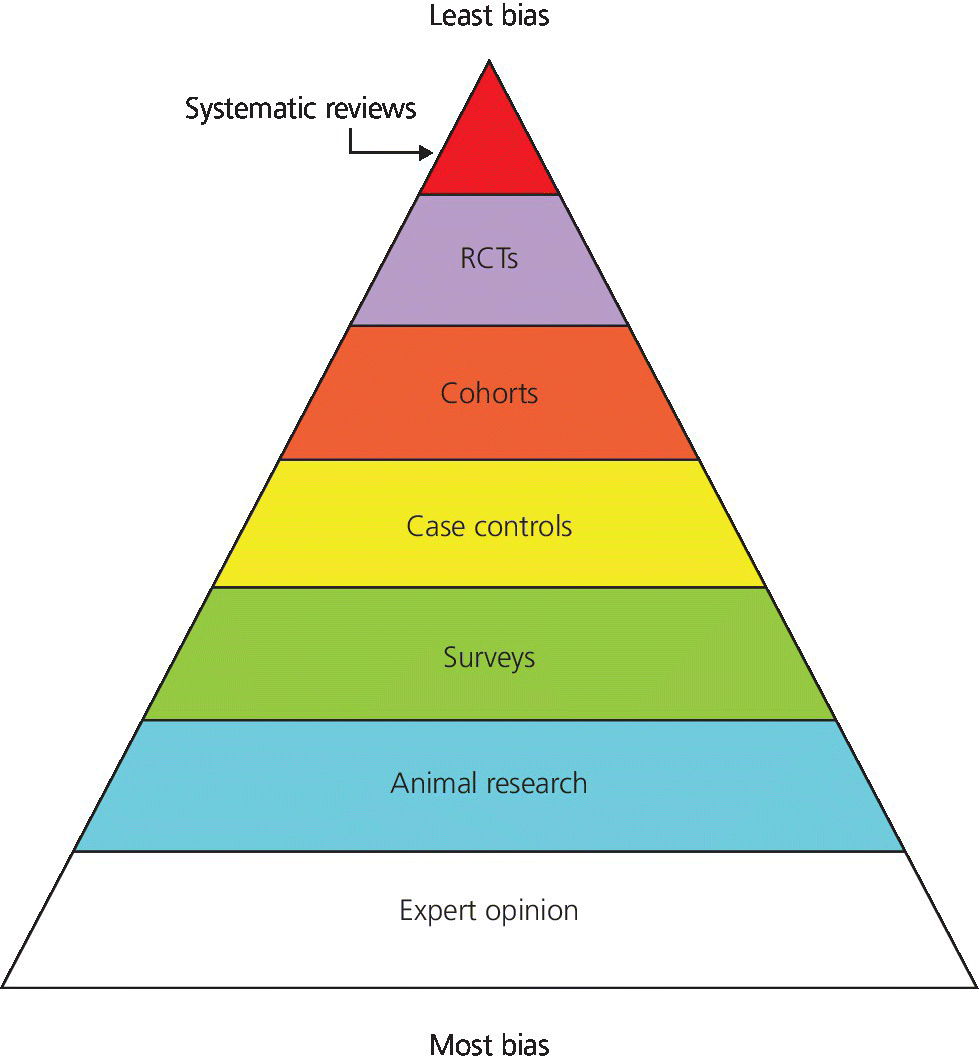
Figure 1.1 Hierarchy of evidence.
Source: Adapted from http://consumers.cochrane.org.
According to the Cochrane Collaboration,1 a Systematic Review (SR) “summarises the results of available carefully designed healthcare studies (controlled trials) and provides a high level of evidence on the effectiveness of healthcare interventions”; and a meta‐analysis (MA) is a SR where the authors pool numerical data. I want to bring your attention to the fact that nowhere in the definition does it mention, or limit itself to, RCTs. SRs and MAs are different from the more typical narrative review where an investigator evaluates all, or much, of the available literature and tenders an “expert opinion” of the results. They usually have loose or no inclusion and exclusion criteria and no “ranking” of the articles being reviewed. For those interested in how one categorizes articles, the following websites would be helpful:
- http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1891443/
- http://www.nature.com/ebd/journal/v10/n1/fig_tab/6400636f1.html#figure‐title
- http://www.ebnp.co.uk/The%20Hierarchy%20of%20Evidence.htm
We can break down studies into analytic or comparative, those that have a comparative group (randomized controlled trials, concurrent cohort studies, and case control studies) and descriptive, those that do not have a comparative group (cross‐sectional surveys, case series, and case reports). Descriptive studies give us useful information about a material, treatment, etc., however, to determine if one material, treatment, etc. is better than another, requires a comparative study.
In addition, studies may be prospective or retrospective. In a prospective study the investigator selects one or more groups (cohorts) and follows them forward in time. In a retrospective study the investigator selects one or more cohorts and looks backwards in time. Prospective studies are considered superior since they can ensure that the cohorts were similar for possible confounding variables at the beginning of the study, that all participants were treated equally, and that dropouts are known and accounted for. Prospective studies allow for randomization or prognostic stratification of the cohorts. Retrospective studies can be very valuable and should not be minimalized, especially in uncovering adverse outcomes that have a low prevalence or take many years to become evident. The adverse effects from smoking have mostly been uncovered by retrospective investigation.
A randomized controlled trial (RCT) is a prospective, comparative study in which the assignment to the treatment or control group is done using a process analogous to flipping a coin. In reality, most projects are randomized utilizing a computer‐generated random assignment protocol. The sole advantage of randomization is that it eliminates allocation bias. Feinstein2 and Brunette3 feel that the universal dependence on RCTs to achieve this is overestimated and prefer prognostic stratification of the matched cohorts for major confounding variables prior to allocation. What soon becomes obvious, however, is that prognostic stratification is not possible for every potential confounding variable, so only “major” ones are usually accounted for.
Doing an RCT is ideal, but has the constraints of time (can we afford to wait the numerous years necessary to design, implement and publish?) and cost (where can you get the funding?). Furthermore, RCTs are only ideal for certain questions, for example one that involves therapy. If our question is one of harm, it would be unethical to randomize a patient to something with a known harmful effect. To my knowledge, there has never been a RCT that proved smoking was harmful. Could you get an Internal Review Board (IRB) or Ethics Committee approval to assign participants to a group that had to smoke two packs of cigarettes a day for 25 years? Yet, does anyone doubt, given the mass of clinical evidence, that it is better to not smoke? Ultimately, the design is determined by the question.
Sackett,4 considered by many to be the father of evidence‐based medicine, in response to the heated dialogue over which design was the best, and in an effort to refocus the time, intellect, energy, and effort being wasted, proposed that “the question being asked determines the appropriate research architecture, strategy, and tactics to be used – not tradition, authority, experts, paradigms, or schools of thought.”
Causation is one of the most difficult things to prove. It is like approaching a single set of railroad tracks. One can feel the warmth of the track and know that a train passed, but in which direction? It is why many studies conclude a “correlation.” In the EBM series authored by the McMaster faculty, the Causation section published in the Canadian Medical Journal had David Sackett using the pseudonym Prof. Kilgore Trout as the corresponding author.5 One can only wonder what motivated him to use the pseudonym rather than his own name to write on this critical topic. Sackett’s love of the works of Kurt Vonnegut is well known and one might wonder if, in fact, his Canadian home named the Trout Research & Education Centre, is based on Kilgore or the fish?
While the design is critical, one must also determine the validity of the methodology. According to Jacob and Carr,6 internal validity is a reflection of how the study was planned and carried out and is threatened by bias and random variation; while external validity defines if the results of the study will be applicable in other clinical settings.
Bias
There are many types of biases and a full explanation of the multitude reported is beyond the scope of this chapter. Still, there are a few that are meaningful to us as clinicians. We can divide bias into the following groups: the reader, the author, and the journal.
The reader
Taleb7 used the following quote to accent that past experience is not always the best method to judge what we are doing at the present time.
But in all my experience, I have never been in an accident…of any sort worth speaking about. I have seen but one vessel in distress in all my years at sea. I never saw a wreck and have never been wrecked nor was I ever in any predicament that threatened to end in disaster of any sort.
E.J. Smith, 1907, Captain RMS Titanic.
The reader is almost always subject to confirmation bias, which is to believe whatever confirms one’s beliefs. It was best stated by Sir Francis Bacon8 : “The human understanding, once it has adopted an opinion, collects any instances that confirm it, and though the contrary instances may be more numerous and more weighty, it either does not notice them or rejects them, in order that this opinion will remain unshaken.” People seek out research in a manner that supports their beliefs. We have all invested time, energy, and money getting a dental education. We have successfully treated patients and are loath to admit that something we have been doing is not as useful, successful, good, etc., as another product, technique, or procedure. This is a form of cognitive dissonance and a common human reaction. It is difficult for a clinician, and especially an educator, to admit that what they have been doing and/or teaching is not currently the best for our patients. Remember, we performed the procedure with “older” information and materials and are evaluating our outcomes or planning new treatment with “newer” evidence. The best recourse is self‐reflection. Keeping up to date with clinically proven advances is our obligation as health providers.
The author
Allocation bias, a type of selection bias, is present when the two or more groups being compared are not similar, especially for confounding variables that could affect the outcome of the study. Familiar examples could be smoking, diabetes, osteoporosis, etc. Theoretically, randomization will account for this and is its major advantage, but only in the presence of a compelling number of participants (N).
The problem of allocation bias was demonstrated in a recent study.9 The investigators were attempting to compare a one‐stage protocol with a two‐stage protocol with respect to marginal bone loss after 5 years; unfortunately the patients in the two‐stage cohort were those who did not have a predetermined insertion torque at placement. As such, the two cohorts were not similar (one‐stage = high insertion torque, two‐stage = low insertion torque) for a major confounding variable and the internal validity of the study is in question.
Chronology bias refers to how long a clinical study ran and whether you, the reader, feel the time span was sufficient to justify the results and/or reveal expected or unexpected untoward responses. For example, company A has introduced a new implant surface that supposedly allows for faster osseointegration. How long would you expect the trial to run in order to accept the results as meaningful? What was their outcome assessment for success? Let’s assume that there was a matched control with an adequate N. Since this is a human study, sacrificing the subjects to get histology would not sit well with your local IRB, but you have confidence that the selected outcome assessment is reliable. They have compelling evidence that a range of 3–6 months proved verifiable in their control group. They run a 6‐month study with all subjects completing the full 6‐month protocol. Do you feel the time is sufficient? Some will say yes and some would feel more comfortable allowing the study to run for 1 year to be certain of the external validity. Some might question whether the new surface will function under occlusal load and the biologic burden of the oral cavity and feel a multiyear protocol is needed.
In a study examining bone loss around implants, what time sequence would you require, 1 year, 2–3 years, 4–5 years, 5+ years? If a study examined the periodontal response to varying emergence profiles on implant‐retained restorations, would you accept a shorter clinical trial than with the previous example? If the study was looking at monolithic zirconia that had surface custom staining and you were concerned about the outer glaze/stain wearing off, how many years would you expect the study to run? If you polled a group of experienced clinicians, you would get different answers to each, so who is correct? Unfortunately, EBD does not give you a definitive answer to this problem. It all comes back to your comfort with the premise and methodology, clinical experience, and need to alter your clinical regimen.
Referral filter bias is a type of selection bias and refers to where the patient is to be treated. For example, tertiary care cancer hospitals like M.D. Anderson or Memorial Slone Kettering have a different patient pool than you would expect to see in your private office. People who get on a plane and travel to the Mayo Clinic are not similar to the ones who are in your office because you practice close to where they work or live. Will the dental school patient be similar to yours? Will the patients in the office of a clinician who does external marketing be similar to yours, or vice versa?
Ideally, clinical projects should be triple blinded in that the person administering the therapy, drug, etc., the patient, and the person doing the outcome assessment are not familiar with what is being tested. The rationale is obvious. I am looking at my work and it all looks great, but you might not be so kind. It is one of the reasons that a case series performed and evaluated by the same group has a lower external and internal validity. In implant therapy studies, blinding often becomes quite difficult. If you are testing a zirconia abutment vs. a titanium abutment, there is a distinct visual difference that is hard to hide. Comparing a locator attachment with a ball attachment is another example that would be impossible to blind, as is one‐stage vs. two‐stage surgery. But, because the study is not blinded does not mean it is not well done and useful. Here we rely on the integrity of the researcher.
Conflict of interest (COI) is easily understood and it is now mandatory to disclose this in most journals. In a 2013 article in JADA,10 the authors examined RCTs in 10 journals, three of which did not have mandatory reporting of COI, and found that “RCTs in which authors have some type of COI are more likely to have results that support the intervention being assessed.” Here we have a major issue that needs to be addressed. Much of the implant clinical research we see is funded by commercial companies. In the US, it is apparent that the National Institute of Dental and Craniofacial Facial Research’s (NIDCR) policy is to fund basic science research and allow companies to fund clinical trials. While this seems counter‐intuitive, it is a fact of life. So we as clinicians, and the patients that we treat, are starting with a decided bias in the research being presented. Often SRs have eliminated RCTs for high risk of bias in other parameters but accept those with industry support. Burying our heads in the sand is not appropriate either. We must be realistic in how we evaluate all forms of bias. As with a lack of blinding, the internal validity suffers, but with both, one must assume the integrity of the researcher is intact. While there is a bad egg in every field, for the most part our colleagues are honest and sincere in their desire to do a study that will answer a needed question, undergo peer review, and stand the test of time. In today’s digital world, once it is written it is there for all to see for all time.
I wouldn’t have seen it if I didn’t believe it. Sherlock Holmes, in the novel A Scandal in Bohemia,11 said, “It is a capital mistake to theorize before one has data. Insensibly one begins to twist facts to suit theories, instead of theories to suit facts.” This is an unfortunate but sometimes unavoidable consequence for the researcher who has invested time, effort, and money in a project and perhaps is unable to clearly see what was happening. Sometimes it is innocuous, like framing the data in a positive manner. An example of this is the researcher who states they had a 70% success rate instead of a 30% failure rate, or justifying the lower success rate by saying that we save the patient time, money etc. But other times it takes on a more disconcerting approach, which could be a type of apophenia, which, according to the Merriam‐Webster Online Dictionary is “the tendency to perceive a connection or meaningful pattern between unrelated or random things (such as objects or ideas).” One of the best examples was given by Cotton12 in a 1988 editorial in the Journal of Dental Research, in which he described an experiment where a frog was trained to jump when told “jump”. After one leg was amputated the frog was still able to jump when told. After two legs were amputated the frog was still able to jump when told. The same occurred after three legs were amputated. After the fourth amputation the frog could not jump so the researchers concluded that quadruple amputation in frogs created deafness. His example explains it all.
The journal
Publication bias is often defined as a preference to publish studies that have a positive finding, and it is true that most studies have historically been positive or neutral. In fact, a recent Cochrane Review13 found that trials with positive findings were “published more often, and more quickly, than trials with negative findings.” There are numerous potential reasons for this finding. Many researchers are reluctant to admit that their premise was incorrect, but these studies have just as much clinical value as positive ones. The negative result always creates a conundrum when the study was funded by a company who now wants to squash the publication and the researcher does not want to risk the loss of future grants. We, as clinicians, should be accepting and thankful to our research colleagues who publish despite these concerns.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


