A child’s difficulty in verbalizing the precise location and nature of facial pain and jaw dysfunction often results in a nondefinitive history, increasing the importance of the dentist’s awareness of the early signs and symptoms of temporomandibular joint disorders (TMD). A focused examination of the masticatory musculature, the temporomandibular joints, and associated capsular and ligamentous structures can reveal if a patient’s symptoms are TMD in origin. An accurate differential diagnosis enables timely referral to appropriate health care providers and minimizes the use of diagnostic imaging.
Key Points
- •
Children seldom seek treatment for temporomandibular joint disorders (TMDs), but the dentist’s awareness of the early signs and symptoms of TMD can facilitate quicker resolution and prevent progression. A child’s difficulty in verbalizing the precise location and nature of facial pain and jaw dysfunction often results in a nondefinitive history, thus increasing the importance of the clinical evaluation.
- •
A focused examination of the masticatory musculature, the temporomandibular joints (TMJs), and associated capsular and ligamentous structures will reveal if a patient’s headaches, otologic symptoms, or facial pains are TMD in origin.
- •
An accurate differential diagnosis enables timely referral to appropriate health care providers, reduces unnecessary consultations, and minimizes the use of diagnostic imaging. Completing a TMD screening history and examination can provide the clinician with the information needed to understand the possible causes and explain the child’s condition.
- •
Guidelines on when to monitor TMDs or recommend treatment are not clearly established for children.
Children seldom seek treatment for temporomandibular joint disorders (TMDs), but the dentist’s awareness of the early signs and symptoms of TMD can facilitate quicker resolution and prevent progression. A child’s difficulty in verbalizing the precise location and nature of facial pain and jaw dysfunction often results in a nondefinitive history, thus increasing the importance of the clinical evaluation. A focused examination of the masticatory musculature, the temporomandibular joints (TMJs), and associated capsular and ligamentous structures will reveal if a patient’s headaches, otologic symptoms, or facial pains are TMD in origin ( Box 1 ). An accurate differential diagnosis enables timely referral to appropriate health care providers, reduces unnecessary consultations, and minimizes the use of diagnostic imaging. Completing a TMD screening history and examination can provide the clinician with the information needed to understand the possible causes and explain the child’s condition. Guidelines for when to monitor TMDs or recommend treatment are not clearly established for children.
-
What are the signs and symptoms of a TMD?
-
Tenderness in the area in front of your ear, especially when you chew, speak, or open your mouth wide to sing or yawn.
-
Headache and discomfort in the muscles on the side of your face or head.
-
Clicking, popping, or grinding sounds when you open or close your mouth.
-
Difficulty opening your mouth wide and catching, sticking, or locking of the jaw.
-
Things you should do if you have a jaw problem
-
Apply moist heat and massage the muscles on the side of your face.
-
Take small bites or cut up food and place it between your back teeth. Avoid large foods that force you to open your mouth wide such as hamburgers and big sandwiches.
-
Avoid biting off food with your front teeth.
-
Eat soft foods such as yogurt, eggs, cereal, oatmeal, soup, and noodles.
-
Avoid chewy foods including licorice, beef jerky, bagels, taffy, gummy bears, bubble gum, French bread, and tough meats.
-
Avoid hard, crunchy foods such as raw vegetables, chips, and nuts.
-
Things you should not do when you have a jaw problem
-
Rest your hand on your chin when sitting at a desk or lying on the floor.
-
Play any wind instrument or violin or sing in a choir if these activities make your jaw hurt more.
-
Bite your fingernails or cuticles as this will aggravate your jaw joint and muscles.
-
Clench or grind your teeth. Remember to keep your lips together and your teeth apart.
Epidemiology
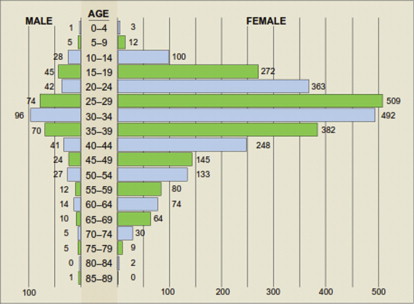
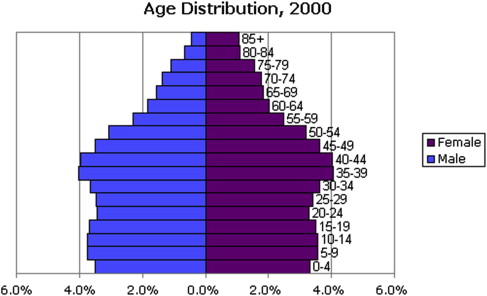
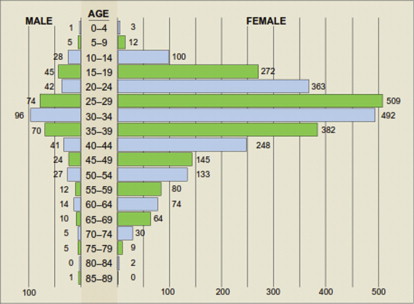
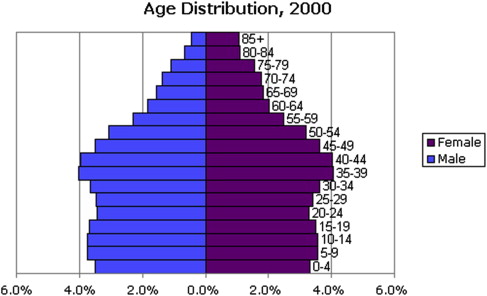
A study of 3428 consecutive patients of all ages enrolled in a health maintenance organization who sought treatment for TMDs ( Fig. 1 ) revealed that 85.4% were female. The skewed age and gender distribution, compared with the general population of the United States ( Fig. 2 ), suggest a hormonal influence. Treatment-seeking peaks occur during the reproductive years, with a mean age of 33.8 years, and this must be taken into account when considering the validity of the proposed causes for TMDs.
The prevalence of signs and symptoms of TMD in children in population-based reports varies considerably. The variation can be explained by the differences in the population investigated, by the examination methods and diagnostic criteria used, and by the interindividual and intraindividual variations of examiners.
Gender differences in the prevalence of TMD are less evident in early childhood and become more accentuated between 15 and 50 years of age, but the female preponderance occurs at all ages. TMD pain in children increases with age in both girls and boys. Females with TMD pain seek care more often than males at all ages but the probability for someone seeking care for TMD correlates more strongly with an increase in the frequency and intensity of facial pain, regardless of age or sex.
Wahlund and colleagues reported that the prevalence of TMD in children and adolescents (12–18 years old) was 7%. Nilsson found that the prevalence of TMD pain in 28,899 youths aged 12–19 years was 4.2% and was significantly higher in girls (6.0%) than boys (2.7%). Studies have correlated more frequent headaches and otologic symptoms with the occurrence of TMD. Köhler and colleagues concluded that the prevalence of severe TMD symptoms and signs in children and adolescents was generally low and did not change significantly during a 20-year follow-up period.
The Research Diagnostic Criteria for TMD (RDC/TMD), used in many studies to define, standardize, and replicate the characteristics of the study samples, are useful in research analysis of groups of patients. The use of standardized diagnostic criteria for defining clinical subtypes of TMD as well as awareness that psychosocial factors are an important factor should lead to better treatment outcomes. Dentists need to focus on the functional aspects of the dental occlusion, the biomechanical, or pathophysiologic aspects of the TMJ articulations, as well as the psychosocial aspects to best manage TMDs. As stated by Klasser and Green, the word biopsychosocial is an excellent descriptor for the condition that TMD pain patients are living with, in that they have a biological problem which may activate pain pathways, with or without a demonstrable pathologic condition, and there may have been psychological antecedents as well as behavioral consequences.
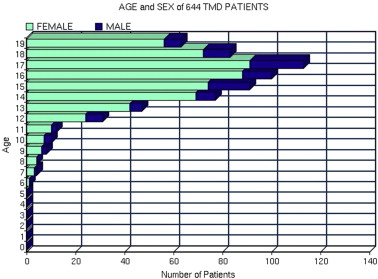
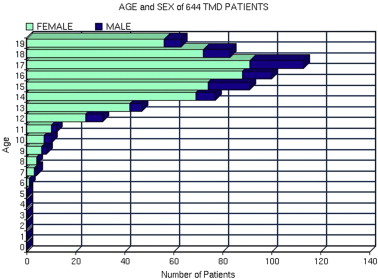
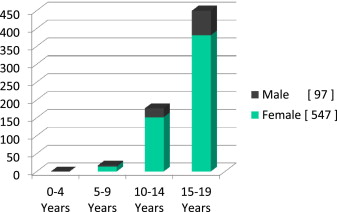
A combination of physical and psychosocial factors contribute to the decision to seek treatment. For children, their parents’ own experience with similar problems influences this decision. Of the 4262 consecutive patients with TMD seeking treatment in the private practice of the author during the past 15 years, 644 patients were less than 20 years of age ( Fig. 3 , Table 1 ), which represents 15.1% of all patients with TMD evaluated; whereas those less than 20 years of age represent 28.6% the general US population in 2000.
| Age in Years | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AGE | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 |
| Male | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 2 | 3 | 2 | 7 | 5 | 8 | 17 | 12 | 22 | 11 | 7 |
| Female | 0 | 0 | 0 | 0 | 0 | 1 | 3 | 4 | 6 | 7 | 10 | 24 | 42 | 69 | 74 | 88 | 91 | 72 | 56 |
| Total | 0 | 0 | 0 | 0 | 1 | 4 | 4 | 8 | 10 | 12 | 31 | 47 | 77 | 91 | 100 | 113 | 83 | 63 | |
| % Male | / | / | / | / | / | / | 25 | 0 | 25 | 30 | 17 | 23 | 11 | 10 | 18 | 12 | 19 | 13 | 11 |
| % Female | / | / | / | / | / | 100 | 75 | 100 | 75 | 70 | 83 | 77 | 89 | 90 | 82 | 88 | 81 | 87 | 89 |
| Age as % of Total | / | / | / | / | / | / | .60 | .60 | 1.2 | 1.5 | 1.9 | 4.8 | 7.4 | 12.0 | 14.1 | 15.5 | 17.7 | 12.9 | 9.8 |
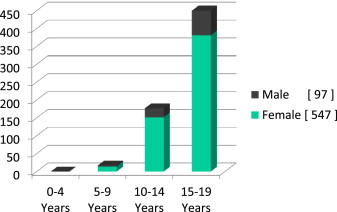
Patients 15–19 years old account for 65.8 of the 644 patients ( Fig. 4 ). No patients younger than 6 years of age were evaluated in last 15 years of private practice. Girls account for 89.9% of those over 15–19 years of age seeking treatment, and 75.5% of patients 6–14 years of age were female.
There are more male births than female births and until the age of 35 years, males represent a larger percentage of the US population. The higher percentage of patients with TMD between the ages of 15 and 19 years and the increased ratio of females both suggest that different factors account for treatment-seeking behavior for TMD in young children than in adolescents. The impact of TMD pain on adolescents differs by age and gender. A questionnaire was mailed to 350 clinic patients aged 12 to 19 years and 350 age-matched and sex-matched controls. There were no age or sex differences in pain intensity, however for those adolescents aged 16–19 years, TMD pain had significantly greater impact on behavioral and psychosocial factors on girls than on boys. Among those aged 16 to 19 years, 32.4% of girls compared with 9.7% of boys reported school absences and analgesic consumption because of their TMD pain. This report is consistent with the finding of a significant increase in treatment seeking by girls aged 13 to 19 years (see Table 1 ).
Late adolescent patients with TMD had higher pain intensity in the orofacial region and reported more impact on activities of daily living, including difficulty in prolonged jaw opening, eating soft/hard foods, and sleeping, than younger patients. Of 167 patients age 16 to 18 years, girls reported significantly more problems than the boys due to headache and neck pain.
Epidemiology
A study of 3428 consecutive patients of all ages enrolled in a health maintenance organization who sought treatment for TMDs ( Fig. 1 ) revealed that 85.4% were female. The skewed age and gender distribution, compared with the general population of the United States ( Fig. 2 ), suggest a hormonal influence. Treatment-seeking peaks occur during the reproductive years, with a mean age of 33.8 years, and this must be taken into account when considering the validity of the proposed causes for TMDs.
The prevalence of signs and symptoms of TMD in children in population-based reports varies considerably. The variation can be explained by the differences in the population investigated, by the examination methods and diagnostic criteria used, and by the interindividual and intraindividual variations of examiners.
Gender differences in the prevalence of TMD are less evident in early childhood and become more accentuated between 15 and 50 years of age, but the female preponderance occurs at all ages. TMD pain in children increases with age in both girls and boys. Females with TMD pain seek care more often than males at all ages but the probability for someone seeking care for TMD correlates more strongly with an increase in the frequency and intensity of facial pain, regardless of age or sex.
Wahlund and colleagues reported that the prevalence of TMD in children and adolescents (12–18 years old) was 7%. Nilsson found that the prevalence of TMD pain in 28,899 youths aged 12–19 years was 4.2% and was significantly higher in girls (6.0%) than boys (2.7%). Studies have correlated more frequent headaches and otologic symptoms with the occurrence of TMD. Köhler and colleagues concluded that the prevalence of severe TMD symptoms and signs in children and adolescents was generally low and did not change significantly during a 20-year follow-up period.
The Research Diagnostic Criteria for TMD (RDC/TMD), used in many studies to define, standardize, and replicate the characteristics of the study samples, are useful in research analysis of groups of patients. The use of standardized diagnostic criteria for defining clinical subtypes of TMD as well as awareness that psychosocial factors are an important factor should lead to better treatment outcomes. Dentists need to focus on the functional aspects of the dental occlusion, the biomechanical, or pathophysiologic aspects of the TMJ articulations, as well as the psychosocial aspects to best manage TMDs. As stated by Klasser and Green, the word biopsychosocial is an excellent descriptor for the condition that TMD pain patients are living with, in that they have a biological problem which may activate pain pathways, with or without a demonstrable pathologic condition, and there may have been psychological antecedents as well as behavioral consequences.
A combination of physical and psychosocial factors contribute to the decision to seek treatment. For children, their parents’ own experience with similar problems influences this decision. Of the 4262 consecutive patients with TMD seeking treatment in the private practice of the author during the past 15 years, 644 patients were less than 20 years of age ( Fig. 3 , Table 1 ), which represents 15.1% of all patients with TMD evaluated; whereas those less than 20 years of age represent 28.6% the general US population in 2000.
| Age in Years | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AGE | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 |
| Male | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 2 | 3 | 2 | 7 | 5 | 8 | 17 | 12 | 22 | 11 | 7 |
| Female | 0 | 0 | 0 | 0 | 0 | 1 | 3 | 4 | 6 | 7 | 10 | 24 | 42 | 69 | 74 | 88 | 91 | 72 | 56 |
| Total | 0 | 0 | 0 | 0 | 1 | 4 | 4 | 8 | 10 | 12 | 31 | 47 | 77 | 91 | 100 | 113 | 83 | 63 | |
| % Male | / | / | / | / | / | / | 25 | 0 | 25 | 30 | 17 | 23 | 11 | 10 | 18 | 12 | 19 | 13 | 11 |
| % Female | / | / | / | / | / | 100 | 75 | 100 | 75 | 70 | 83 | 77 | 89 | 90 | 82 | 88 | 81 | 87 | 89 |
| Age as % of Total | / | / | / | / | / | / | .60 | .60 | 1.2 | 1.5 | 1.9 | 4.8 | 7.4 | 12.0 | 14.1 | 15.5 | 17.7 | 12.9 | 9.8 |
Patients 15–19 years old account for 65.8 of the 644 patients ( Fig. 4 ). No patients younger than 6 years of age were evaluated in last 15 years of private practice. Girls account for 89.9% of those over 15–19 years of age seeking treatment, and 75.5% of patients 6–14 years of age were female.
There are more male births than female births and until the age of 35 years, males represent a larger percentage of the US population. The higher percentage of patients with TMD between the ages of 15 and 19 years and the increased ratio of females both suggest that different factors account for treatment-seeking behavior for TMD in young children than in adolescents. The impact of TMD pain on adolescents differs by age and gender. A questionnaire was mailed to 350 clinic patients aged 12 to 19 years and 350 age-matched and sex-matched controls. There were no age or sex differences in pain intensity, however for those adolescents aged 16–19 years, TMD pain had significantly greater impact on behavioral and psychosocial factors on girls than on boys. Among those aged 16 to 19 years, 32.4% of girls compared with 9.7% of boys reported school absences and analgesic consumption because of their TMD pain. This report is consistent with the finding of a significant increase in treatment seeking by girls aged 13 to 19 years (see Table 1 ).
Late adolescent patients with TMD had higher pain intensity in the orofacial region and reported more impact on activities of daily living, including difficulty in prolonged jaw opening, eating soft/hard foods, and sleeping, than younger patients. Of 167 patients age 16 to 18 years, girls reported significantly more problems than the boys due to headache and neck pain.
Etiology
Greene pointed out that the inability to identify precise causes or the lack of a perfect theoretical model does not prevent rendering sensible and often successful treatment of most patients with TMD. Historically, malocclusion has been considered a primary cause of TMD, but the occurrence of malocclusion, occlusal interferences, and missing teeth is nearly equal for males and females. However, most published studies evaluate static, morphologic variables and overlook the importance of the dynamic relationship between the joints and teeth during function. Restriction in the anterior envelope of function and dysfunctional occlusal contacts that alter the final path of closure during mastication both take on greater importance with the eruption of the permanent teeth. The increase in treatment seeking for TMD at 13 years of age ( Table 1 ) coincides with full eruption of the second molars and the acceleration of growth of the mandible. Hormonal fluctuations in females have been implicated in altered pain perception and increased treatment seeking. Micro and macro trauma are known causes of TMD as are systemic factors and disease.
It is useful to categorize ( Table 2 ) the causes as predisposing, precipitating and perpetuating.
-
Predisposing (or risk) factors for TMDs can be
- •
Systemic (affecting the entire body or a particular body system)
- •
Psychosocial (interaction of psychological and social variables)
- •
Physiologic (cellular and metabolic processes, neuromuscular)
- •
Structural (dental occlusion, musculoskeletal, articular, developmental anomalies).
- •
-
Precipitating (or initiating) factors often involve trauma or overuse. Repetitive activities with the jaw in a sustained or abnormal posture or under abnormal load, such as when playing a wind instrument or violin, or sleep posture can trigger a painful TMD episode.
-
Perpetuating (or sustaining) factors often include parafunction, overuse, systemic disease, occlusal factors, or psychological distress.
| Cause | Examples | Predisposing | Precipitating | Perpetuating |
|---|---|---|---|---|
| Trauma, micro | Repetitive strain (eg, wind musician, violin, singing, fingernail biting, scuba diving, snorkeling, swimming) | ✓ | ✓ | ✓ |
| Trauma, macro | External blow (eg, falls, motor vehicle accident, sports injury, physical abuse, intubation, tooth extraction, prolonged mouth opening, cervical traction, neck brace) | ✓ | ||
| Ligament laxity, systemic (hypermobility) | Hormonal variation in ligament laxity, Breighton syndrome | ✓ | ✓ | |
| Stress-induced parafunction | Tooth clenching, grinding, tapping | ✓ | ✓ | ✓ |
| Sleep-induced bruxism | Involuntary nocturnal tooth grinding | ✓ | ✓ | ✓ |
| Restrictive anterior bite relationship | Deep overbite, retruded upper incisors, mandibular skeletal hyperplasia | ✓ | ✓ | |
| Loss of posterior tooth support | Missing or extracted molars without prosthetic replacement | ✓ | ✓ | ✓ |
| Excessive horizontal overjet | >6-mm distance between upper and lower incisors | ✓ | ✓ | |
| Abnormal condylar form, developmental or congenital | Bifid condyle, condylar hyperplasia, condylar hypoplasias, tumors | ✓ | ✓ | |
| Systemic inflammatory and metabolic polyarthritis | Rheumatoid arthritis, juvenile idiopathic arthritis, scleroderma, psoriatic arthritis, ankylosing spondylitis, Reiter syndrome, systemic lupus erythematosus, gout | ✓ | ✓ | ✓ |
| Infectious arthritis | Lyme disease, sexually transmitted disease, gonococcal arthritis. Chlamydia trachomatis | ✓ | ||
| Central nervous system–mediated maxillofacial movement disorders and palsy | Hypokinesia (eg, Parkinson disease, muscular dystrophy), hyperkinesias (eg, tics, tardive dyskinesia, dystonia, chorea, myoclonus), palsy (cerebral palsy, facial palsy) | ✓ | ✓ | ✓ |
Parafunction
Parafunctional activities are nonfunctional oromandibular activities that include jaw clenching, tooth grinding, tooth tapping, cheek biting, lip biting, and object biting occurring alone or in combinations. Bruxism is a movement disorder characterized by grinding and clenching of teeth ( Box 2 ). Awake bruxism is found more in females than in males, whereas sleep bruxism (SB) shows no such sex prevalence. Data gathered from awake subjects cannot predict SB behavior and self-reports of bruxism during sleep are not reliable. Cause of bruxism has been reported in 3 categories: psychosocial factors, peripheral occlusal factors, and neurophysiologic factors. Most SB episodes are associated with the arousal response, a sudden change in the depth of the sleep during which the individual either arrives in the lighter sleep stage or actually wakes up ( Box 3 ). The micro-arousals are short lasting, with periods of bruxism lasting 3 to 15 s with cortical activation associated with an increase in the activity of the sympathetic nervous system. This centrally mediated bruxism is often accompanied by increased muscle activity and gross body movements including involuntary leg movements, increased heart rate, and respiratory changes.
-
What are the signs and symptoms of bruxism?
-
Bruxism activity while you are asleep, especially tooth grinding, occurs more often if you experience restless sleep. Tooth grinding during your sleep is triggered by impulses from the brain and it is not really a habit.
-
Daytime bruxism is more likely to manifest as tooth clenching or bracing your jaw with your mouth part-way open or off to the side.
-
Bruxism often leads to tooth sensitivity to hot and cold and to bite pressure.
-
Cracks in your teeth or flat spots (facets) are another sign of bruxism.
-
Awakening with a headache or unexplained earache accompanied by a stiff jaw strongly suggests nighttime bruxism.
-
What should you do if your clench or grind your teeth?
-
Sleep-related bruxism can be hard to control. You can focus on preventing damage to your teeth and reduce the associated muscle discomfort by using a protective plastic guard over your teeth.
-
To control daytime clenching or bracing, you have to become aware of the activity. Have those around you remind you if they see your jaw set tight. When you catch yourself clenching remember to keep your lips together and teeth apart.
-
Consider attending relaxation therapy, biofeedback, or yoga classes to help reduce stress. Increase your physical exercise to improve your sleep.
-
Avoid excessive caffeine intake or drinking caffeinated beverages within 3 hours of bedtime. Be aware that chocolate and many soda drinks contain caffeine.
-
Create a quiet and dark sleep environment and establish a regular bedtime. Do not shortchange yourself on sleep.
-
Restless or disturbed sleep usually increases bruxism (tooth clenching or grinding). A good night’s sleep can be achieved by following these simple guidelines:
-
Go to bed at about the same time every night and get up from bed at about the same time each day; by doing so the body clock remains synchronized with the outside environment. Everyone has a circadian rhythm, an internal 24-hour clock that plays a critical role in when we fall asleep and when we wake up. By sticking to a regular waking and sleeping time, the body becomes adapted to this schedule.
-
Exercise regularly. Studies have shown that regular exercise encourages restful sleep. Exercise should be done early in the evening or in the morning. Do not exercise just before bedtime as this stimulates the body and makes it more difficult to fall sleep. Make the bedroom as restful as possible by keeping the temperature cool and reducing noise and outside light to a minimum.
-
Caffeine is a central nervous system stimulant, temporarily warding off drowsiness and restoring alertness. Avoid chocolate and beverages containing caffeine, such as coffee, tea, soft drinks, and energy drinks for at least 4 hours before bedtime. Do not use alcohol to help you sleep as alcohol consumption leads to fragmented sleep and it often worsens snoring and sleep apnea.
-
Do not undertake stimulating activities just before bed. Exciting games or movies or engaging in important family discussions stimulate the mind and may make it more difficult to fall asleep.
-
A warm bath or shower before bedtime increases body temperature and the subsequent decrease in temperature promotes sleep.
-
If unable to fall asleep within 20 to 30 minutes, get up and engage in some activity in another room. Do not stay in the bedroom trying to force yourself to fall sleep. Only return to the bedroom when you are sleepy.
Bruxism should not be considered a pathologic condition and is not an indicator of psychopathology and most individuals with bruxism do not develop pain. Unless bruxism is causing headaches, facial pain, TMJ instability, severe tooth attrition, fracture of dental restorations, or tooth mobility, it does not require treatment. When necessary, the focus of therapy is to prevent damage to the teeth and supporting structures by managing the forces, and this can be accomplished with an intraoral appliance (IOA). Injection of botulinum toxin, which acts as a paralytic, into the temporalis and masseter muscles has been used in patients with cerebral palsy to reduce intraoral soft-tissue injury and tooth damage as well as for patients with bruxism-related headaches. However, the effect of this neurotoxin is localized at the motor endplate and although the amount of force generated is reduced, it does not alter the bruxism activity and is effective for only 3 to 4 months. Bruxism can also be reduced with the use of sedative and anxiolytic drugs, but patient compliance is low because of side effects. Maintenance of the drugs’ therapeutic efficacy, their long-term tolerability, and the risk of addiction need further investigation. Pharmacologic management of bruxism in children is not an appropriate treatment, except for short-term use for situational anxiety. No effective treatment to permanently eliminate sleep bruxism has been identified.
Malocclusion
The clear lack of an association between occlusal anomalies and TMD may be due to the frequent deviation from the norm and because inadequate and invalid study designs, including the failure to take into account the development stage of the occlusion, have led to false-negative results. A sample of 4724 children (2353 girls and 2371 boys) aged 5 to 17 years were grouped by the stage of dental development (deciduous, early mixed, late mixed, and permanent dentition). The registrations included functional occlusion (anterior and lateral sliding, interferences), dental wear, mandibular mobility (maximal opening, deflection), and TMJ and muscular pain provoked by palpation. Mild clinical signs were recorded in 22.8% of the children; only 2.8% had moderate to severe findings and multiple clinical signs. The prevalence of TMD increased during the developmental stages, with girls more affected than boys. Significant associations were found between TMD and posterior crossbite, anterior openbite, Angle Class III malocclusion, and extreme maxillary overjet greater than 6 mm.
The occurrence of occlusal anomalies is similar for both sexes, so occlusal factors do not explain the predominance of TMD pain among girls. Although the role of occlusion as a predisposing factor cannot be confirmed by conclusive scientific evidence, some occlusal features may place greater adaptive demands on the masticatory system. It is proposed that most individuals compensate without problems, but adaptation in others may lead to greater risk of dysfunction. Some occlusal anomalies may be a result rather than a cause of TMD.
The relationship between dental occlusion and TMDs has been one of the most controversial topics in the dental community. The Study of Health in Pomerania (Germany), a cross-sectional survey of 4289 adults (aged 20–81 years), revealed associations between 15 occlusion-related variables and TMD signs or symptoms. However, statistical associations do not prove causality. Only bruxism, loss of posterior support, and unilateral posterior crossbite show some consistency across studies. On the other hand, several reported occlusal features seem to be a consequence of TMDs, not their cause. Biological plausibility for an occlusal etiology is difficult to establish, because TMDs are much more common among women than men. Symptom improvement after insertion of an IOA or after occlusal adjustment does not prove an occlusal cause, because the amelioration may be the result of appliance-induced changes in vertical dimension, or altered proprioception with decreased muscle activity. In addition, TMD symptoms often abate even in the absence of therapy.
Systemic Factors
Most connective tissue diseases that affect other articulations also occur in the TMJ, including rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, mixed connective tissue disease, juvenile idiopathic arthritis, and psoriatic arthropathy. Typical imaging findings are joint-space narrowing, and condylar erosion, flattening, and sclerosis. Connective tissue diseases are a group of closely related conditions, with many overlapping clinical features that involve the skin, joints, muscles, or blood. Serologic examination can reveal connective tissue diseases that are associated with a variety of antinuclear antigens (ANA) and other related antibodies. However, the ANA test lacks specificity and the presence of the antibody is not necessarily diagnostic for a specific disease because these antibodies may be found in patients with other autoimmune diseases such as hepatitis C, may be induced by medication, and may even be present in otherwise healthy individuals.
Generalized joint hypermobility (GJH) was evaluated as a risk factor for TMD in 895 subjects (20–60 years of age). Hypermobile subjects had a higher risk for reproducible reciprocal TMJ clicking associated with disk displacement with reduction (odds ratio [OR] = 1.68) compared with those without hypermobile joints. No association was observed between hypermobility and myalgia/arthralgia; thus, GJH was found to be associated with nonpainful subtypes of TMD.
Children (n = 1833) aged 4 to 18 years were evaluated for GJH; the prevalence rate of symptomatic hypermobility was 13.8% for girls and 8.2% for boys. Besides gender (OR = 2.07), risk factors for symptomatic hypermobility were race (OR = 2.61 for nonwhites) and was associated with masticatory muscle pain (OR = 1.95).
Trauma
In a survey of 2374 students, 715 had positive symptoms for TMD. They were classified into 7 groups: group 1, those with clicking only; group 2, only pain in the temporomandibular joint; group 3, only difficulty with mouth opening; group 4, clicking and pain; group 5, clicking and difficulty with mouth opening; group 6, difficulty with mouth opening and pain; and group 7, all 3 symptoms. TMD symptoms were significantly associated with a history of jaw injury with; the ORs by group were: group 2, 2.25; group 3, 2.47; group 6, 3.38; and group 7, 2.01. Experience of third molar removal was significantly associated with the onset of TMD (OR = 1.81) for group 1. Experiences of jaw injury and third molar removal might be cumulative and precipitating events in TMD. No association was found between orthodontic experience and TMD in any group.
The epidemiology of facial injuries in children and adolescents (from birth to 18 years) was evaluated using the National Trauma Data Bank (2001–2005) to examine facial fracture pattern, mechanism, and concomitant injury by age. A total of 12,739 (4.6%) facial fractures were identified among 277,008 pediatric trauma patient admissions. The proportion of patients with facial fractures increased substantially with age. The most common facial fractures were mandible (32.7%), nasal (30.2%), and maxillary/zygoma (28.6%). Motor vehicle collision (55.1%), assault (14.5%), and falls (8.6%) were the most common mechanisms for facial fracture in all pediatric age groups. The second most common cause of bony facial injury varied with age. Fall was the second most common mechanism (23.4%) among infants and toddlers (0–4 years). Bicycle-related collisions and pedestrians struck by motor vehicles were the second most common mechanisms for school-aged children (5–14 years). For older teenagers (15–18 years), the second most common mechanism was a fight or an assault (21.7%). The male sex predominated through all age groups and for all types of injuries.
Related to increasing age, many of these patients reported more than one trauma, and as pointed out by Akhter and colleagues, there is a cumulative effect of trauma as a causative factor for TMD. Of the in 644 children ( Fig. 3 ) seeking TMD treatment, 13.8% presented with chin scars and 37.7% reported a history of facial trauma that may have contributed to their TMD. Although not statistically significant, because of the small number of males in the sample, blows to the face are more common in males.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


