| Key Points |
 The impact of chronic pain on sleep can be described as a vicious cycle, with mutual deleterious influences causing an increase in pain and disrupted sleep. The impact of chronic pain on sleep can be described as a vicious cycle, with mutual deleterious influences causing an increase in pain and disrupted sleep. |
 The role of the clinician is to identify orofacial pain patients complaining of poor sleep and refer them to a sleep laboratory for further evaluation of a suspected sleep disorder. The role of the clinician is to identify orofacial pain patients complaining of poor sleep and refer them to a sleep laboratory for further evaluation of a suspected sleep disorder. |
 Sleep hygiene advice and cognitive behavioral treatments or medications may help restore sleep quality. Sleep hygiene advice and cognitive behavioral treatments or medications may help restore sleep quality. |
 Pain and sleep management need to be customized to each patient based on his or her psychosocial and medical histories. Pain and sleep management need to be customized to each patient based on his or her psychosocial and medical histories. |
Approximately one-third of patients with temporomandibular disorders (TMDs) or other conditions manifesting orofacial pain report poor sleep quality or an unrefreshed feeling on awakening. The impact of chronic pain on sleep can be described as a vicious cycle, with mutual deleterious influences between pain and disrupted sleep. A few quantitative sleep variables (eg, total sleep time, slow-wave sleep, sleep stage duration, presence of breathing events, and periodic limb movements [PLMs]) characterize the pain-related disruption of sleep. The role of the clinician is to identify patients complaining of poor sleep and, after excluding the role of concomitant musculoskeletal or neuropathic pain or comorbid conditions (eg, complaints related to fatigue, depression, anxiety, and poor sleep), refer the patients to a sleep laboratory when a sleep disorder is suspected, such as insomnia (ICD-10 G47.0; ICD-9 780.52), sleep apnea (ICD-10 G47.3; ICD-9 780.57), or PLM (ICD-10 G47.61; ICD-9 327.51), according to The International Classification of Diseases, Tenth (ICD-10) and Ninth (ICD-9) Editions. The aim of this chapter is to provide a critical overview of the current understanding of pain and sleep interactions and to discuss evidence-based and empirical knowledge to help clinicians recognize, diagnose, and manage poor sleep where indicated in patients with chronic orofacial pain conditions.
Sleep is a natural physiologic activity essential for good quality of life and indeed species survival. Animals totally deprived from sleep become sick from infection or organic dysfunctions within a few weeks. Sleep is vital for recovery from fatigue and for memory consolidation, tissue repair, and brain function at cellular and brain network levels. Its duration is variable from individual to individual. Most adult humans sleep between 6 and 9 hours a night; below or above this range, humans tend to report more pain.1,2 Teenagers tend to have an irregular circadian sleep (ICD-10 G47.23; ICD-9 327.33) schedule that may predispose them to some pains. Without enough sleep, humans tend to be less functional and tend to report cognitive problems, mood alterations, immune dysfunction, and somatic pain–related complaints within 3 to 4 days.3,4
Sleep and wakefulness are under the regulation of a circadian process lasting approximately 24 hours. Typically, sleep and wake cycles are regulated by an oscillatory behavior at the level of the hypothalamus suprachiasmatic nucleus in the higher regions of the central nervous system (CNS). Human sleep and wake rhythms are tuned by the sun and moon (ie, 24-hour light/ dark cycle) but also by external cues, such as sounds.5,6 The importance of understanding circadian rhythm is that pain patients may have circadian phase mismatch, which could explain their symptoms. A person who goes to sleep later every night may have a hard time waking up early; this sleep cycle is called circadian phase delayed (ICD-10 G47.21; ICD-9 327.31). A person who goes to sleep earlier every day may wake up earlier; this sleep cycle is called circadian phase advanced (ICD-10 G47.22; ICD-9 327.32).2,7
Sleep is divided into rapid eye movement (REM) and non-REM (NREM). During a typical night, there are four to five NREM to REM cycles, termed the ultradian rhythm cycle, in contrast to the 24-hour circadian cycle that is under the moon and sun time schedule. NREM sleep is further divided into light sleep (stages N1 and N2) and deep sleep (N3, formerly called stages 3 and 4, which is dominated by slow-wave brain activity) (Fig 11-1). REM sleep is a stage that is often called paradoxical sleep, because the CNS and the autonomic nervous system are highly active but all skeletal muscles are in a hypotonic state, as if the body were paralyzed.5
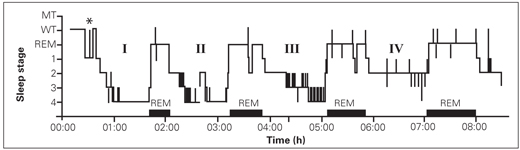
Fig 11-1 Histogram of four ultradian oscillations (I to IV) between NREM (stages 1 to 4 or N1, N2, and N3 for stages 3 and 4) and REM (horizontal full bars) sleep in a normal adult sleep cycle. MT—movement time; WT—wake time; *—12 brief awakening episodes. (Reprinted from Lavigne and Manzini8 with permission.)
In healthy adults, the majority of body movements (eg, of limbs and jaws) tend to occur in light sleep, more specifically in relation to sleep stage shifts, such as from deeper to lighter sleep. The occurrence of movements is periodic during sleep and follows a cyclic pattern. Every 20 to 40 seconds, sleep oscillates from quiet periods to more active ones. The active physiologic periods, lasting 3 to 10 seconds, are called arousals; in normal patients, these tend to reappear 7 to 15 times per hour of sleep. Such active periods are “windows” in a sense that the sleeping individual can readjust his or her body position, reset body temperature, and, if any harmful event is perceived, become fully awake (ie, a fight-or-flight reaction could be triggered).
About 80% of sleep bruxism (ICD-10 G47.63; ICD-9 327.53) events (ie, repetitive jaw muscle contractions with or without tooth grinding) are observed during such recurrent arousal periods. A method has been developed to monitor such a cyclic alternating pattern (CAP) during sleep. A CAP comprises an active phase A that is subdivided into three periods: A1 (dominated by slow-wave brain activity, which preserves sleep), A2 (the transition period between A1 and A3), and A3 (the arousal dominant period).9 Phase B of a CAP is the quiet period of sleep. Sleep bruxism seems to occur preferentially in CAP A2 and especially in A3, thereby providing a physiologic window to facilitate the onset of rhythmic movements during sleep.10 It is important to understand that sleep bruxism tends to occur in transition from deeper to lighter sleep and when arousal pressure is greater. Patients with chronic pain and more specifically fibromyalgia (ie, chronic widespread musculoskeletal pain [CWP]) also tend to present with 50% more phase A periods than healthy adults.9 In contrast, during normal healthy adult sleep, nociception is partially attenuated to preserve sleep continuity, with higher thresholds and lower response rates to noxious stimuli in light sleep (stages N1 and N2), even higher thresholds in deep sleep (stage N3), and variable thresholds in REM sleep.9,11,12
Normal adult sleep onset usually appears within 20 to 30 minutes of when an individual goes to bed. Insomnia may be suspected when sleep onset is longer than 30 minutes occurring three to five times a week or if spontaneous awakening is present during the night without the ability to resume sleeping.7,13 About 10% of the general population suffers from chronic insomnia, but the prevalence is reported to be around 30% in chronic pain patients.11
Pain conditions can alter normal sleep patterns. The effect of acute pain on sleep seems to follow a linear model: pain precedes poor sleep (ie, cause and effect sequence). Hopefully, sleep returns to normal when the acute pain is resolved. However, in the presence of chronic pain (pain lasting for at least 3 months or longer), a circular model seems to predominate; however, a linear model may still be present in some patients. In the circular model, a night of poor sleep is followed by a day with complaints of more intense and variable pain, which is then followed by a night of nonrestorative sleep with morning-related complaints of unrefreshing sleep.
Approximately one-to two-thirds of chronic musculoskeletal pain patients report poor sleep quality.11Nonrestorative sleep is defined as an unrefreshed feeling on awakening, and it is present in about 10% of the general population, with a higher risk as individuals age. Unrefreshing sleep is a frequent complaint found in shift workers who work during the night, in patients who sleep over 9 hours per night, and in those with insomnia-related symptoms or with fatigue and mood alterations.3,11 The presence of restorative sleep (ie, sleep that leaves the individual feeling refreshed, rested, and re-energized) appears to predict the resolution of CWP in some patients.14 It remains to be demonstrated whether some subjects are genetically protected against the deleterious impact of chronic pain on sleep.11,15
Various factors may contribute to the interaction of pain and poor sleep; lifestyle, beliefs, difficulties in coping with anxiety, poor physical fitness, and chronic fatigue may be risk factors for insomnia, a condition found in 36% of patients with TMDs.15,16 In addition, sleep comorbidities include PLMs and sleep-disordered breathing (apnea/hypopnea or upper airway resistance), which can exacerbate the pain–poor sleep interaction. PLMs and sleep apnea/hypopnea have been reported in both TMD and CWP/fibromyalgia patients with greater frequency than in the controls.16,17 The diagnostic threshold for sleep apnea (ICD-10 G47.3; ICD-9 780.58) or sleep-related movement disorders (ICD-10 G47.6; ICD-9 780.58) is most frequently isolated in diagnostic decision-making. A PLM index over 10 events per hour of sleep (from an electromyogram of the tibialis leg muscle) is the new lower threshold for polysomnographic diagnosis of PLMs.18 Sleep apnea/hypopnea is diagnosed if there are five events per hour of sleep in adults or per 1 to 2 hours of sleep in children; no clear consensus has been reached yet for adolescents.13
Compared with a general practice of adult patients with CWP, community cases have a higher risk (odds ratio > 3) of reporting comorbid conditions, such as fatigue, headache, gastrointestinal problems, and sleep disturbances.19 Orofacial pain patients also report more problems in coping with fatigue, psychologic distress, headaches, and abdominal pains.16,20–22 Patients in a family medical practice who had hypertension, pain syndromes (eg, back pain, arthritis), and depression also had more sleep disturbance–related complaints.23 Several sleep-related problems, such as sleepiness, dozing off during daily activity, frequent awakenings during the night, restless leg syndrome–related complaints (ie, the awake symptoms in approximately 80% of PLM cases), and signs of sleep-related breathing disturbances (eg, loud snoring and cessation of breathing suggestive of apnea), were higher in pain patients.7,11,15 Clinicians working with pain patients should recognize and understand the influences of these comorbidities on their differential diagnoses (Box 11-1) and treatment planning for orofacial pain patients.7,11 The Epworth Sleepiness Scale is a simple-to-use screening questionnaire to guide the clinician on the extent of daytime sleepiness; scores over 10 to 12 are suggestive of a sleep-related disorder and warrant medical evaluation.24 The STOP-Bang sleep questionnaire may help the clinician discern between simple snoring and sleep apnea.25,26
| Box 11-1 Elements for assessment of orofacial pain and sleep complaints |
| • Identify the nature of the pain and sleep complaints. Ask the patient to keep a 24-hour diary, if possible, to monitor periods of pain exacerbation, time and duration of daily naps, sleep time, and wake time. • Review medication type and dose, time of intake, and use of other treatments for pain or sleep complaints, such as physical therapy or cognitive behavioral therapy. • Identify mood alterations (eg, depression, anxiety). • Identify stressful life events or traumatic events. • Identify poor sleep behavior or hygiene (eg, irregular sleep schedule; disrupted sleep environment; frequent use of caffeine, alcohol, or medications that alter sleep). • Identify the extent of sleepiness (eg, falling asleep easily during daily activities or while watching TV, driving-related sleepiness) using the Epworth Sleepiness Scale or STOP-Bang questionnaire. • Assess the risk of insomnia (ie, more than 30 minutes’ delay in falling asleep or difficulty resuming sleep after awakening during the night). • Assess the presence of any PLMs (eg, leg kicks that will increase sleep fragmentation above a cumulative threshold of events per hour of sleep). • Assess if any snoring, alone or with sleep-disordered breathing (upper airway resistance or apnea/ hypopnea, ie, cessation of breathing with reduced oxygenation with concomitant sleepiness), may contribute to increased sleep fragmentation and poor sleep quality complaints. • Identify clinical risk factors associated with sleep-related breathing disorders, such as retrognathia, a deep and narrow palate and narrow arch, and large tonsils and adenoids. |
< div class='tao-gold-member'>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


