Introduction
Our objective was to analyze the surface and bulk properties alterations of clinically aged composites used for fixed retention.
Methods
Twenty-six lingual retainers bonded for different time periods (2.2-17.4 years) were retrieved from postorthodontic patients. Fifteen lingual retainers had been cemented by a chemically cured adhesive (Maximum Cure, Reliance Orthodontic Products, Itasca, Ill), and 11 were treated with a photo-cured adhesive (Flow-Tain, Reliance Orthodontic Products). The first group was in service for 2.8 to 17.4 years and the second for 2.2 to 5.4 years. Five specimens from each material were prepared and used as the control (or reference) group. The debonded surfaces from enamel were studied by attenuated total reflectance Fourier transform infrared spectroscopy (n = 3 per material per group), low-vacuum scanning electron microscopy, and energy dispersive x-ray microanalysis (n = 3 per material per group). All specimens were used for the assessment of Vickers hardness, indentation modulus, and elastic index with the instrumented indentation testing method. The values of Vickers hardness, indentation modulus, and elastic index were compared between the retrieved and the reference groups with 1-way analysis of variance and the Student-Newman-Keuls multiple comparison test (α = 0.05).
Results
The attenuated total reflectance Fourier transform infrared spectroscopy analysis showed that both retrieved composites demonstrated reduced unsaturation in comparison with the corresponding reference specimens. Some bonded surfaces showed development of organic integuments. All retrieved specimens showed reduced silicon content. Barium was identified only in the photo-cured group. No significant differences were found between the reference and retrieved groups in Vickers hardness, indentation modulus, and elastic index.
Conclusions
Despite the changes in composition, the mechanical properties of the materials tested remained unaffected by intraoral aging.
Highlights
- •
Multistranded wires were bonded to lingual enamel for orthodontic retention.
- •
Effects of intraoral aging on light- and chemically cured composites were compared.
- •
Some differences in structure and composition occurred after prolonged exposure.
- •
Most mechanical properties were unaffected.
Changes in dental arch dimensions due to age are a common occurrence affecting both untreated and orthodontically treated persons. With age, decreases in arch length and width are usually witnessed, causing considerable anterior crowding and increased incisor irregularity. Because development of unsatisfactory dental alignment is often the medium- to long-term treatment outcome, there has been a consensus among clinicians that retention should last as long as perfect alignment is desired.
Lingual fixed retainers have thus become the only orthodontic device designed to remain bonded over many years and even decades. Based on the prevailing literature, it appears correct to state that no associations between bonded retainers and periodontal diseases could be substantiated. Also, no evidence indicates that fixed retention is a cause for increased incidence of dental caries or white spot lesions. Because recent evidence suggests that a bonded retainer may fail to preserve its passivity, the choice of inappropriate wire, material fatigue induced through physiologic masticatory forces, and bonding failure may be sources of developing undesirable effects.
It is well known that dental composite resins are subjected to intraoral degradation from exposure to mechanical, thermal, and chemical agents. The latter may significantly reduce bond strength, probably through diffusion into the composite-tissue interface.
Despite several in-vitro investigations focusing selectively on chemical, thermal, or mechanical stress-related impact, there is limited information on the effects of intraoral aging on composite properties in general, and nothing about intraoral aging of composites used to bond retainers in particular.
The purpose of this study was to assess the changes in the composition and mechanical properties of composite resins used for bonding orthodontic retainers after intraoral aging. Since differences in the curing mechanism may affect the composite properties, both light-cured and chemically cured materials were analyzed. The null hypothesis was that there would be no significant differences in the properties of the materials tested between the retrieved and the control samples.
Material and methods
Forty patients finished with orthodontic treatment and retention were selected according to the following inclusion criteria: bonded lingual retainers intended to be removed; no active caries, restorations, or fractures of mandibular anterior teeth; and adequate oral hygiene. The fixed retainers consisted of 0.022 × 0.022-in multistranded stainless steel wires (Tru-Chrome; Rocky Mountain Orthodontics, Denver, Colo) and had been adapted at chairside to fit passively. During bonding, the lingual surfaces of the 6 mandibular anterior teeth had been pumiced, rinsed with water, air-dried, and etched with 37% orthophosphoric acid liquid. The retainers had been bonded to enamel with (1) an unfilled chemically cured, 2-liquid bonding resin and a 2-paste orthodontic adhesive (Maximum Cure and Excel; Reliance Orthodontic Products, Itasca, Ill), and (2) an unfilled light-cured bonding resin and a light-cured orthodontic adhesive (Assure and Flow-Tain; Reliance Orthodontics Products). The compositions of the materials used in the study are listed in Table I .
| Ingredient | Excel | Flow-Tain | Maximum Cure: Liquid A | Maximum Cure: Liquid B | Assure | |
|---|---|---|---|---|---|---|
| Paste A | Paste B | |||||
| Resin content (wt%) | ||||||
| BisGMA | – | >20 | – | 50-80 | 50-80 | – |
| EBDMA | – | – | >10 | – | – | – |
| TEGDMA | 5-15 | >5 | <15 | – | – | – |
| MMA | – | – | 20-40 | 20-40 | – | |
| MMA-F | – | – | – | 2-5 | – | – |
| HEMA (stabilized) | – | – | – | – | – | 20-30 in acetone |
| Filler content (wt%) | ||||||
| Silica | 40-70 | 40-80 | – | – | – | – |
| Glass filler | – | – | >50 | – | – | – |
| Sodium fluoride | <1 | – | – | – | – | – |
Approval of the appropriate ethical committee was obtained for the retrieval (University of Athens, number 227, January 20, 2015). Bonding and retrieval of the retainers were performed by the same orthodontist (N.P.); removal of the fixed retainers was not related to the scope of this study. The materials used, the process adopted, and the date of insertion of the fixed retainer were meticulously recorded, and the retrieval process consisted of careful removal of the lingual retainers with a How’s pliers. The patient’s oral hygiene was assessed before the removal and throughout the maintenance period as well as by examination by the general practitioner.
After retrieval, the materials were forwarded to the laboratory for analysis. The enamel bonded surfaces of all composite retainer elements were examined under a stereomicroscope (M80; Leica, Wetzlar, Germany) at 16-times magnification. From the retrieved retainers, 15 lingual retainers bonded with Excel (EX-INV) and 11 with Flow-Tain (FT-INV) (total, 26) were collected; the remaining were excluded for reasons related to the lack of adequate mass of adhesive attached to the wires, broken adhesive, cohesive failure of the adhesive on the enamel, and fully or partially debonded adhesive from the enamel attached to the retainer. The intraoral aging periods ranged from 2.8 to 17.4 years for the EX-INV retainers and from 2.2 to 5.4 years for the FT-INV retainers.
Specimens of each adhesive material were cured in vitro as follows. Microscopic glass slides were covered with transparent polyester film strips and received 2 spacers (1.5 mm), 1 on each side. The adhesive resin pastes (approximately 2 mm in length × 2 mm in width; n = 3) were placed and covered with a second strip and slide. For the light-cured material, photopolymerization was performed with a light-emitting diode curing unit (Bluephase G2; Ivoclar Vivadent, Schaan, Liechtenstein) emitting 1.2 W per square centimeter of irradiance at the standard irradiation mode for 20 seconds. These specimens were stored in an incubator at 37°C for 7 days under dark and dry conditions, and were used as references for Excel (EX-REF) and Flow-Tain (FT-REF), respectively.
Attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR) was performed on the composite surfaces debonded from the lingual enamel surfaces to identify the molecular compositions and the degrees of carbon double-bond conversion. Three specimens from each retrieved set and control group were analyzed, to characterize the changes in molecular composition. Spectra acquisitions were obtained with a Fourier transform infrared spectrometer (Spectrum GX; PerkinElmer, Baconsfield, United Kingdom), using a microattenuated total reflection accessory (Golden Gate; Specac, Fort Washington, Pa) with a 1 × 1 mm diamond reflective element under the following conditions: 4000 to 650 cm −1 range, 4 cm −1 resolution, 20 scans coaddition, and 2-μm depth of analysis at 1000 cm −1 . All spectra were subjected to attenuated total reflection and baseline corrections. The peak absorbance height ratio of the aliphatic C = C (1638 cm −1 ) to aromatic C..C (1608 cm −1 ) groups used to measure the degree of cure was used to calculate the percentage differences in the degree of cure between the retrieved and the control specimens.
The same samples were further investigated for their structure and elemental composition, using a variable pressure scanning electron microscope (Quanta 200; FEI, Hillsboro, Ore) with a large field detector for secondary electron imaging and a solid-state detector for backscattered electron imaging. The operating parameters were 1 Pa chamber pressure (low-vacuum, 20 kV beam acceleration voltage, 105 μA beam current, and 800-times nominal magnification). The elemental composition was investigated by x-ray energy dispersive microanalysis. Spectra were collected using an X Flash 6|10 silicon drift detector (Bruker, Berlin, Germany) with a slew window under the same operating conditions, using a 64 × 64-μm sampling window and 200-second acquisition time. The elemental quantification was carried out in a standardless mode using atomic number, absorbance, and fluorescence correction factors.
All specimens were embedded in self-cure acrylic resin (10 mm thick) and, after metallographic grinding and polishing (300-1200 grit-size silicon-carbon papers, under water rinsing plus 5 minutes of sonication in water), were subjected to instrumented indentation testing with a Vickers indenter. One force-indentation depth curve was recorded from each specimen under a 4.9-N load and a 2-second dwell time, using a universal hardness testing machine (ZHU 0.2/Z2.5; Zwick Roell, Ulm, Germany). The indentation modulus, elastic index (defined as the elastic [area under the curve at the elastic limit] to total work ratio), and Vickers hardness (HV 5 ) were determined according to the ISO 14577-1 specification.
Statistical analysis
The values of the indentation modulus, elastic index, and Vickers hardness were statistically analyzed by 1-way analysis of variance with the independent parameter the aging status (in-vivo aged, control). The Student-Newman-Keuls test was used for multiple comparisons. For the percentage differences in the degree of cure, an unpaired t test was used. Data normality and equal variance were assessed by the Kolmogorov-Smirnov and Levene tests, respectively. Statistical analysis was performed by SigmaStat software (version 3.1; Systat Software, San Jose, Calif).
Results
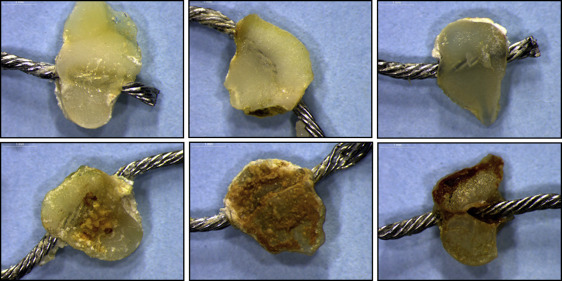
Figure 1 illustrates the stereoscopic images of retrieved composite surfaces bonded to enamel. Clear debonded surfaces were used for the analysis, whereas contaminated surfaces were excluded from the study.
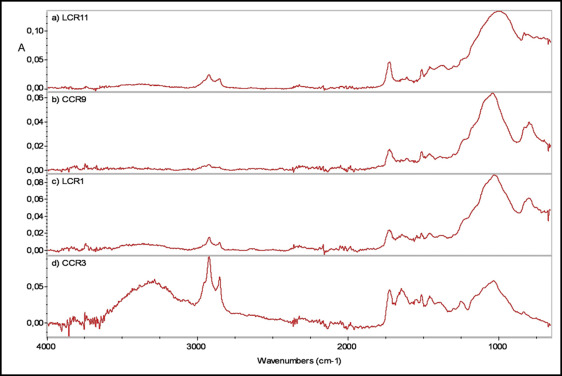
Representative ATR-FTIR spectra from the reference and in-vivo aged materials are shown in Figure 2 . The peak assignments include the following groups: OH free (3746 cm −1 ), OH hydrogen bonded (3380 cm −1 ), CH 2 /CH 3 (2920, 2853, 1465-1430 cm −1 ), C = O (1724, 1320, 1290 cm −1 ), C = C (1636, 980-950, 809 cm −1 ), aromatic C..C (3060, 1608, 1510, 770, 700 cm −1 ), aromatic-C-CH (1300 cm −1 ), aromatic-O-CH (1250 cm −1 ), CH-OH (1180-1160 cm −1 ), C-O-C (1100-1050 cm −1 ), and Si-O- (1080-1020, 790-800 cm −1 ), the latter attributed to silica and glass-filler interferences ( Fig 2 , A-C ). One specimen demonstrated organic (proteinaceous) contamination ( Fig 2 , D ) as evidenced by the increased relative intensity of the peaks at 3500 to 3000 cm −1 (OH and N-H), 1740 cm −1 (C = O), 1650 cm −1 (C = O of amide I), 1540 and 1250 cm −1 (C = O, N-H of amide II and III, respectively), and 1200 to 1100 cm −1 (C-OH). The spectra of the retrieved materials showed a greater contribution of the Si-O- peak relative to the C = O component. Expanded spectra of a retrieved ( Fig 3 , EX-INV) and a control ( Fig 3 , EX-REF) adhesive at the region used for assessment of the degree of cure are shown in Figure 3 . The results of the percentage differences in the degree of cure were (mean, standard deviation): Excel = +10.2 (3.1)% and Flow-Tain = +7.6 (2.4)%, with the positive values indicating higher conversion in the in-vivo aged materials. No statistically significant difference was found between the 2 means ( P >0.05).
Secondary electron and backscattered electron images from the reference and retrieved Excel material are depicted in Figure 4 . The backscattered electron images clearly demonstrate the filler particles with irregular shapes and higher mean atomic numbers (lighter gray scale). The main differences between the 2 images were the increased porosity in the retrieved material and the presence of some fractured ridges associated with the debonding procedure. The corresponding images for the Flow-Tain material (FT-REF and FT-INV) are illustrated in Figure 5 . The smooth morphology of the control surface demonstrated few regions with lighter gray scale, assigned to filler particles. The retrieved surfaces, though, demonstrated a complex pattern with microporosity, pitting, and cracking.




