Abstract
Benign lymphoepithelial lesions (BLLs) usually affect the parotid glands, resulting in their asymmetrical enlargement. The condition can be associated with Sjögren’s syndrome (SS), which in turn may predispose the affected patient to the development of malignant lymphoma. BLLs arising in the intraoral minor salivary glands are rare; such a lesion should prompt the work-up for SS. In some instances, a BLL of a minor salivary gland could represent the initial stage of a mucosa-associated lymphoid tissue (MALT) lymphoma. A case of BLL arising in the palatal minor salivary glands is reported here. The importance of an appropriate diagnostic work-up is emphasized and the relationship of BLL with SS is reinforced.
The condition today identified as a benign lymphoepithelial lesion (BLL) was first described by Johann Mikulicz in the 19th century as Mikulicz’s disease, characterized by an increase in the volume of the parotid glands. The term BLL was suggested by Godwin in 1952. BLLs show a predilection for the major salivary glands, particularly the parotid, and usually occur unilaterally, but may eventually be bilateral. Middle-aged women are most frequently affected; lesions involving the minor salivary glands are extremely rare, and the lacrimal glands may also be affected. Interestingly, based on current knowledge, the report of Mikulicz should now be classified as a mucosa-associated lymphoid tissue (MALT) lymphoma.
Clinically, the BLL presents as a firm, diffuse, and asymptomatic swelling of the affected gland, eventually leading to mild pain. The histopathological findings reveal an intense lymphocytic infiltrate involving the salivary gland acini, causing parenchymal atrophy. Although these acini may be destroyed, the ductal epithelium persists and becomes hyperplastic, forming epimyoepithelial islands.
BLLs are commonly associated with Sjögren’s syndrome (SS). Furthermore, cases previously diagnosed as a BLL may be re-classified as the initial stages of lymphoma, particularly MALT lymphoma.
A rare case of a palatal BLL linked to polyautoimmunity is presented herein, and the importance of an adequate diagnostic investigation is discussed.
Case report
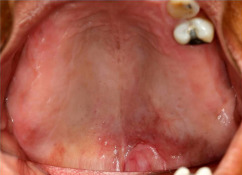
A 55-year-old woman was referred for the chief complaint of an asymptomatic palatal swelling lasting for approximately 6 months. Her past medical history revealed a heart murmur and systemic arterial hypertension, controlled with the use of captopril and hydrochlorothiazide. The patient had smoked around 15 cigarettes a day for the past 40 years, but denied the use of alcohol and/or other recreational drugs. An extraoral evaluation showed no relevant findings. Physical examination of the mouth revealed a diffuse firm nodule on the palate, measuring approximately 5 cm × 3 cm, covered by normal mucosa except for a slightly reddish central area ( Fig. 1 ). There were no signs of increased volume of the major salivary glands or cervicofacial lymphadenomegaly.
The patient had already undergone a computed tomography (CT) scan and magnetic resonance imaging (MRI) of the affected region, as requested previously by her general practitioner. The CT scan revealed a homogeneous hypodense image in the left posterior region of the palate, with no evidence of palatal cortical bone perforation. The MRI STIR sequences showed an area of hyperintense signal in the left palate, and post-contrast images highlighted a lesion with well-defined limits measuring 4.1 cm in largest diameter, as well as discrete contrast enhancement in the parotid glands. Based on the clinical and imaging findings, the differential diagnosis included pleomorphic adenoma, mucoepidermoid carcinoma, and adenoid cystic carcinoma.
A palatal incisional biopsy was performed under local anaesthesia. The histopathological analysis revealed a fragment of palatal mucosa partially covered by hyperkeratotic squamous epithelium and groups of salivary gland acini surrounded by a dense lymphocytic infiltrate. Dense clusters of epimyoepithelial cells surrounded by lymphoid follicles were also noted ( Fig. 2 ). The features on microscopy were characteristic of a BLL. Immunohistochemical analysis revealed positivity for pan-cytokeratins (AE1/AE3) in the epimyoepithelial islands, while the lymphocytic infiltrate was formed of T-lymphocytes (CD3+) and B-lymphocytes (CD20+), without restriction of light chains, confirming it to be a polyclonal and apparently benign infiltrate.

Based on the microscopy results, a clinical diagnosis of SS was considered. The patient was then questioned about having a dry mouth and/or dry eyes, and she reported mild xerostomia (not interfering with sleeping, and eating dry foods without drinking water was still possible), as well as fatigue and a cold feeling in her hands. Resting sialometry, a Schirmer test, minor salivary gland biopsy (lower lip), and serological analyses (anti-nuclear antibody (ANA), rheumatoid factor, anti-Sjögren’s syndrome-related antigen A (anti-SSA), and anti-Sjögren’s syndrome-related antigen B (anti-SSB)) were performed. The results of these tests are shown in Tables 1 and 2 , and these confirmed the diagnosis of SS in agreement with the criteria of the American–European Consensus Group and American College of Rheumatology.
| Criteria | Test | Reference | Result | Evaluation |
|---|---|---|---|---|
| Serology | Anti-SSA | Negative: <7.0 U/ml | Anti-SSA: 240.9 U/ml | Positive |
| Anti-SSB | Inconclusive: 7.1 < result < 10.0 U/ml Positive: >10.1 U/ml |
Anti-SSB: 108 U/ml | ||
| Rheumatoid factor | <14.0 IU/ml = negative | 0.0 IU/ml | Negative | |
| Anti-nuclear antibody | Negative | Negative | Negative | |
| Histopathology | Focus score (FS) | Positive: FS ≥1.0 | FS = 1.3 (labial glands) Confluent inflammatory infiltrate (palatal glands) |
Positive |
| Sialometry | Unstimulated salivary flow test | >1.5 ml/15 min | 1.2 ml/15 min | Positive |
| Schirmer test | >5 mm/min | Right eye = 4 mm/min Left eye = 5 mm/min |
Positive | |
| Xerostomia | No xerostomia | Yes (mild) | Positive | |
| Xerophthalmia | No xerophthalmia | No | Negative |
| AECG, 2002 a | Patient result | ACR, 2012 b | Patient result |
|---|---|---|---|
| Ocular symptoms | Negative | Histopathology | Positive |
| Oral symptoms | Positive | Autoantibodies | Positive |
| Ocular signs | Positive | Ocular staining score | Not evaluated |
| Histopathology | Positive | ||
| Salivary gland involvement | Positive | ||
| Autoantibodies | Positive |
a AECG, 2002 is fulfilled when four of the six criteria are positive, including histopathology or autoantibodies.
b ACR, 2012 is fulfilled when two of the three criteria are positive.
Once the diagnosis of SS had been established, and based on the elevated prevalence of associated autoimmune diseases with this condition, anti-thyroglobulin and anti-thyroid peroxidase were tested, which led to the diagnosis of Hashimoto’s thyroiditis. The patient was referred to the rheumatology and ophthalmology services for further evaluation. Pilocarpine was prescribed for salivary flow control, and fluoride trays were made for caries control. The patient was started on hydroxychloroquine 200 mg twice daily for the management of SS. Levothyroxine 88 mg once daily was found to be adequate for controlling the thyroid hypofunction as well as the fatigue. A partial reduction of the BLL was achieved, and no additional swelling was noted after 14 months of follow-up.
Discussion
Nodular lesions in the palatal region frequently cause immediate concern, because this site is commonly affected by minor salivary gland tumours. In fact, approximately 50% of these tumours are malignant and require more radical approaches.
Nevertheless, the occurrence of BLLs related to the minor salivary glands is extremely rare, as these are more common in the major salivary glands, particularly the parotid glands. In a search of the literature, a total of 10 additional case reports of BLLs involving the minor salivary glands, published between 1960 and 2015, were identified. Of the total 11 cases, including the case presented herein, nine involved the palate (81.8%) and seven cases occurred in women (63.6%). The affected patients had a mean age of 66 years, ranging from 24 to 84 years, and the disease evolution ranged from 2 to 18 months ( Table 3 ). Only five patients (including the present one) (45.5%) were diagnosed with SS. However, some were not adequately evaluated, which suggests that this number could be higher. In short, intraoral BLLs associated or not with SS are very rare, but it is crucial that a diagnosis of BLL in the orofacial region leads to a clinical investigation for SS, even in the absence of classical clinical signs of the disease.



