This article describes how dentists can recognize osteoporosis before fractures develop, and discusses whether osteoporosis affects tooth loss or inhibits implant osseointegration. Some success in diagnosing osteoporosis has been obtained using clinical questionnaires that attempt to identify those who have strong risk factors for the disease, and analysis of the sparse trabeculation and thinning of the mandibular cortex often seen in dental panoramic radiographs. The role of osteoporosis in periodontal disease is unclear as there are many conflicting reports, but the evidence suggests that tooth loss may be more prevalent in patients with osteoporosis.
- •
Osteoporosis can be diagnosed using clinical questionnaires that attempt to identify those who have strong risk factors for the disease. Lifestyle factors such as the excessive use of alcohol and smoking are known to be risk factors for osteoporosis.
- •
Osteoporosis can be diagnosed through an analysis of the sparse trabeculation and thinning of the mandibular cortex often seen in dental panoramic radiographs.
- •
As a result of the radiographic and clinical findings, the dentist may suspect a patient has an increased risk of osteoporotic fractures and refer them for further tests, preventive advice, and treatment.
- •
There is no evidence that osteoporosis initiates periodontal disease. The role of osteoporosis in periodontal disease is unclear as there are many conflicting reports, but the evidence suggests that tooth loss may be more prevalent in patients with osteoporosis.
- •
There is no evidence that osteoporosis affects the clinical success rate of dental implants.
Osteoporosis assessment tools
Osteoporosis is a disease characterized by a severe loss of bone mineral density that can eventually result in fracture. The femur and spine are most commonly affected, and bone density measurements at these sites are expressed as T-score values. These values are the number of standard deviations by which the bone mineral density value lies below the sex-matched young adult value. Therefore as the T score becomes more negative, the patient is judged to be more severely affected by osteoporosis. According to the World Health Organization (WHO), patients with a T score of less than −2.5 are osteoporotic, whereas those with a T score between −1 and −2.5 have a bone mineral density less than normal and are osteopenic.
The sensitivity of a test measures the proportion of people with osteoporosis that the test can correctly identify, whereas specificity provides a measure of a test’s ability to correctly identify healthy people who do not have osteoporosis. By plotting sensitivity against (1 − specificity), the diagnostic accuracy of the test can be calculated as the area under the curve, a technique called receiver-operating characteristic curve (ROC) analysis. A test with a value of 0.5 has no diagnostic ability, and the accuracy of the test increases as the ROC value approaches unity.
Risk factors
The presence of osteoporosis and the likelihood that an individual will develop a fracture can be identified. Numerous assessment tools have been developed based on the patient’s risk factors, namely body weight, age, current estrogen use, rheumatoid arthritis, ethnicity, and a history of nontraumatic fracture, and these tools have been compared against the WHO gold-standard diagnosis of osteoporosis. None of these tests are sufficiently accurate to provide a confirmatory diagnosis of osteoporosis. Rather, their purpose is to identify those people at increased risk of an osteoporotic hip fracture so that interceptive therapy can be given. However, opinion is divided as to where to place the diagnostic threshold. If the objective is to obtain a test with maximum sensitivity, this can result in a large number of inappropriate false-positive referrals. The patients who are affected have to undergo needless worry. In addition, the referral of significant numbers of healthy individuals uses scant health care resources and is not cost effective. Conversely, using a high-specificity and low-sensitivity threshold will result in a failure to identify many women with osteoporosis, a proportion of whom will develop hip fractures in the future.
Dentists can use information from the patient’s dental panoramic radiograph and an assessment of their clinical risk factors to determine whether they are at risk of osteoporosis. It is not recommended that a radiograph is taken specifically for an osteoporosis diagnosis, because the test is not as accurate as the medical criterion test, but it is quick and convenient for the patient.
In a large study population of 7779 women in the United States, Gourlay and colleagues found that body weight had an area under the ROC curve of 0.73 (95% confidence interval [CI] 0.72–0.75) in identifying low bone density in women aged 67 years and older. The use of multiple clinical risk factors has been used to improve the osteoporosis diagnostic tool. Combinations of clinical factors such as age, weight, use of hormone replacement therapy, and a history of low trauma fracture are used in the Osteoporosis Index of Risk (OSIRIS) index. Karayianni and colleagues analyzed 653 women aged 45 to 70 years and showed that OSIRIS had an area under the ROC curve of 0.83 (95% CI 0.81–0.87) in detecting osteoporosis at either the hip, femoral neck, or spine.
Radiographic Changes in Dental Panoramic Radiographs
The inferior cortical border of the mandible is thinned in osteoporosis, and this can be detected on panoramic radiographs. Where a thin mandibular cortex is visible, the dentist should investigate the clinical risk factors to determine whether the patient should be referred for further investigation and diagnosis.
Fig. 1 A illustrates the clearly distinct outline of the normal inferior mandibular cortex. Fig. 1 B shows the indistinct endosteal border of the mandibular cortex in a patient with osteoporosis of the hip. Such a radiographic finding is often, but not always, observed. Fig. 1 C is a more detailed image of this region showing the multiple layered appearance and porous nature of the cortex. A series of patchy radiopaque “residues” are also seen, representing the remains of the resorbed cortex that now form an intermediate, coarsely structured cancellous bone.
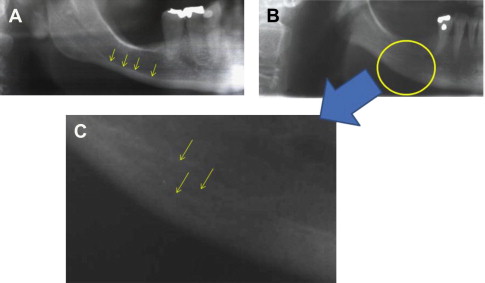
Can dental radiographs be used to identify patients with osteoporosis?
Roberts and colleagues evaluated the change in mandibular cortical width in 4949 dental panoramic tomograms of patients aged 15 to 94 years. In women, there was no change in cortical width during adulthood until age 42.5 years, when bone loss accelerated thereafter. This pattern of cortical thinning also occurs in the hip, where it may result in weakening and bone fracture following trauma.
If dentists are to be more involved in proactively identifying those with osteoporosis, patients with mandibular cortical thinning should be referred to their physician for further investigation. Accurate measurement of the cortical width can be difficult, given the indistinct nature of the endosteal cortical border (see Fig. 1 ); therefore Horner and colleagues proposed a 3-mm threshold as a compromise between detecting the maximum number of “true” osteoporotic patients and avoiding unnecessarily alarming patients who were subsequently found to have a normal bone density. This approach produced low sensitivity (8%) at detecting osteopenia at either the hip or spine, but specificity was high (98.7%). Others have proposed higher cortical-width thresholds. Lee and colleagues used a threshold of 3.9 mm, and found a sensitivity of 72.9% and specificity of 70.2% in detecting low bone mineral density at the femoral neck. Using this higher threshold increased the sensitivity of the osteoporosis detection method, but decreased the specificity. Damilakis and Vlasiadis found that a threshold of 4.1 mm produced sensitivity of 58.8% and specificity of 66.7% at detecting patients with osteoporosis at either the lumbar spine or the hip. Taguchi and colleagues found that a cortical-width threshold of 4.3 mm produced sensitivity of 90% and specificity of 45.3% for identifying women with osteoporosis. Whatever the threshold chosen, the main conclusion from these studies is that a dental panoramic radiograph is not justified solely for the purposes of triage screening for osteoporosis, because of the high percentage of false positives that would result. But when a radiograph is taken for unrelated reasons and the dentist makes an incidental finding of cortical thinning and porosity, the patient may be at high risk of osteoporosis, and a referral to the patient’s physician may be indicated.
The mandible is composed of 2 thick cortical plates with a sandwich of cancellous bone. At first glance, the cortical width might be thought to be a better predictor of osteoporosis at the femoral neck than at the lumbar spine, given the highly cortical nature of the former. However, there is no clear evidence for a difference in the detection rate of osteoporosis at the 2 sites.
Other studies have found that a sparse cancellous bone pattern in the mandibular premolar region is associated with an increased number of self-reported fractures. This pattern is seen as large intertrabecular spaces in the interdental area of the mandible. The loss of connectedness of the cancellous architecture in the mandible may reflect a similar general pattern of bone loss elsewhere in the aging skeleton.
Future research is also needed to identify those factors that may influence a patient’s access to osteoporosis diagnosis and treatment services when referred by the dentist; for example, the support of the physician, the available resources, the type of health care system, the patient’s consent, and the remuneration system offered to the dentist. Diagnosis of a patient’s risk of osteoporosis will increasingly use computer-assisted diagnosis to automatically detect the cortices on the panoramic radiograph, measure cortical width, and alert the dentist as to whether the patient has a high risk of osteoporosis. Such computer software already exists, and also allows data from the cortical-width measurements to be combined with clinical risk factors to produce a powerful diagnostic tool.
Clinical Risk Factors for Osteoporosis
The major and minor risk factors for osteoporosis are listed in Table 1 . One of the main risk factors is a low body weight or body mass index (BMI).
A low body weight is a strong risk factor for osteoporosis. Those young women with anorexia nervosa are at an increased risk of premenopausal osteoporosis. Rapid bone loss occurs early, but bone density increases following weight gain.
| Major Clinical Risk Factors | Less Important Clinical Risk Factors |
|---|---|
| Old age | Dementia |
| Low body weight | High alcohol intake |
| Ovariectomy at an early age | Little physical activity |
| Previous low-impact bone fracture | |
| Rheumatoid arthritis and corticosteroids |
Are those with osteoporosis more likely to suffer from periodontal disease?
Periodontal disease is triggered by plaque and poor dental hygiene. As periodontal bone loss can continue in some patients despite good oral-hygiene practices, many have considered whether osteoporosis could be a significant risk factor for periodontal disease. Osteoporosis causes a reduction in bone mass throughout the body, and the increased porosity of the alveolar bone surrounding the teeth may make it less resistant to resorption with plaque-induced inflammation. Unfortunately, most of the studies are cross-sectional in design and definitive conclusions are often not possible because of the small sample sizes. Pilgram and colleagues examined 135 patients, and found weak correlations between clinical attachment loss and bone mineral density. A larger study involving 778 subjects, about half (53%) of whom were women, found a weak association between osteoporosis and periodontitis in women (crude odds ratio [OR] 1.8, 95% CI 1.1–3.3).
Cross-sectional studies can be used to study risk factors (eg, osteoporosis) on an outcome (eg, periodontal disease or tooth loss), but this design is weak. To minimize bias, the assessment of the outcome must be blinded to prognostic information. Most studies comparing osteoporosis and periodontal disease are retrospective analyses of databases. When determining whether osteoporosis is an important prognostic factor in determining tooth loss, it is important to adjust for other prognostic variables, for example, oral hygiene and smoking. Multiple regression analysis is therefore usually required, but when comparing these studies investigators have included a variety of different variables. Could a declining alveolar bone density and osteoporosis account for the increased number of teeth lost by elderly women in some surveys? Tooth loss is the result of many social and clinical factors that interact in a complicated way, and it is difficult to isolate the effect of osteoporosis. The patient’s caries and periodontal disease, as well as attitude and personal finance issues, are often the main factors in determining whether a patient has a tooth removed.
One of the first early studies found a low, but statistically significant, correlation between the periodontal index of posterior teeth and osteoporosis of the metacarpal bone, although the smoking status of the patients was not assessed. Other studies have found a small additional effect of osteoporosis, even after adjustment for smoking and age. Both smoking and increasing age have a negative effect on both osteoporosis and periodontal disease. The methods of assessing the severity and extent of periodontal disease and osteoporosis in these studies vary widely, with consequently a large variation in the comparison groups.
There is general agreement in the literature that osteoporosis does not initiate periodontal disease, but there is considerable controversy as to whether osteoporosis can accelerate the progression of periodontal disease. Kribbs examined 85 women with osteoporosis and 27 normal women, and found no difference in the periodontal measurements between the 2 groups. The women with osteoporosis had a history of vertebral compression fractures, but the severity of the osteoporosis or bone mass at this site was not available. If osteoporosis has a small effect on the progression of periodontal disease, the effect is more likely to be observed in the severely osteoporotic elderly. Von Wowern and colleagues found that a group of osteoporotic women had a significantly lower bone mineral content in their forearm compared with a control group, and there was a significantly greater loss of periodontal attachment in the osteoporotic women, but no differences were found with respect to plaque score and gingival bleeding.
Osteoporosis does not initiate periodontal disease. Its role, if any, in aggravating preexisting periodontal disease is controversial.
In a study by Elders and colleagues, no correlation was seen between the mean probing depth and the lumbar spine bone mineral density in a sample of 286 women, of whom 60 (21%) were edentulous. Only premenopausal or perimenopausal women were studied, as only those aged 46 to 55 years were included. Studies involving younger women are unlikely to involve severely affected osteoporotic individuals, as osteoporosis is mainly a disease of the elderly. Despite this, Taikaishi and colleagues found an association between alveolar bone mineral density and periodontal pocket depth in a sample of 40 perimenopausal and postmenopausal women aged 50 to 69 years. Direct evidence for a causal relationship between declining estrogen levels and reduced alveolar bone density was found in the more porous alveolar bone of ovariectomized monkeys. The less dense alveolar bone may be more prone to periodontal disease.
Many of the population studies were not conducted in populations being seen specifically for dental services. For example, Gomes-Filho and colleagues examined patients attending a reproduction assistance program. In the group with periodontal disease, 139 (83.3%) postmenopausal women had a very high prevalence of osteoporosis or osteopenia, compared with those with no periodontal disease (65.9%). Other studies are ancillary studies of large osteoporotic fracture studies, for example, the Pittsburgh Clinical Center for the Study of Osteoporotic Fractures. In this 2-year longitudinal study, no difference in hip bone mineral density was found between women with and without periodontal disease. Differences in the assessment of periodontal disease, skeletal bone density, and age group studied limit any comparison between studies. Inagaki and colleagues used a community-based Oral Health Screening Program to recruit 101 postmenopausal women. The investigators assessed the patients’ osteoporotic status by means of computed densitometry of the metacarpal bone, and did not use the WHO classification. The age-adjusted odds of having fewer than 20 teeth were greater among those postmenopausal women with a very low bone mineral density in comparison with the normal group (OR = 5.9). However, the 95% CIs for this were wide (1.2–28.6), indicating that a larger sample size is required. Nineteen women were present in the very low bone mineral density group, of whom 6 had fewer than 20 teeth. The odds of tooth loss and periodontal disease were significantly higher in postmenopausal Japanese women with reduced metacarpal bone mineral density. Statistical adjustment for smoking status and prevalence of teeth with caries and restorations did not affect the relationship between severity of periodontal disease and metacarpal bone density.
In a multicenter study funded by the European Union (Osteodent Study), 665 women aged 45 to 70 years were recruited. Bone density was measured at the total hip, femoral neck, and lumbar spine. On average, the osteoporotic subjects had about 3 fewer teeth than normal subjects. If those with edentulousness are excluded, the osteoporotic group had a mean of about 2 fewer teeth. After adjusting for age, smoking, and center, there was a significant association between osteoporosis and having fewer than 28 teeth remaining ( P = .011), which may indicate that osteoporosis has an effect on particular tooth groups (eg, molar teeth rather than anterior teeth).
Astrom and colleagues examined the dental status of 14,375 individuals in a Swedish community. For both men and women the incidence of fracture was correlated with the degree of tooth loss, but the statistical relationship of both factors is not proof of a causative relationship. There is an increasing tooth loss and bone fracture incidence with age, but both may be occurring independently. However, in this study, in those older than 70 years women generally had a fewer mean number of teeth at each age than men. In most studies, it is a small minority of the study population that accounts for the majority of the tooth loss.
In a cross-sectional study, Bollen and colleagues reported no significant difference in the number of teeth between those with and without bone fractures. Patients were asked to recall their previous fractures, and this potentially selective recall may have introduced error. May and colleagues found a significant association in 608 men and 874 women, aged 65 to 76 years, between the bone mineral density at the hip and spine and the patients’ self-reported tooth loss. Adjustment for confounding variables such as age and cigarette smoking did not alter the result. It is not known whether any misclassification of self-reported tooth number produced a random error in this study. The investigators argued that if all patients were inconsistent in stating how many teeth they had lost, a random error would result that would add variability to the data, and no significant association would have resulted. In this study, no information was provided on how these patients had lost their teeth. Trauma, caries, and periodontal disease are usually the main contributing factors to the number of teeth lost by patients. These factors are active throughout life, unlike a low bone mineral density, which usually develops only later in life. Klemetti and Vainio reported no association between tooth loss and bone mineral density at the hip and lumbar spine. Similarly, Earnshaw and colleagues found no relationship between tooth count and bone mineral density in early postmenopausal women.
Krall and colleagues found that systemic bone loss predicted tooth loss in a sample of 189 postmenopausal women aged 41 to 71 years in a 7-year prospective study. Subjects in this study were included if they had a normal spinal bone mineral density and a low dietary calcium intake, but this may mean that the study participants were unrepresentative of the general population of women from the United States. The number of teeth lost during the study was assessed by questionnaire, which may have also introduced some measurement error. There were no measurements in the sample of periodontal disease status and no investigation of the reasons for the tooth loss.
Yoshihara and colleagues examined 600 people aged 70 years, and found a significant relationship between the number of remaining teeth and bone mineral density of the os calcis. In this study, measurement at the os calcis was undertaken using ultrasound densitometry, which is not considered to be the gold-standard measurement technique for bone mineral density. This study demonstrated a clinically significant reduction of 2 to 3 teeth when comparing the mean tooth number for the female osteopenia group (15.97, SD = 9.98) with that of the control group (18.31, SD = 8.06). However, given the large standard deviation associated with the number of teeth, it would not be accurate to say to an individual patient with osteoporosis that they would be likely to lose a further 2 to 3 teeth as a result of this disease.
The United Kingdom Adult Dental Health Survey has shown that women have fewer sound and untreated teeth than do men. Osteoporosis may be the cause of this difference, as women are more frequently affected by osteoporosis. Alternatively, women may be more demanding about aesthetics and request the extraction of irregular teeth. The latter explanation would predict that more elderly women than men of the same age have missing anterior teeth, but have a similar mean number of posterior teeth.
A prospective cohort study of 1341 recruited postmenopausal women were followed up for a mean of 5.1 years to investigate the factors predisposing to tooth loss. Preexisting periodontal disease, plaque, a history of smoking, diabetes, and previous missing teeth at the baseline examination were all important in predicting future tooth loss. A lower BMI is associated with osteoporosis, but in this study a higher BMI was associated with an increased incidence of tooth loss. The effect was small (about one tooth on average was lost for a 5 kg/m 2 increase in BMI). In a longitudinal 5-year study involving postmenopausal Japanese women, Iwasaki and colleagues found that those with the greater reduction in bone mineral density over this period were more likely to lose teeth, although the effect was small. The relative risk for the highest tertile percentage change from baseline bone density was 1.38 (95% CI 1.11–1.72) for the lumbar spine and 1.27 (95% CI 1.01–1.59) for the femoral neck. The result remained significant after controlling for menopausal age, high alcohol consumption, vitamin D and calcium intake, BMI, and diabetes. Smokers were excluded from the study.
In research there tends to be a bias against publishing studies that show no statistically significant effects, so it can be difficult to assess the true association between variables. Studies that do not show any association between variables or any benefit of a particular drug intervention tend to remain unpublished. There are, of course, exceptions to this general statement. For example, in a Japanese cross-sectional study, there was no significant difference in the number of teeth remaining between those who used estrogen and those who did not. There are studies showing the opposite, that is, a positive beneficial effect of estrogen replacement on tooth loss.
In conclusion, there is some evidence for a small clinical effect of osteoporosis on increasing tooth loss. However, further laboratory investigations are required to provide a biological mechanism of action that will, in turn, supply a clinically testable hypothesis.
Do oral implants have less success when used in osteoporotic patients?
Many factors interact to determine the success rate of oral implants. The failure rate of this form of treatment is very low; therefore if the effect of a factor on the rate of implant success is to be determined, the studies must be large and therefore expensive. However, if a very large study is required to demonstrate a small statistically significant effect, the clinical importance of that factor must also be separately considered. When considering the published studies, the question has to be asked whether other potentially more influential factors have been accounted for in the analysis or in the experimental methodology. Lee and colleagues concluded, in a retrospective study, that implant treatment should not be considered to be a particularly high-risk procedure for older patients with controlled systemic conditions. However, they collected data from only 35 patients who were followed for a mean period of only 32.7 months. If a dental implant is placed in cancellous bone that has sparse trabeculation and is of low density, it will provide poor support for an implant in function and may result in its loss. Glauser and colleagues found that only two-thirds of implants placed in the posterior maxilla were successful, probably because of the low-density bone that is usually found in this region.
The importance of the density of the host bone is well known in orthopedic surgery. In the older osteoporotic patient with a hip fracture, the bone quality is extremely important in determining the success or failure of internal fixation. Complications of hip-fracture fixation are common. Where bone support is critically low, the surgeon may be unable to place any internal fixation device. The Singh index has been used in assessment of femoral trabecular bone structure, but has the disadvantage that there is considerable interobserver variability with its use.
Risk factors
The presence of osteoporosis and the likelihood that an individual will develop a fracture can be identified. Numerous assessment tools have been developed based on the patient’s risk factors, namely body weight, age, current estrogen use, rheumatoid arthritis, ethnicity, and a history of nontraumatic fracture, and these tools have been compared against the WHO gold-standard diagnosis of osteoporosis. None of these tests are sufficiently accurate to provide a confirmatory diagnosis of osteoporosis. Rather, their purpose is to identify those people at increased risk of an osteoporotic hip fracture so that interceptive therapy can be given. However, opinion is divided as to where to place the diagnostic threshold. If the objective is to obtain a test with maximum sensitivity, this can result in a large number of inappropriate false-positive referrals. The patients who are affected have to undergo needless worry. In addition, the referral of significant numbers of healthy individuals uses scant health care resources and is not cost effective. Conversely, using a high-specificity and low-sensitivity threshold will result in a failure to identify many women with osteoporosis, a proportion of whom will develop hip fractures in the future.
Dentists can use information from the patient’s dental panoramic radiograph and an assessment of their clinical risk factors to determine whether they are at risk of osteoporosis. It is not recommended that a radiograph is taken specifically for an osteoporosis diagnosis, because the test is not as accurate as the medical criterion test, but it is quick and convenient for the patient.
In a large study population of 7779 women in the United States, Gourlay and colleagues found that body weight had an area under the ROC curve of 0.73 (95% confidence interval [CI] 0.72–0.75) in identifying low bone density in women aged 67 years and older. The use of multiple clinical risk factors has been used to improve the osteoporosis diagnostic tool. Combinations of clinical factors such as age, weight, use of hormone replacement therapy, and a history of low trauma fracture are used in the Osteoporosis Index of Risk (OSIRIS) index. Karayianni and colleagues analyzed 653 women aged 45 to 70 years and showed that OSIRIS had an area under the ROC curve of 0.83 (95% CI 0.81–0.87) in detecting osteoporosis at either the hip, femoral neck, or spine.
Radiographic Changes in Dental Panoramic Radiographs
The inferior cortical border of the mandible is thinned in osteoporosis, and this can be detected on panoramic radiographs. Where a thin mandibular cortex is visible, the dentist should investigate the clinical risk factors to determine whether the patient should be referred for further investigation and diagnosis.
Fig. 1 A illustrates the clearly distinct outline of the normal inferior mandibular cortex. Fig. 1 B shows the indistinct endosteal border of the mandibular cortex in a patient with osteoporosis of the hip. Such a radiographic finding is often, but not always, observed. Fig. 1 C is a more detailed image of this region showing the multiple layered appearance and porous nature of the cortex. A series of patchy radiopaque “residues” are also seen, representing the remains of the resorbed cortex that now form an intermediate, coarsely structured cancellous bone.
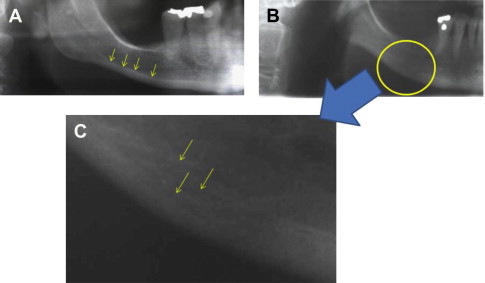
Can dental radiographs be used to identify patients with osteoporosis?
Roberts and colleagues evaluated the change in mandibular cortical width in 4949 dental panoramic tomograms of patients aged 15 to 94 years. In women, there was no change in cortical width during adulthood until age 42.5 years, when bone loss accelerated thereafter. This pattern of cortical thinning also occurs in the hip, where it may result in weakening and bone fracture following trauma.
If dentists are to be more involved in proactively identifying those with osteoporosis, patients with mandibular cortical thinning should be referred to their physician for further investigation. Accurate measurement of the cortical width can be difficult, given the indistinct nature of the endosteal cortical border (see Fig. 1 ); therefore Horner and colleagues proposed a 3-mm threshold as a compromise between detecting the maximum number of “true” osteoporotic patients and avoiding unnecessarily alarming patients who were subsequently found to have a normal bone density. This approach produced low sensitivity (8%) at detecting osteopenia at either the hip or spine, but specificity was high (98.7%). Others have proposed higher cortical-width thresholds. Lee and colleagues used a threshold of 3.9 mm, and found a sensitivity of 72.9% and specificity of 70.2% in detecting low bone mineral density at the femoral neck. Using this higher threshold increased the sensitivity of the osteoporosis detection method, but decreased the specificity. Damilakis and Vlasiadis found that a threshold of 4.1 mm produced sensitivity of 58.8% and specificity of 66.7% at detecting patients with osteoporosis at either the lumbar spine or the hip. Taguchi and colleagues found that a cortical-width threshold of 4.3 mm produced sensitivity of 90% and specificity of 45.3% for identifying women with osteoporosis. Whatever the threshold chosen, the main conclusion from these studies is that a dental panoramic radiograph is not justified solely for the purposes of triage screening for osteoporosis, because of the high percentage of false positives that would result. But when a radiograph is taken for unrelated reasons and the dentist makes an incidental finding of cortical thinning and porosity, the patient may be at high risk of osteoporosis, and a referral to the patient’s physician may be indicated.
The mandible is composed of 2 thick cortical plates with a sandwich of cancellous bone. At first glance, the cortical width might be thought to be a better predictor of osteoporosis at the femoral neck than at the lumbar spine, given the highly cortical nature of the former. However, there is no clear evidence for a difference in the detection rate of osteoporosis at the 2 sites.
Other studies have found that a sparse cancellous bone pattern in the mandibular premolar region is associated with an increased number of self-reported fractures. This pattern is seen as large intertrabecular spaces in the interdental area of the mandible. The loss of connectedness of the cancellous architecture in the mandible may reflect a similar general pattern of bone loss elsewhere in the aging skeleton.
Future research is also needed to identify those factors that may influence a patient’s access to osteoporosis diagnosis and treatment services when referred by the dentist; for example, the support of the physician, the available resources, the type of health care system, the patient’s consent, and the remuneration system offered to the dentist. Diagnosis of a patient’s risk of osteoporosis will increasingly use computer-assisted diagnosis to automatically detect the cortices on the panoramic radiograph, measure cortical width, and alert the dentist as to whether the patient has a high risk of osteoporosis. Such computer software already exists, and also allows data from the cortical-width measurements to be combined with clinical risk factors to produce a powerful diagnostic tool.
Clinical Risk Factors for Osteoporosis
The major and minor risk factors for osteoporosis are listed in Table 1 . One of the main risk factors is a low body weight or body mass index (BMI).
A low body weight is a strong risk factor for osteoporosis. Those young women with anorexia nervosa are at an increased risk of premenopausal osteoporosis. Rapid bone loss occurs early, but bone density increases following weight gain.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


