Introduction
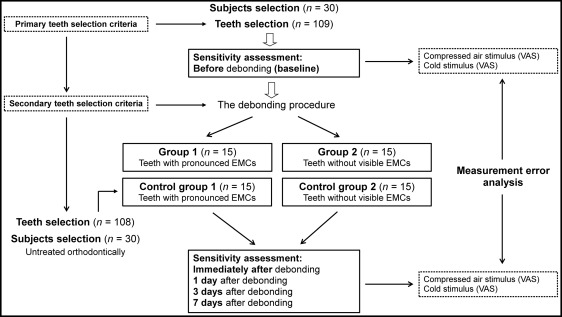
Our aim was to assess the possible changes in sensitivity of teeth with and without visible enamel microcracks (EMCs) up to 1 week after the removal of metal brackets.
Methods
After debonding, 15 patients possessing teeth with visible EMCs and 15 subjects whose teeth were free of EMCs were enrolled in the study. For each experimental group, a control group was formed. The assessments of tooth sensitivity elicited by compressed air and cold testing were performed 5 times: just before debonding, immediately after debonding, and at 1, 3, and 7 days after debonding. Tooth sensitivity was recorded on a 100-mm visual analog scale.
Results
For the patients without visible EMCs, discomfort peaked immediately after debonding and started to decrease on day 1; at 1 week after debonding, the visual analog scale scores were lower than just before debonding and immediately after debonding. For the subjects possessing teeth with visible EMCs, the pattern of sensitivity dynamic was inherently the same. However, the patients with visible EMCs showed higher visual analog scale values at each time interval.
Conclusions
Debonding leads to a short-term increase in tooth sensitivity. EMCs, a form of enamel damage, do not predispose to greater sensitivity perception in relation to bracket removal.
Highlights
- •
Tooth sensitivity was evaluated up to 1 week after removal of metal brackets.
- •
Discomfort of teeth with and without visible enamel microcracks was assessed.
- •
Debonding led to a short-term (7 days) increase in tooth sensitivity.
- •
Patients with visible enamel microcracks showed higher absolute discomfort values.
- •
The pattern of sensitivity is inherently the same for all subjects.
One factor that discourages patients from seeking orthodontic treatment is the anxiety and fear of related pain and discomfort from the orthodontic appliance. Studies about patients experiencing pain or discomfort during orthodontic therapy have shown values up to 95%. The levels and durations of discomfort during elastic separation, placement of different bracket systems, activation of archwires, and use of intermaxillary elastics are presented in detail in previously published investigations. However, the pain experienced during debonding is currently poorly quantified. A few studies have assessed the level of discomfort during debonding with different instruments or biting on a soft acrylic wafer at the time of bracket removal. Possible changes in the pain dynamic with time after debonding have been not observed. Therefore, there is little knowledge regarding the duration and the intensity of discomfort experienced during the time period after bracket removal.
The perception of pain during orthodontic force application has generally been attributed to mechanical damage and inflammatory reactions in the periodontium. However, there is evidence suggesting that elements in the dental pulp may contribute as well. A trend has been noted where reports of greater orthodontic tooth pain were associated with increased pulpal sensitivity evoked by electrical tooth stimulation.
Caries, restorations, enamel irregularities, and tooth structure defects might cause variations in tooth sensitivity assessment. Enamel microcracks (EMCs), a form of enamel damage after debonding, are a concern for patients receiving orthodontic care. It has been shown that EMCs may jeopardize the integrity of the enamel and cause stains and plaque accumulation on the fractured surfaces. The susceptibility to carious lesions increases and affects the appearance of the teeth. Pronounced EMCs (visible with the naked eye under normal room illumination) are often noticed by the patients at the beginning of the orthodontic treatment or after the removal of fixed appliances. Current knowledge indicates that measured quantitative pronounced EMC characteristics (width of EMCs is expressed in microns and length in millimeters) are greater in comparison with weak EMCs (not apparent under normal room illumination but visible by scanning electron microscopy), but this does not predispose to increased EMCs after debonding. However, if a visible EMC is located on more than a third of the buccal tooth surface, its inclination does not exceed 30° to 45°, and ceramic brackets are used, the combination of these EMC characteristics might lead to a greater risk of more tooth surface damage after debonding up to 20.4%. Still, it is unknown whether the teeth with visible EMCs might be more susceptible to discomfort and pain during the orthodontic treatment.
Therefore, the aims of this study were to (1) evaluate the sensitivity of teeth with pronounced EMCs and to compare it with teeth without visible EMCs just before debonding and up to 1 week after the removal of fixed appliances; (2) determine the time for a clinically significant reduction in teeth sensitivity; (3) determine whether there is a correlation between the tooth sensitivity values just before debonding and the likelihood of their increasing during the removal of brackets; and (4) find out whether insensitive teeth with and without visible EMCs just before debonding might progress to sensitive ones immediately after debonding.
Material and methods
A detailed medical history, including questions about tooth sensitivity before and during orthodontic treatment with fixed appliances, was taken of each patient in a questionnaire ( Table I ). The selection criteria for the subjects and the teeth are listed in Table II . All eligible patients were given verbal and written information about the study and the opportunity to ask further questions.
| Patient name: | Date: |
| Birth date: | Gender: |
| Date of bonding procedure: | Date of debonding procedure: |
| Duration of treatment: | Type of brackets: |
|
|
| Selection criteria for subjects |
| Informed consent from patients and parents |
| Patients aged 14-24 years |
| Patients bonded with metal brackets (Discovery; Dentaurum, Ispringen, Germany) on 1 or both dental arches using 34.5% phosphoric acid gel (Vococid; Voco, Cuxhaven, Germany) and light-cure adhesive (Transbond XT; 3M Unitek, Monrovia, Calif). During orthodontic treatment, rebonding procedure not performed, and the duration did not exceed 36 months. |
| No current or previous professional desensitizing treatment and use of over-the-counter desensitizing products within the previous 6 weeks |
| No long-term use of anti-inflammatory, analgesic, and psychotropic drugs |
| No pregnancy or breast-feeding |
| No recorded eating disorders or excessive dietary or environmental exposure to acid |
| No history of systemic conditions that may cause dentin hypersensitivity (eg, chronic acid regurgitation) |
| Selection criteria for teeth |
| Primary teeth selection criteria |
| Maxillary and mandibular premolars have intact buccal enamel with no white spots, signs of hypoplasia or erosion, or wedge-shaped defects |
| No pretreatment with chemical agents (ie, hydrogen peroxide) |
| No previous orthodontic, endodontic, or restorative treatment |
| No signs of gingival recession |
| Secondary teeth selection criterion |
| Enamel microcracks visible on the buccal enamel surface |
The sample size was estimated using the sample size calculator, by which a sample size of 60 was required to detect differences in sensitivity intensity values with a 5% confidence interval (CI) and 90% confidence level (population size, 76). Thus, 60 patients were included in the study; 30 received orthodontic treatment, and the rest served as the controls (untreated orthodontically).
The investigation was performed according to the protocol in Figure 1 . Metal brackets were removed with the conventional utility Weingart pliers (Dentaurum, Ispringen, Germany) by hand (the mesial and distal edges of the bracket wings were squeezed gently until the bracket became free), and all visible residual adhesive was carefully removed using a slow-speed hand piece and a carbide finishing bur. The orthodontic archwire was removed before the debonding procedure. The teeth were kept out of occlusion (biting on a cotton roll) during the debonding. In the weeks before the removal of the brackets, the patients were in a passive phase of orthodontic treatment (no activation procedures, such as changes of archwires, ligatures, or elastics, were performed; and the same dietary instructions, such as avoidance of solid, extremely hot or cold foods, were given). After debonding, orthodontically treated patients were divided into 2 groups of 15: group 1, patients possessing teeth with visible EMCs (mean age, 18.5 ± 3.62 years; average time of treatment, 21.2 months); and group 2, patients whose teeth were free of visible EMCs (mean age, 17.1 ± 3.72 years; average time of treatment, 20.8 months). The identification of EMCs with a naked eye under normal room illumination was the main criterion for assigning patients to 1 of the 2 groups. The distribution of EMCs among premolars included in the study is shown in Table III .
| Distribution of visible enamel microcracks (%) | Experimental group 1 (n = 15) | Control group 1 (n = 15) |
|---|---|---|
| <25 | – | – |
| 25-50 | 12 (80%) | 9 (60%) |
| >50 | 3 (20%) | 6 (40%) |
Thirty orthodontically untreated patients were divided into 2 groups of 15: control group 1, patients having teeth with EMCs (mean age, 18.9 ± 3.15 years); and control group 2, subjects without EMCs (mean age, 18.0 ± 3.76 years). The protocol of the study was approved by the ethical review board of the Institute of Odontology at Vilnius University in Vilnius, Lithuania.
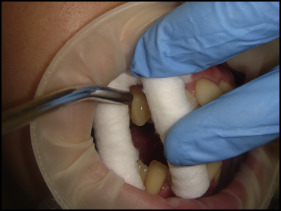
Tooth sensitivity was assessed by timed applications of compressed air (applied first) and cold stimulus (freshly melted ice water). Thermal stimuli (compressed air and cold) application procedures are shown in Table IV and Figure 2 . The intensity of the evoked discomfort or sensitivity by compressed air and cold stimulus was measured using a 100-mm long visual analog scale (VAS). The sensitivity assessment was performed at 5 time points: just before debonding (baseline), immediately after debonding, and at 1, 3, and 7 days after debonding. The VAS scores of each patient (all evaluated teeth) from every group (experimental and control) were averaged to determine the mean sensitivity intensity score at each time point. Orthodontically treated patients were blinded to the presence of visible EMCs and so were the subjects in the control groups. After enrollment in the investigation, the patients were given identical oral hygiene instructions. No desensitizing agents were permitted, including dentifrices and analgesics, during the study period.
| Compressed air | Cold | |
|---|---|---|
| Temperature (°C) | 19-24 | 0 |
| Application procedure | Compressed air from a dental syringe at a right angle to the buccal cervical surface of the tooth and a distance of about 1-3 mm | With a pipette or syringe, application of freshly melted water on the cervical third of the buccal tooth surface |
| Application duration (s) | 1 | ≤3 |
| Application procedure characteristics | The buccal cervical surfaces of the premolars were selected as sites for the sensitivity evaluation | |
| Application procedure was performed for every tooth individually starting from the maxillary right second premolar and finishing with the mandibular right second premolar (maximum of 8 teeth per patient) | ||
| Before testing, the patients were asked to raise their right hand at the moment sensation was first felt, at which time the application of the stimulus was discontinued | ||
| Ten minutes of recovery was provided between stimulus applications for each tooth | ||
Statistical analysis
Statistical analysis was carried out using the SPSS statistical package (version 17.0; SPSS, Chicago, Ill). A paired-samples t test was performed to evaluate differences between tooth sensitivity values at the different time points of the same group. An independent-samples t test was applied to compare mean sensitivity intensity values between the 2 unrelated groups on the same continuous, dependent variable. Spearman correlation coefficients of the baseline tooth sensitivity values and their changes during debonding were calculated. For graphic representation, error bars were used in which the 95% confidence interval was demonstrated. In case of overlapping confidence intervals, there was no statistical difference between the 2 comparison groups. In other cases with no overlapping confidence intervals, the statistical significance of the differences was evident with 95% probability. Significance for all statistical tests was predetermined to be P ≤0.05.
Results
Visibility of the EMCs was assessed repeatedly by the same investigator (I.D.) 1, 3, and 7 days after debonding before the tooth sensitivity assessment procedures. For standardization, the same time of the day, tooth position (patient’s head position), and its isolation conditions were used. After repeated EMC visibility evaluations, no significant discrepancies between the results were observed.
Each patient was assessed by 1 operator (I.D.) during the entire study. The pain levels from the VAS scale were measured using a digital caliper by the same examiner. Measurement error was analyzed using the method suggested by Dahlberg. Measurements (sensitivity intensity values from the VAS scale) of 5 patients were repeated 2 weeks later, and the estimated error between measurements was calculated. Measurement error was found to be 0.12 mm, and the effect of this error on the reliability of the sensitivity intensity measurements was deemed nonsignificant.
The mean sensitivity intensity values compared with the stimulus applied and time for the patients having teeth with visible EMCs are shown in Table V . The sensitivity was greatest immediately after debonding followed by a decrease on day 1 with higher mean sensitivity intensity values for the cold stimulation group. Tooth sensitivity elicited by cold testing demonstrated gradual changes (corresponding to straight lines in Fig 3 ); at day 7, the recorded discomfort values were lower than just before debonding ( P ≤0.05) and immediately after debonding ( P ≤0.05). Differences in the control group were nonsignificant. The reductions in sensitivity intensity scores were 33% or greater at day 3 (41.98%, air stimulation; 37.78%, cold stimulation) and at day 7 (85.71%, air stimulation; 37.04%, cold stimulation). After debonding, 1 tooth (2.38%) became sensitive to air stimulation, and 2 teeth (4.76%) became sensitive to cold stimulation.



