Diagnosis and Management of TMDs
| Key Points |
 Temporomandibular disorders (TMDs) are often remitting, self-limiting, or fluctuating over time and rarely result in disabling conditions. Temporomandibular disorders (TMDs) are often remitting, self-limiting, or fluctuating over time and rarely result in disabling conditions. |
 There is no single cause for all TMDs, and the influence of dental occlusion on TMDs is relatively low. There is no single cause for all TMDs, and the influence of dental occlusion on TMDs is relatively low. |
 The clinician must be cautioned against providing unnecessary treatments for subclinical TMDs, because benign TMD signs and symptoms are very common among the general population. The clinician must be cautioned against providing unnecessary treatments for subclinical TMDs, because benign TMD signs and symptoms are very common among the general population. |
 Between 3.6% and 7.0% of the general population has a TMD severe enough to seek treatment. Between 3.6% and 7.0% of the general population has a TMD severe enough to seek treatment. |
 Patients with pain-free temporomandibular joint (TMJ) clicking generally do not need treatment; reassurance and education about this benign condition usually suffice. Patients with pain-free temporomandibular joint (TMJ) clicking generally do not need treatment; reassurance and education about this benign condition usually suffice. |
 If jaw pain does not increase with jaw function, it is probably not a TMD. If jaw pain does not increase with jaw function, it is probably not a TMD. |
 Management of TMDs should include the control of contributing factors, such as oral parafunctional habits. Management of TMDs should include the control of contributing factors, such as oral parafunctional habits. |
 The use of nicotine and the presence of sleep disorders, anxiety, and depression may influence TMD symptoms and prognosis; hence, these issues should be addressed. The use of nicotine and the presence of sleep disorders, anxiety, and depression may influence TMD symptoms and prognosis; hence, these issues should be addressed. |
 Management of TMDs may include self-management instructions, oral appliances, pharmacotherapy, and physical therapy. Management of TMDs may include self-management instructions, oral appliances, pharmacotherapy, and physical therapy. |
 Generally, surgical management for TMDs is indicated only after reasonable nonsurgical efforts have failed and when the patient’s quality of life is significantly affected. Generally, surgical management for TMDs is indicated only after reasonable nonsurgical efforts have failed and when the patient’s quality of life is significantly affected. |
 Radiographically visible condylar changes should not be used as a guide for treatment. Radiographically visible condylar changes should not be used as a guide for treatment. |
Anatomy of the Masticatory Structures
Craniomandibular articulation occurs in the TMJs, two of the most complex joints in the body. Each TMJ provides for hinging (rotation) movements in one plane, which is a criterion for a ginglymoid joint. At the same time, however, the TMJ provides for gliding (translation) movements, which is a criterion for an arthrodial joint. Thus, the TMJ is technically considered a ginglymoarthrodial joint.1 The TMJ is formed by the mandibular condyle fitting into the mandibular (glenoid) fossa of the temporal bone1 (Fig 8-1). Separating these two bones from direct contact is the interposed articular disc (sometimes inappropriately referred to as meniscus). The articular portion of the healthy disc is composed of dense, fibrous connective tissue, devoid of any nerves or vessels; conversely, the posterior attachment of the disc is richly vascularized and innervated.2–4 Collateral ligaments attach the disc to the condyle both medially and laterally. These ligaments permit rotational movement of the disc on the condyle during opening and closing of the mouth. This so-called condyle-disc complex translates out of the fossa during extended mouth opening1 (Fig 8-2). Therefore, in the normal joint, rotational movement occurs between the condyle and the inferior surface of the disc during early opening (the inferior joint space), and translation takes place in the space between the superior surface of the disc and the fossa (the superior joint space) during later opening. Movement of the joint is lubricated by synovial fluid, which also acts as a medium for transporting nutrients to and waste products from the articular surfaces.
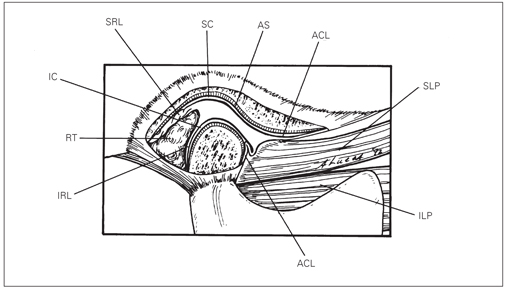
Fig 8-1 Normal anatomy of the TMJ. RT—retrodiscal tissues; SRL—superior retrodiscal lamina (elastic); IRL—inferior retrodiscal lamina (collagenous); ACL—anterior capsular ligament (collagenous); SLP and ILP—superior and inferior lateral pterygoid muscles; AS—articular surface; SC and IC—superior and inferior joint cavities; DL—discal (collateral) ligament. (Reproduced with permission from Okeson.1)
Fig 8-2 Normal functional movement of the condyle and disc during the full range of opening and closing. Note that the disc is rotated posteriorly on the condyle as the condyle is translated out of the fossa. The closing movement is the exact opposite of opening. (Reproduced with permission from Okeson.1)
Unlike most synovial joints, the articulating surfaces of the TMJs are lined with dense fibrocartilage instead of hyaline cartilage.5 This is an important feature, because fibrocartilage has a greater ability to repair itself than hyaline cartilage. This implies that the management of arthritic conditions of the TMJ may be different from that of other synovial joints.6
Movement of the TMJs is achieved by a group of skeletal muscles referred to as the muscles of mastication. These muscles are comparable to other skeletal muscles in physiology and ergonomics.7 Although the muscles of mastication are the primary muscles that provide mandibular movement, other associated muscles of the head and neck furnish secondary support during mastication. The masticatory muscles include the masseter, medial pterygoid, and temporal muscles, which predominantly elevate the mandible (mouth closing); the digastric muscles, which assist in mandibular depression (mouth opening); the inferior lateral pterygoid muscles, which assist in protrusive and lateral movements of the mandible; and the superior lateral pterygoid muscles, which provide stabilization for the condyle and disc during function.8–11 The masticatory muscles are recruited in a variety of functional behaviors that include talking, chewing, and swallowing.12 A number of muscle behaviors are nonfunctional (parafunctional), defined under the broad term of bruxism, and include grinding, clenching, or rhythmic, chewing-like, empty-mouth movements.13,14
TMDs encompass a group of musculoskeletal and neuromuscular conditions that involve the TMJs, the masticatory muscles, and all associated tissues.15 TMDs have been identified as a major cause of nondental pain in the orofacial region and are considered to be a subclassification of musculoskeletal disorders.16 TMDs represent clusters of related disorders in the masticatory system that have many common symptoms. The most frequent presenting symptom is pain, usually localized in the muscles of mastication and/or the preauricular area. Chewing or other mandibular activity usually aggravates the pain. In addition to complaints of pain, patients with these disorders frequently have a limited range of mandibular movement and TMJ sounds most frequently described as “clicking,” “popping,” “grating,” or “crepitus.”
Common patient complaints include jaw ache, earache, headache, and facial pain. Nonpainful masticatory muscle hypertrophy and abnormal occlusal wear associated with oral parafunction, such as bruxism (jaw clenching and tooth grinding), may be related problems. Pain or dysfunction due to nonmusculoskeletal causes, such as otolaryngologic, neurologic, vascular, neoplastic, or infectious disease, in the orofacial region is not considered a primary TMD even though musculoskeletal pain may be present. However, TMDs often coexist with other craniofacial and orofacial pain disorders.
Reports on prevalence of TMDs from cross-sectional epidemiologic studies vary considerably from study to study because of differences in descriptive terminology, data collection, analytic approaches (eg, single-factor versus multiple-factor analysis), and the individual factors selected for study. A recent systematic review including only studies adopting the Research Diagnostic Criteria for TMD (RDC/ TMD)17 reported a prevalence of up to 13% for masticatory muscle pain, up to 16% for disc derangement disorders, and up to 9% for TMJ pain disorders in the general population.18 While the prevalence of the different diagnoses in TMD patients varied widely, the results of a meta-analysis showed a prevalence of 45%, 41%, and 34% for muscle disorders, disc derangement disorders, and joint pain disorders, respectively.18 Other studies show that TMDs are primarily a condition of young and middle-aged adults rather than of children or the elderly and are approximately twice more common in women than in men.19,20 In addition, TMDs are often remitting, self-limiting, or fluctuating over time.21,22 The progression to a potentially more serious nonreducing disc status is relatively uncommon,23–26 and there is evidence suggesting that progression to chronic and disabling intra-capsular TMJ disease is uncommon.22,26,27
Only 3.6% to 7.0% of individuals with TMDs are estimated to be in need of treatment,28–33 and the annual incidence rate is estimated to be 2.0%.29,34,35 Because joint sounds are common, often pain free, and not progressive, it is important to avoid overtreatment of benign chronic reducing and nonreducing disc displacements in the absence of pain and/ or impaired function.26 Magnetic resonance imaging (MRI) indicates that up to 35% of asymptomatic individuals appear to have disc displacements.36
Painful TMD conditions, such as myofascial pain and arthralgia, have been associated with trauma, parafunction, physical symptoms (somatization), and the female sex.37 Also, individuals developing TMDs are more likely to describe comorbidities such as headaches, muscle soreness, and other body pains.38,39 Cigarette smoking was associated with increased risk of TMDs in young adults40 and higher levels of pain, psychosocial distress, and sleep disturbances in TMD patients.41
The identification of an unambiguous, universal cause of TMDs is lacking. For this reason, most of the factors discussed in this section are not proven causes but are rather associated with TMDs. Factors that cause the onset of TMDs are called initiating factors, factors that increase the risk of TMDs are called predisposing factors, and factors that interfere with healing or enhance the progression of TMDs are called perpetuating factors. Individual factors, under different circumstances, may serve any or all of these roles. There is not a single etiologic factor or a unique theoretical model that can explain the onset of TMDs.
Bone and TMJ soft tissue remodeling and muscle tone regulation are all adaptive physiologic responses to insult or change. Loss of structural integrity, altered function, or biomechanical stresses in the system can compromise adaptability and increase the likelihood of dysfunction or pathology.42 Direct extrinsic trauma to any component of the masticatory system can initiate loss of structural integrity and concomitantly alter function, thereby reducing the adaptive capacity in the system. In addition, there are other contributing anatomical, systemic, pathophysiologic, and psychosocial factors that may sufficiently reduce the adaptive capacity of the masticatory system and result in TMDs.
Trauma
Trauma is described as any force applied to the masticatory structures that exceeds that of normal functional loading. Most trauma can be divided into three types: (1) direct trauma, which results from a sudden and usually isolated blow to the structures, (2) indirect trauma, which is associated with a sudden blow but without direct contact to the affected structures, and (3) microtrauma, which is the result of prolonged, repeated force over time.
Direct trauma
There is general agreement that direct trauma (macrotrauma) to the mandible or the TMJ produces injury via impact and is accompanied in close temporal proximity with signs and symptoms of inflammation. If the forces lead to structural failure, loss of function may quickly follow. Direct trauma resulting in mandibular fracture in adults has been associated with increased signs and symptoms of TMDs; however, these may not cause patients to seek treatment.43 In children, condylar and subcondylar fractures were associated with increased TMD symptoms over time in girls but not in boys.44 More significant fractures may result in disc displacement.45
Patients with TMDs report physical trauma more often than patients without TMDs.46 The accuracy of recall of TMD symptoms associated with a traumatic event may be compromised,47 making it difficult to clearly link the traumatic event with the symptom onset. Other forms of trauma, such as wide or prolonged opening,48 third molar extraction,49 and intubation, have been reportedly associated with TMDs. Self-reported jaw injury due to yawning or prolonged opening is significantly higher in patients with TMDs than in healthy patients.46 Transient and permanent dysfunction of the TMJ after upper airway management procedures has been reported50,51; however, controlled trials and longitudinal studies are not available in the English-language literature.
Indirect trauma
Acceleration-deceleration (flexion-extension) injury (whiplash) with no direct blow to the face may cause symptoms consistent with TMDs, but significant controversy persists as to whether there could be a direct causal relationship. Prospective controlled studies link52 and show limited risk53 for the development of TMD symptoms after whiplash. Review of the literature has highlighted these controversies.54 However, there is some evidence that postwhiplash TMDs can have a different and potentially more protracted clinical course than non-trauma-associated TMDs.55 Although symptoms in the mandible may be referred from injured cervical structures, a direct causal relationship between mandibular symptoms and indirect trauma has yet to be established.53,56,57
Computer simulation suggests that low-velocity rear-end impact in motor vehicle crashes does not cause mandibular movement or stresses beyond physiologic range.58 In support of this finding, human volunteers in simulated rear-end crash tests failed to demonstrate mandibular movement beyond physiologic limits.59 Thus, while evidence is lacking for a mandibular strain without a direct blow to the mandible following a low-velocity motor vehicle accident, there are recognized pathways of heterotopic pain from the cervical area to the trigeminal area.16,60 It is therefore not uncommon to observe symptoms of TMDs following acceleration-deceleration injury to the neck without direct trauma to the face or jaw. The etiologic significance of nonimpact injuries is uncertain, and much misinformation is being provided to patients without scientific studies to support the claims.
Microtrauma
Microtrauma has been hypothesized to originate from sustained and repetitious adverse loading of the masticatory system through postural imbalances or from parafunctional habits. It has been suggested that postural habits such as forward head position or phone-bracing may create muscle and joint strain and lead to musculoskeletal pain, including headache, in the TMD patient.61
Parafunctional habits have been most frequently assessed by indirect means, such as self-report, questionnaires, reports by a bedroom partner, or tooth wear. These indirect measures of parafunctional habits have provided conflicting reports as to the relationship between TMD symptoms and the presence of parafunctional habits. When myofascial TMD patients were compared with controls using polysomnography, the patients self-reported more sleep bruxism than the controls; however, the polysomnography did not confirm these results.62
Parafunctional habits such as teeth clenching, teeth grinding, lip biting, and abnormal posturing of the mandible are common and usually do not result in TMD symptoms.63–65 However, parafunctional habits have been suggested as initiating or perpetuating factors in certain subgroups of TMD patients.14,65–75 Although the available research and clinical observations generally support this contention, the exact role of parafunctional habits in TMDs remains unclear, because few studies have directly assessed these behaviors. Attrition severity secondary to bruxism cannot distinguish TMD patients from asymptomatic subjects,76,77 and muscle hyperactivity has not been shown to be associated with arthrogenous TMJ disorders.78 Furthermore, clenching does not cause neuromuscular fatigue, because muscles compensate for sustained muscle activity by derecruitment of motor neurons or through slower firing rates.79
Despite the lack of evidence that non-experimentally induced parafunction or clenching can cause TMDs,80,81 some studies have shown that experimentally induced parafunction can result in transient pain similar to that reported by patients with TMDs.72,82 However, the impact of these studies is limited by their small sample size.
The intensity and frequency of oral parafunctional activity may be exacerbated by stress and anxiety, sleep disorders, and medications (eg, neuroleptics, alcohol, and other substances),14 although the relationship between sleep bruxism and psychologic factors has been questioned.83,84 Some forms of masticatory muscle hyperactivity have been associated with emotional behavior and may be mediated via the cortex through the hypothalamus.85 Intense and persistent parafunction can also occur in patients with neurologic disorders such as cerebral palsy and extrapyramidal disorders such as orofacial dyskinesia and epilepsy.86 Conversely, sleep bruxism has not been related to facial type or head form.87
The most commonly believed indication of past sleep bruxism severity is dental attrition.88 However, dental attrition can also be partly explained by overbite and overjet changes that correlate with age89 and sex,90,91 protrusive guidance schemes,92,93 dentofacial morphology,94 erosive diets,95 the bite force ability,93,96 and environmental factors.97,98 In addition, anthropologists argue that if bruxism, which is nearly universal in humans, is pathologic, natural selection should have eliminated it by now.99 Others have also suggested beneficial consequences of bruxism, by allowing better chewing efficiency from flattened occlusal surfaces.100
Whether sleep bruxism is a pathogenic disorder or a normal physiologic process is unclear. Recent evidence suggests that sleep bruxism may be associated with increased salivation during sleep, resulting in lubrication of oropharyngeal structures,101 increased space in the upper airways to aid with airway patency,102,103 or both.104 Furthermore, it has been suggested that sleep bruxism plays a protective role as a compensatory mechanism to protect the airway in patients with sleep-related breathing disorders.105 Therefore, the issue of whether sleep bruxism without the presentation of significant problems requires management is debatable. A more pragmatic approach may be to view this as an issue regarding “consequence management,” taking into account risk or side effect–benefit ratio.106
Another problem arises when attrition is used to suggest current bruxism levels.68 Attrition appears to be episodic in nature and occurs in bursts due to as yet unspecified factors,24,107 and thus any noted attrition may not necessarily represent ongoing habits. Continued research with more direct measurements of parafunction (eg, portable electromyography, sleep laboratory, and direct observation) will be necessary to clarify the specific role of current parafunction.108–110
Anatomical factors
Skeletal relationships
Skeletal factors comprise adverse biomechanical relationships that can be genetic, developmental, or iatrogenic in origin. Severe skeletal malformations, interarch and intra-arch discrepancies, and past injuries to the teeth may play a role in TMDs. This role, however, may be less strong than previously believed. For example, while it is known that disc displacement is common in children with facial skeletal abnormalities, such as retrognathia,111 it cannot be said that these anatomical anomalies are etiologic. In addition, patients with a disc displacement as well as other types of TMDs generally do not have an increased prevalence of forward head posture.112
A steep articular eminence has also been proposed as an etiologic factor in internal derangement of the TMJ. In asymptomatic patients, a steeper eminence was associated with an increased posterior rotation of the disc, posing a potential anatomical risk factor.113 However, several studies have shown that the eminence was less steep in TMJs with disc displacement without reduction and TMJs with osseous changes compared with TMJs with disc displacement with reduction or TMJs without osseous changes, indicating adaptive remodeling.114–116 In addition, unilateral joint sounds were associated with the side with the less steep condylar movement path.117
Occlusal relationships
The dental profession historically has viewed occlusal variation as a primary etiologic factor for TMDs. Occlusal features such as working and nonworking posterior contacts and discrepancies between the retruded contact position (RCP) and the intercuspal position (ICP) have been commonly identified as predisposing, initiating, and perpetuating factors. However, the current available evidence suggests that the influence of the occlusion on the onset and development of TMDs is low.118,119 Among the occlusal factors evaluated, loss of posterior support and unilateral crossbite show some association across studies.118
Several occlusal factors, such as large overjet, minimal anterior overlap and anterior skeletal open bite, unilateral posterior crossbite, occlusal slides greater than 2 mm, and lack of firm posterior tooth contact, are more prevalent in TMD patients than in convenience samples, possibly due to condylar positional changes following intracapsular alterations associated with the disease process itself. Therefore, these occlusal factors may be the result rather than the cause of the disease.118,120,121 Previous studies have lacked reliable occlusal measurement techniques and data collection methods, which may explain the variability of some findings. Nevertheless, whether these findings are considered individually or simultaneously, little evidence is available to strongly associate occlusal and other factors that are traditionally implicated in TMD etiology.
Some specific occlusal variants explain between 10% and 25% of specific diagnoses.122 A small increased risk for osteoarthritic changes122,123 and myofascial pain122,124 is associated with RCP-ICP slides over 2 mm, internal derangements with unilateral maxillary palatal crossbite, osteoarthritic changes and myofascial pain with overjet over 6 mm, and internal derangement and osteoarthritic changes with more than six missing posterior teeth.122 The greatest contribution was found for anterior open bite to define osteoarthritic changes and myofascial pain patients. However, most of the associations noted were judged to be secondary to joint alterations and therefore not etiologic. In summary, the contribution of occlusion in the etiology of TMDs appears to be minimal.125
Pathophysiologic factors
Systemic factors
Systemic pathophysiologic conditions may influence local TMDs and should generally be managed in cooperation with the patient’s primary care physician or other medical specialist. These can include degenerative, endocrine, infectious, metabolic, neoplastic, neurologic, rheumatologic, and vascular disorders. Systemic factors can act simultaneously at central and local (peripheral) levels.126,127
Generalized joint laxity (hypermobility) has been cited as a possible contributing factor to TMDs128,129 and has been proven to be significantly more prevalent in patients with internal derangements than in patients with other types of TMDs or in normal controls.24,130–133 Altered collagen metabolism may also play a role in joint laxity,131 and the collagen composition in TMJs with painful disc displacement has been found to differ from that of asymptomatic joints.134 Nevertheless, there is only a weak correlation between the mobility of peripheral joints or the trunk and mandibular mobility,135–137 and research has yet to demonstrate that joint laxity can predict the potential for development of TMDs.
Local (peripheral) factors
Local pathophysiologic factors of TMDs, such as masticatory efficiency, appear to be multifactorial and involve such a large span of individual variation that it is difficult to establish norms.138 While chewing efficiency is not affected by the extent of the occlusal contact area100,139 or the number and extensiveness of restorations,138 it is enhanced by greater numbers of chewing units and fewer than five missing posterior teeth.140 The threshold for impaired chewing is at fewer than three posterior chewing units.141 In addition, chewing force is also influenced by sex,142,143 age,144 and pain levels.145–149
Masticatory muscle tenderness is not always related to variation in muscle activity124,127 or the site or side of reported tenderness.150,151 While the masseter muscle may react to proximal muscle pain, the anterior temporalis muscle does not, and any associations may be parallel developments rather than etiologic. Muscle tenderness does not appear to be the result of inflammation but is probably related to prolonged central hyperexcitability and altered central nervous system processing following peripheral tissue injury.151 Cervical muscle activity has been shown to influence masticatory muscle activity,152,153 probably involving a primary afferent reflex response.154 Thus, a primary cervical or TMJ disorder may precipitate a secondary masticatory muscle condition. Muscle hyperalgesia can also result from TMJ inflammation.155
Of great concern to the clinician is the distinction between pathologic and adaptive responses to disease in the TMJ. Histologic studies suggest that cartilage thickness and composition adapt to shearing stresses during functional loading.156–159 Maintenance of an intact articular surface is to be expected, even in the face of osteoarthritic changes,121,160,161 allowing for both stable morphologic relationships and histologic compatibility between the articulating components. Morphologic change, therefore, while mostly irreversible, usually achieves and maintains stability and should be considered adaptive.162 The goal of treating osteoarthritic changes in this light should not be to restore earlier morphology but to encourage the body’s adaptive response to pathophysiologic processes.
In the early stages of disc derangement, signs of osteoarthritis are not apparent. Disc derangements with reduction may persist for many years without development of radiographically visible changes or symptoms.163 Disc derangements in later stages with osteoarthritic changes are possibly parallel but may also represent independent processes,164 and 50% will show some active cellular osteoblastic or osteoclastic activity.165 Osteoarthritic changes, alterations in synovial fluid viscosity, and inadequate or altered lubrication may initiate derangement of the TMJ articular disc.166,167 Synovial fluid analyses attempting to correlate biochemical signs of inflammation with pain reveal abnormal concentrations of plasma proteins168–170 or neurotransmitters and inflammatory cytokines.171–173 Other studies have evaluated the degradation of various enzymes and other metabolic byproducts as well as the type of pain transmitters causing pain, inflammation, and degeneration in the TMJ.174–177
Frictional “sticking” of the disc has been proposed73 to cause TMJ internal derangement. The forces depend on the type of clenching task, with greater impact at the ICP and during a unilateral molar clench.178,179 According to experimental models and animal studies, the forces are also increased by reduced congruity between the opposing surfaces,180–183 and by flat unrounded surfaces,179 and are affected by disc thickness and area182 as well as by mandibular deformation during clenching.184 Interestingly, nonworking tooth contacts have been hypothesized to reduce loads within the joint and act as a stress breaker for the clenching forces.179
Intracapsular pressure may also affect TMDs.185,186 With joint movement, the alternating pressure acts as a pump for joint lubrication, nutrition, blood supply, drug delivery, metabolic waste removal, and even condylar growth. Thus, any interruption through immobilization or prolonged clenching may initiate or advance TMD signs and symptoms.
Female hormones have been mentioned as having a role in TMJ disc disease, but the question is as yet undecided because the presence of both estrogen and progesterone receptors within the articular disc has been both confirmed187 and denied.188 Randomized controlled trials (RCTs) indicate that estrogen does not play a role in the etiology of TMDs,189,190 whereas cohort studies and case-control studies show the opposite.191,192
The etiologic explanations for progression from disc displacement to osteoarthritis and osteoarthritic changes are multifactorial and include failure of the reparative articular chondrocyte response, due to metabolic dysfunction, and relative or absolute overloading, due to excessive mechanical forces, leading to articular cartilage biochemical failure.42,193 Remodeling, in contrast, is a physiologic response to accommodate an altered disc position.121 Thus, it is probable that a mechanical breakdown in the articular disc, such as a perforation, rather than an unusual disc position leads to osteoarthritis and/or osteoarthritic changes following disc displacement.194 It is not certain, however, that all gross abnormalities of disc morphology will lead to osteoarthritis, because the TMJ may be capable of healing disc perforations within a relatively short time according to one experimental animal study.195
Mechanical stress may also lead to the accumulation of damaging free radicals in affected articular tissues of susceptible individuals. This condition is called oxidative stress.196 Dijkgraaf et al197 have proposed that free radicals may be responsible for the formation of adhesions in the TMJ by cross-linking of proteins.
Genetic factors
Little research is available with regard to genetic susceptibility for TMDs. A recent study examined the relationship between catechol-O-methyl transferase (COMT) polymorphism, pain sensitivity, and the risk of TMD development. Three genetic variants (haplotypes) of the gene encoding COMT were identified and designated as “low pain sensitivity,” “average pain sensitivity,” and “high pain sensitivity.”198 The haplotypes were associated with experimental pain sensitivity, and the presence of even a single “low pain sensitivity” haplotype was shown to reduce the risk of developing myogenous TMDs. Further studies are needed to identify how the presence of the different haplotypes relates to the risk of developing TMDs. Moreover, the Orofacial Pain: Prospective Evaluation and Risk Assessment (OPPERA) Study has genotyped 3,295 single nucleotide polymorphisms representing 358 genes involved in biologic systems associated with pain perception; this is the most extensive panel of candidate genes, strongly indicating that multiple genetic and biologic pathways contribute to the risk for TMDs.199
Psychosocial factors
Psychosocial factors include individual, interpersonal, and situational variables that impact the patient’s capacity to function adaptively. General distress is the most salient single factor across most individuals with chronic TMD pain.200 General distress is readily assessed (see chapters 2 and 12), and certainly other factors may contribute actively to the measured distress, such as personality characteristics, enduring stressors, a physical response to stress, or limited coping skills.201–206
There is evidence that some TMD patients experience more anxiety than healthy control groups and that some TMD and orofacial pain symptoms may be only one of several somatic manifestations of emotional distress.207–211 Some muscle pain, in fact, may be caused by excessive sympathetic nervous system activity as an overresponse to life stressors, and the attention focused on the pain can adversely affect the intensity of the pain.212–215 Patients with such complaints often have a history of other stress-related disorders.211,216 Depression and anxiety related to other major life events may alter the patient’s perception and tolerance of physical symptoms, causing them to seek more care for what is presented as a problem of the body.217
Patients with chronic TMDs have been found to have psychosocial and behavioral characteristics similar to patients with lower back pain and headache.218,219 In general, TMD patients are not significantly different from healthy individuals in personality type, and they do not differ from other pain patients in responses to illness, attitudes toward health care, or ways of coping with stress.64,206,220–222 Any psychologic impairment may be merely associated with the persistence of pain.145,217,223
Environmental contingencies can greatly complicate treatment by affecting an individual’s perception of and response to pain and disease. Some patients may experience a lessening of distress to the extent that psychogenic symptoms decrease or resolve preexisting psychologic and interpersonal conflicts. This primary gain of symptom formation is to be distinguished from the secondary gain of social benefits experienced by patients once a disorder is established.224–226 Secondary gain includes being exempt from ordinary daily responsibilities, being compensated monetarily from insurance or litigation, using the rationalization of “being ill” to avoid unpleasant tasks, and gaining attention from family, friends, or health care workers.226,227 What the clinician interprets as “pain” in response to a clinical examination procedure designed to evoke pain may be less related to any local nociceptive mechanisms and more a function of pain behavior: how the individual presents his or her distress as motivated by factors beyond the stated complaint.
The OPPERA group, using a case-control analytical approach as a foundation for their research, found that TMD patients are different from controls across multiple phenotypic domains, including sociodemographic factors, clinical variables, psychologic functioning, pain sensitivity, autonomic responses, and genetic associations. All of these elements are providing cutting-edge information on the biologic pathways of TMD pain that may elucidate TMD pathophysiology.199
Diagnostic Classification of TMDs
The classification of TMDs is hampered by limited knowledge of the etiology and natural progression of these disorders, and yet advancement in our knowledge is dependent on an accepted taxonomy and corresponding diagnostic criteria.17 Diagnostic criteria allow comparisons of patient populations in different studies and provide a common language for developing a conceptual framework to use in the clinic.228 Any classification system or taxonomy must be considered an evolving framework that will be modified by new findings and increased levels of understanding. As part of the development of the Diagnostic Criteria for TMDs (DC/TMD),229 several international consensus workshops were conducted by the International RDC-TMD Consortium Network of the International Association for Dental Research (IADR) and the Orofacial Pain Special Interest Group of the International Association for the Study of Pain (IASP) in 2009 (Miami), 2011 (San Diego), and 2012 (Iguazu Falls, Brazil). At these workshops, the American Academy of Orofacial Pain (AAOP) TMD taxonomy and criteria230 were revised in order to create an expanded taxonomy for TMDs based on principles of clear diagnostic criteria that can be operationalized for standardized assessment (see Box 3-1). This allows the taxonomy to better serve both clinical practice and further research into these conditions. The expanded taxonomy was approved by the AAOP Council in 2012. The DC/TMD was developed in parallel for the more common TMDs. The expanded taxonomy, a description of its development, and an explanation with the rationale for changes made relative to the prior version230 will soon be available.231 The expanded taxonomy incorporates the diagnostic citeria for the common TMDs as fully described in Tables 1 and 2 of the DC/TMD. 229
It is possible that one set of diagnostic criteria may not satisfy all circumstances to which it might be applied.232 For example, the requirements for diagnostic sensitivity and specificity are often different for the clinical researcher performing a clinical trial and the clinician treating patients. The researcher needs inclusion criteria with high specificity at the expense of sensitivity, resulting in a homogenous test population with a high probability of having the disease in question. In contrast, the clinician needs to identify patients presenting with the entire spectrum of a particular disease. This will likely require higher sensitivity at the expense of reduced specificity. However, the clinician must be cautioned against unnecessary treatment of subclinical disease based on the presence of benign signs and symptoms that are very common in the general population. The criteria listed here were made available by the consensus work groups prior to publication in peer-reviewed journals to ensure consistency between this revised edition and future publications; however, revisions to the expanded DC/TMD may be present in the published versions.229,231 The following outlines and criteria are in part derived from the specific text in Tables 1 and 2 of the Schiffman et al paper,229 for the common TMDs, and from the full classification,231 for the uncommon TMDs.
In the following classification, TMDs are divided into TMJ disorders and masticatory muscle disorders, headache disorders, and associated structures. The International Classification of Diseases, Tenth and Ninth Editions (ICD-10 and ICD-9) codes, required for medical insurance, are given for each specific disorder.
TMJ disorders
1. Joint pain (ICD-10 M26.62; ICD-9 524.62)
Previously called synovitis, capsulitis, and ret-rodiscitis, joint pain is defined as an inflammation of the synovial lining of the TMJ that can be due to infection, an immunologic condition secondary to cartilage degeneration, or trauma. Joint pain is characterized by localized pain that is exacerbated by function and superior or posterior joint loading. On occasion, there will be a fluctuating swelling (due to effusion) that decreases the ability to occlude on the ipsilateral posterior teeth.
A. Arthralgia. Arthralgia is pain of joint origin effected by jaw movement, function, or parafunction and replication of this pain with provocation testing. Limitation of mandibular movements secondary to pain may be present.
In order to diagnose arthralgia, the history must be positive for both of the following:
• Pain in the jaw, temple, in front of the ear, or in the ear in the last 30 days, with examiner confirmation of pain in a masticatory structure
• Pain modified with jaw movement, function, or parafunction
In addition, examination of the TMJ must elicit a report of familiar pain with at least one of the following provocation tests:
• Palpation of the lateral pole or around the lateral pole
• Maximum unassisted or assisted opening, right or left lateral movements, or protrusive movements
B. Arthritis. Arthritis is pain of joint origin with clinical characteristics of inflammation or infection: edema, erythema, and/or increased temperature. It may arise in association with trauma. Limitation of mandibular movements secondary to pain may be present. Although not required for diagnosis, pain with a compression test can help to corroborate this diagnosis. Associated symptoms can include ipsilateral posterior open bite if an effusion is present. This disorder is also referred to as synovitis or capsulitis.
In order to diagnose arthritis, the patient must have arthralgia as defined above, and examination and laboratory testing must be positive for both of the following:
• Presence of edema, erythema, and/or increased temperature over the joint
• Negative serologic results for rheumatologic disease
When this diagnosis needs to be confirmed, laboratory testing will show elevated levels of interleukin 1β (IL-1β), interleukin 1 receptor antagonist (IL-1Ra), IL-6, tumor necrosis factor (TNF), serotonin, or glutamate or reduced levels of soluble IL-1 receptor II (IL-1sRII) or soluble TNF receptor II (TNFsRII) in the synovial fluid.
2. Joint disorders
Articular disc displacement is the most common TMJ arthropathy and is characterized by several stages of clinical dysfunction that involve the condyle-disc relationship. It involves an abnormal relationship or misalignment of the articular disc relative to the condyle. Although posterior233,234 and mediolateral235–238 displacements of the articular disc have been described, the usual direction for displacement is anteriorly or anteromedially.239,240 Nevertheless, pain or mandibular movement symptoms are not specific for disc derangement disorders,241 and the disc position is not related to any presenting symptoms.242
The causes of disc displacement are not established; however, it is postulated that in the majority of cases, elongated or torn ligaments binding the disc to the condyle permit the disc to become displaced.243 Lubrication impairment is also suggested to be a possible etiologic factor of disc displacement.73,167 Osteoarthritis may also precipitate disc displacement. Disc displacement is subdivided into disc displacement with reduction and disc displacement without reduction.
A. Disc-condyle complex disorders (ICD-10 M26.62; ICD-9 524.63).
i. Disc displacement with reduction. In the closed-mouth position, the disc is in an anterior position relative to the condylar head, and the disc reduces upon opening of the mouth. Medial and lateral displacement of the disc may also be present. Clicking, popping, or snapping noises may occur with disc reduction. Although not required for this diagnosis, elimination of the opening and closing noise, if present, with protrusion can help to corroborate this diagnosis.
In order to diagnose disc displacement with reduction, the history must be positive for joint noise in the last 30 days. In addition, examination must confirm at least one of the following:
• Clicking, popping, and/or snapping noise detected during opening and closing with palpation during at least one of three repetitions of jaw opening and closing
• Clicking, popping, and/or snapping noise detected during opening or closing with palpation during at least one of three repetitions of jaw opening and closing; and clicking, popping, and/or snapping noise detected with palpation during at least one of three repetitions of left lateral, right lateral, or protrusion movements
When this diagnosis needs to be confirmed, MRI of the TMJ will reveal both of the following:
• In the maximal intercuspal position, the posterior band of the disc is located anterior to the 11:30 position and the condyle is not seated under the intermediate zone of the disc.
• On full opening, the intermediate zone of the disc is located between the condylar head and the articular eminence.
ii. Disc displacement with reduction with intermittent locking. In the closed-mouth position, the disc is in an anterior position relative to the condylar head, and the disc intermittently reduces with opening of the mouth. When the disc does not reduce, it is associated with limited mandibular opening. Medial and lateral displacement of the disc may also be present. Clicking, popping, or snapping noises may occur with disc reduction. Although not required for this diagnosis, occurrence of intermittent closed lock during the clinical examination can help to corroborate this diagnosis.
In order to diagnose disc displacement with reduction with intermittent locking, the history must be positive for both of the following:
• Joint noise in the last 30 days
• Intermittent locking with limited opening, even if momentary, in the last 30 days
In addition, the examination must confirm disc displacement with reduction as defined above.
When this diagnosis needs to be confirmed, the imaging criteria are the same as those for disc displacement with reduction.
iii. Disc displacement without reduction with limited opening. In the closed-mouth position, the disc is in an anterior position relative to the condylar head, and the disc does not reduce with opening of the mouth. Medial and lateral displacement of the disc may also be present. This disorder is associated with limited mandibular opening. Although not required for this diagnosis, the presence of deflection on opening to the affected side and limited lateral movements especially to the contralateral side help to corroborate this diagnosis. Note that limitation of movement cannot be reliably determined from imaging; subjects may not open maximally because of the need to sustain mouth opening during some imaging procedures. This disorder is also called closed lock.
In order to diagnose disc displacement without reduction with limited opening, the history must be positive for both of the following:
• Jaw lock or catch so that the mouth will not open all the way
• Limitation in jaw opening severe enough to interfere with the ability to eat
In addition, the examination must confirm that the maximum assisted opening (passive stretch) is less than 40 mm, including the vertical incisal overlap.
When this diagnosis needs to be confirmed, MRI of the TMJ will reveal both of the following:
• In the maximal intercuspal position, the posterior band of the disc is located anterior to the 11:30 position and the intermediate zone of the disc is located anterior to the condylar head.
• On full opening, the intermediate zone of the disc is located anterior to the condylar head.
iv. Disc displacement without reduction without limited opening. In the closed-mouth position, the disc is in an anterior position relative to the condylar head, and the disc does not reduce with opening of the mouth. Medial and lateral displacement of the disc may also be present. This disorder is not associated with limited mandibular opening.
In order to diagnose disc displacement without reduction without limited opening, the history as defined for disc displacement without reduction with limited opening must be confirmed. In addition, examination must confirm that the maximum assisted opening (passive stretch) is at least 40 mm, including the vertical incisal overlap.
When this diagnosis needs to be confirmed, the imaging criteria are the same as those for disc displacement without reduction with limited opening.
B. Other hypomobility disorders (ICD-10 M26.61; ICD-9 524.61). Intra-articular fibrous adhesions and ankyloses are characterized by restricted mandibular movement with deflection to the affected side on opening that may result as a long-term sequela of trauma, including mandibular fracture. Hypomobility is firm and unyielding due to intra-articular fibrous adhesions, more widespread fibrotic changes in the capsular ligaments (fibrous ankylosis), and/ or, less frequently, the formation of a bony mass that results in fusion of the joint components (osseous ankylosis). The condition usually is not associated with pain. The most frequent cause of TMJ ankylosis is macrotrauma; less frequent causes are infection of the mastoid or middle ear, systemic disease, and inadequate surgical treatment of the condylar area.
i. Adhesions/adherence. Fibrous adhesions within the TMJ are thought to occur mainly in the superior compartment. They produce a decreased movement of the disc-condyle complex. Adhesions may occur secondary to joint inflammation that results from direct trauma or systemic conditions such as a polyarthritic disease. They may be associated with disc-condyle complex disorders.
In order to diagnose adhesions, the patient must have both of the following:
• History of loss of jaw mobility
• Positive diagnosis of a disc-complex disorder
In addition, the examination must confirm all of the following:
• Limited range of motion on opening
• Marked deflection to the affected side
• Markedly limited laterotrusion to the contralateral side
When this diagnosis needs to be confirmed, arthrography will demonstrate the presence of adhesions, or magnetic resonance arthrography will demonstrate either punctuate, bandlike lower signal intensity in the contrast material or irregularity of the contrast material–filled joint space or limited distensibility.
ii. Ankylosis. TMJ ankyloses are differentiated by the type of tissues causing the ankylosis (eg, fibrous or osseous). Fibrous ankylosis is more common. In fibrous ankylosis, no radiographic finding other than the absence of ipsilateral condylar translation on opening is found. Osseous ankylosis is characterized by radiographic evidence of bone proliferation with marked deflection to the affected side and markedly limited laterotrusion to the contralateral side.
In order to diagnose fibrous ankylosis, the patient must have a history of progressive loss of jaw mobility. In addition, the examination must confirm all of the following:
• Limited range of motion on opening
• Marked deflection to the affected side
• Markedly limited laterotrusion to the contralateral side
In addition, the imaging must show both of the following:
• Decreased to complete lack of ipsilateral condylar translation on opening
• A disc space between the ipsilateral condyle and the eminence
Note: In cases of bilateral involvement, asymmetries in mandibular movements during clinical examination will be less pronounced or absent.
In order to diagnose osseous ankylosis, the patient must have a history of progressive loss of jaw mobility. In addition, the examination must confirm limited jaw mobility with all movements, and the imaging must show bone proliferation with obliteration of part or all of the joint space.
C. Hypermobility disorders. Hypermobility disorders include two types of TMJ dislocations in which the disc-condyle complex is positioned anterior to the articular eminence and is unable to return to a closed position without a specific maneuver by the patient (ie, subluxation or partial dislocation) or by the clinician (ie, luxation or dislocation). The latter disorder is also referred to as open lock. Note that the condyle is frequently anterior to the eminence at full mouth opening and thus by itself is not a predictor of hypermobility disorders. The duration of dislocation may be momentary or prolonged. Pain may occur at the time of dislocation with residual pain following the episode.
Examples of hypermobility disorders are the following:
a. Closed dislocation (ICD-10 S03.0XXA; ICD-9 830.0)
b. Recurrent dislocation (ICD-10 M26.69; ICD-9 524.69)
c. Ligamentous laxity (ICD-10 M24.20; ICD-9 728.4)
i. Subluxation (ICD-10 S03.0XXA; ICD-9 830.0). Subluxation is a condition in which the disc-condyle complex is positioned anterior to the articular eminence and is unable to return to the fossa without a specific manipulative maneuver by the patient.
In order to diagnose subluxation, the patient must report both of the following:
• Inability to close from wide opening in the last 30 days
• Mouth closing can be achieved with a specific mandibular maneuver
Examination is required only when the disorder is present clinically. Examination will confirm the inability to return to a normal closed-mouth position without the patient’s performing a specific manipulative maneuver.
ii. Luxation (ICD-10 S03.0XXA; ICD-9 830.0). Luxation is a condition in which the disc-condyle complex is positioned anterior to the articular eminence and is unable to return to the fossa without a specific manipulative maneuver by the clinician.
In order to diagnose luxation, the patient must report the inability to close from wide opening, and examination must confirm both of the following:
• Mouth is wide open or in a protruded jaw position
• Mouth closing is achieved with manual manipulation by the clinician
Diagnosis can be made by history only when manipulation by a clinician was required in the past. When this diagnosis needs to be confirmed, imaging will show that the condyle is anterior to the articular eminence during an attempt to close the mouth.
3. Joint diseases
Osteoarthritis, also known as osteoarthrosis or degenerative joint disease (DJD), is defined as a degenerative condition of the joint characterized by deterioration and abrasion of articular tissue and concomitant remodeling of the underlying subchondral bone due to overloading of the remodeling mechanism. The progressive loss of articular cartilage in the osteoarthritic TMJ results from an imbalance between predominantly chondrocyte-controlled reparative and degradative processes.244 The process accelerates as proteoglycan depletion, collagen fiber network disintegration, and fatty degeneration weaken the functional capacity of the articular cartilage. Different kinds of biochemical markers have been determined in the synovial fluid of TMJs with osteoarthritis. These include IL-6,245 tissue inhibitor of metalloproteinase-1 (TIMP-1),245–247 matrix metalloproteinases,248,249 heat shock protein (HSP),250 transforming growth factor β1 (TGF-β1),251 bone morphogenetic protein 2 (BMP-2),252 chondroitin-4-sulfate (C4S) and chondroitin-6-sulfate (C6S),253 keratan sulfate (KS),254 and human leukocyte antigen D (HLA-DR).255 The clinical and diagnostic value of these markers remain to be evaluated, but similar markers have been present in other joint diseases. Radiographic evidence typically lags behind articular tissue changes.256 The early changes in the synovial membrane, such as synovial intima hyperplasia and cell hypertrophy with subsequent loss of fibrous material in the intima matrix,257 and in the articular cartilage are only detectable with biopsy and arthroscopy.193,258 Thus, osteoarthrosis frequently escapes early clinical detection.259
A. Degenerative joint disease (ICD-10 M19.91; ICD-9 715.18 localized/primary). DJD is a degenerative disorder involving the joint characterized by deterioration of articular tissue with concomitant osseous changes in the condyle and the articular eminence. It may be secondary to a systemic disorder. DJD without pain is classified as osteoarthrosis, while DJD with pain is classified as osteoarthritis.
i. Osteoarthrosis. In order to diagnose osteoarthrosis, the history must be positive for joint noise in the last 30 days. In addition, examination must confirm at least one of the following:
• Crepitus detected with palpation during maximum unassisted opening, maximum assisted opening, right or left lateral movements, or protrusive movements
• Patient report of crunching, grinding, or grating noises during the examination
Computerized tomography (CT) imaging of the TMJ will show at least one of the following:
• Subchondral cyst
• Erosion
• Generalized sclerosis
• Osteophyte
Laboratory testing will confirm negative serologic results for rheumatologic disease.
Note: Flattening and/or cortical sclerosis is considered an indeterminant finding for DJD and may represent normal variation, aging, remodeling, or a precursor to DJD. Also, DJD can result in malocclusions including anterior open bite, especially when present bilaterally, and contralateral posterior open bite when present unilaterally.
ii. Osteoarthritis. In order to diagnose osteoarthritis, the history must be positive for both of the following:
• Presence of joint pain as defined for arthralgia
• Presence of joint noise in the last 30 days
In addition, examination must confirm both of the following:
• Arthralgia as defined previously
• Same as those criteria defined for osteoarthrosis
Imaging will also show the same criteria as those defined for osteoarthrosis.
B. Condylysis (ICD-10 M26.69; ICD-9 524.69).
A rare idiopathic degenerative condition termed condylysis occurs spontaneously, primarily in adolescent girls,260–262 and is suggested clinically by anterior bite opening and a rapid development of molar laterotrusive facets.76 Normal condylar development proceeds until the sudden lytic event occurs, causing the condyle to become progressively smaller and in some cases even disappear. Condylysis is not usually associated with ankylosis, erosive changes in the fossae, or a positive serologic result.263,264
Neither any history nor examination diagnostic criteria have been defined. However, imaging will show evidence of resorption of part or all of the condyle, and laboratory testing will confirm negative serologic results for rheumatologic disease.
C. Osteochondritis dissecans (ICD-10 M93.20; ICD-9 732.7). Osteochondritis dissecans is a joint condition in which a piece of cartilage, along with a small bone fragment, break loose from the end of the bone and result in loose osteochondral fragments within the joint. It usually occurs in the knee and elbow and is often related to sports. Case reports describe the condition in the TMJ, but little is known about signs and symptoms.
Neither any history nor examination diagnostic criteria have been defined. However, imaging will show evidence of loose osteochondral fragments within the joint, and laboratory testing will confirm negative serologic results for rheumatologic disease.
D. Osteonecrosis (ICD-10 M87.08; ICD-9 733.45). Osteonecrosis is a painful condition most commonly affecting the ends of long bones such as the femur. Other common sites include the humerus and the knees. The condition is also found in the mandibular condyle, as shown on MRI scans as a decreased signal in proton density and on T1-weighted images and can be combined with an increased signal on T2-weighted images (edema). The cause, clinical significance, and the need for treatment are unknown.
In order to diagnose osteonecrosis, the patient must report joint pain, examination must confirm arthralgia, and radiography must show a decreased signal in proton density on MRI scans and on T1-weighted images, which can be combined with an increased signal on T2-weighted images. In addition, laboratory testing will confirm negative serologic results for rheumatologic disease.
E. Systemic arthritides (rheumatoid arthritis: ICD-10 M06.9; ICD-9 714.0). Systemic arthritides is joint inflammation resulting in pain or structural changes caused by a generalized systemic inflammatory disease, including rheumatoid arthritis (RA), juvenile idiopathic arthritis, spondyloarthropathies (eg, ankylosing spondylitis, psoriatic arthritis, infectious arthritis, Reiter syndrome), and crystal-induced disease (eg, gout, chondrocalcinosis). Other rheumatologically related diseases that may affect the TMJ include autoimmune disorders and other mixed connective tissue diseases (eg, scleroderma, Sjögren’s syndrome, lupus erythematosus).265 This group of arthritides therefore comprises multiple diagnostic categories that are best diagnosed and managed by rheumatologists regarding the general/systemic therapy. Clinical signs and symptoms of an ongoing chronic (TMJ) inflammation are variable between patients and within a patient over time. They can vary from no sign/symptom to only pain to only swelling/exudate to only tissue degradation to only growth disturbance. Resorption of condylar structures may be associated with malocclusion, such as a progressive anterior open bite. A diagnostic instrument should aim to identify patients with chronic inflammation early and accurately, should not exclude patients with chronic arthritis of long duration, and should cover not only RA but the whole range of chronic inflammatory states.
In order to diagnose systemic arthritides, the patient must have both of the following:
• Diagnosis of a systemic inflammatory joint disease by a rheumatologist
• Diagnosis of DJD
The examination and imaging will show the same criteria as those defined for DJD. The laboratory criteria include elevated levels of IL-1β, IL-1Ra, IL-6, TNF, serotonin, or glutamate or reduced levels of IL-1sRII or TNFsRII in the synovial fluid.
F. Neoplasm (benign: ICD-10 D16.5; ICD-9 213.1; malignant: ICD-10 C41.1; ICD-9 170.1).
A neoplasm is new, often uncontrolled growth of abnormal tissue, in this case arising or involving the TMJ or supporting structures. Neoplasms in this area may be benign, malignant, or metastatic. Although neoplasia as an underlying cause of TMJ dysfunction is rare, it is well known in the literature.266,267 Approximately 3% of malignant neoplasms metastasize to the jaws.268–272 Neoplasms most frequently extending to the TMJ region, causing pain and dysfunction, include squamous cell carcinomas of the maxillofacial region and primary nasopharyngeal tumors.16,273–281 Neoplasms arising in the parotid gland, such as adenoid cystic carcinomas and mucoepidermoid carcinomas, may also produce TMJ pain and dysfunction.282–284 The most common signs and symptoms include reduced opening, crepitation, occlusal changes, pain with function, and swelling.285 If the condyle is involved, there is frequently development of a facial asymmetry with a midline shift similar to that seen in condylar hyperplasia.286 The most common treatment is surgery. Diagnostic imaging and biopsy are essential when a neoplasm is suspected.
G. Synovial chondromatosis (ICD-10 D48.0; ICD-9 238.0). Synovial chondromatosis is a cartilaginous metaplasia of the mesenchymal remnants of the synovial tissue of the joint. Its main characteristic is the formation of cartilaginous nodules that may be pedunculated and/or detached from the synovial membrane, becoming loose bodies within the joint space. Calcification of the cartilage can occur (ie, osteochondromatosis). The disease may be associated with malocclusion, such as a progressive ipsilateral posterior open bite. Imaging is needed to establish the diagnosis.
In order to diagnose synovial chondromatosis, the history be positive for at least one of the following:
• Report of preauricular swelling
• Arthralgia
• Progressive limitation in mouth opening
• Joint noise in the past month
In addition, the examination must confirm at least one of the following:
• Preauricular swelling
• Arthralgia
• Maximum assisted opening (passive stretch) less than 40 mm, including the vertical incisal overlap
• Crepitus
Imaging will show the following:
• MRI: multiple chondroid nodules, joint effusion, and amorphous iso-intensity signal tissues within the joint space and capsule
• CT/Cone-beam CT: loose calcified bodies in the soft tissues of the TMJ
Histologic examination will confirm cartilaginous metaplasia.
4. Fractures
Direct traumatic force can injure all related bony components of the masticatory system (ie, temporal bone, maxilla, zygoma, sphenoid bone, and mandible). This trauma can be related to the following conditions: fracture, dislocation, contusion, and/or laceration of the articular surfaces, ligaments, or the disc, with or without intra-articular hemarthrosis. Sequelae could include adhesions, ankylosis, occlusal abnormalities, or joint degeneration.185,287 Patients with nonsurgically treated dislocated fractures may be prone to symptoms of TMDs, functional disorders, and occlusal disorders.287 Fractures of the condylar process may result in facial asymmetry, in general with greater skeletal changes when the fracture occurs earlier in life. Closed treatments have reportedly also resulted in facial asymmetries, even in adults.288
Examples of fractures are the following:
A. Closed fracture of the condylar process (ICD-10 S02.61XA; ICD-9 802.21)
B. Closed fracture of the subcondylar process (ICD-10 S02.62XA; ICD-9 802.22)
C. Open fracture of the condylar process (ICD-10 S02.61XB; ICD-9 802.31)
D. Open fracture of the subcondylar process (ICD-10 S02.62XB; ICD-9 802.32)
In order to diagnose a fracture, the history must be positive for at least one of the following:
• Trauma to the orofacial region and associated preauricular swelling
• Arthralgia
• Limited mouth opening
In addition, the examination must confirm at least one of the following:
• Preauricular swelling
• Arthralgia
• Maximum assisted opening (passive stretch) less than 40 mm, including vertical incisal overlap
Imaging will show evidence of fracture.
5. Congenital/developmental disorders
A. Aplasia (ICD-10 Q67.4; ICD-9 754.0). Aplasia is defined as a typically unilateral absence of the condyle and incomplete development of the articular fossa and eminence, resulting in facial asymmetries. It is commonly associated with other congenital anomalies (eg, oculo-auriculovertebral spectrum [Goldenhar syndrome], hemifacial microsomia, and mandibulo-facial dysostosis [Treacher Collins syndrome]). It is occasionally bilateral; in such cases, asymmetry is not present, but micrognathia is the dominant clinical manifestation. The condition may be associated with malocclusion, which may include open bite.
In order to diagnose aplasia, the history must be positive for the following:
• Progressive development of mandibular asymmetry or micrognathia from birth or early childhood
• Development of malocclusion, which may include posterior open bite
In addition, the examination must confirm this history. Imaging will show both of the following:
• Severe hypoplasia of the fossa
• Aplasia of the condyle
B. Hypoplasia (ICD-10 M27.8; ICD-9 526.89).Hypoplasia is defined as incomplete development or underdevelopment of the cranial bones or the mandible. Growth is proportionately reduced and less severe than in aplasia. Condylar hypoplasia can be secondary to facial trauma. The condition may be associated with malocclusion, which may include open bite. Sensitivity and specificity have not been established.
In order to diagnose hypoplasia, the history must be positive for the following:
• Progressive development of mandibular asymmetry or micrognathia from birth or early childhood
• Development of malocclusion, which may include posterior open bite
In addition, the examination must confirm this history. Imaging will show at least one of the following:
• Hypoplasia of the fossa
• Hypoplasia of the condyle
• Shortened mandibular ramus height
C. Hyperplasia (ICD-10 M27.8; ICD-9 526.89).
Hyperplasia is an overdevelopment of the cranial bones or mandible. It is a non-neoplastic increase in the number of normal cells. It can occur unilaterally or bilaterally as a localized enlargement, such as condylar hyperplasia, or as an overdevelopment of the entire mandible or side of the face. Sensitivity and specificity have not been established.
In order to diagnose hyperplasia, the history must be positive for progressive development of mandibular or facial asymmetry, and the examination must confirm this history. Imaging will show the following:
• Asymmetry in mandibular ramus height
• Hot spots on technetium scan (for active conditions only)
Masticatory muscle disorders
The mechanisms that produce pain in skeletal muscles are still not well understood. Overuse of a normally perfused muscle or ischemia of a normally working muscle may cause pain.289–292 Sympathetic and fusimotor reflexes can produce changes in the blood supply and muscle tone.293,294 Furthermore, different psychologic or emotional states can alter muscle tone.295,296 Neurons that mediate pain from skeletal muscles are subject to strong modulatory influences. Endogenous substances (eg, bradykinin, serotonin, prostaglandins, neuropeptides, and substance P) can sensitize the nociceptive endings very easily. Painful muscle conditions not only lead to increased sensitivity of peripheral nociceptors but also produce hyperexcitability in the central nervous system, resulting in localized hyperalgesia and allodynia.33,289,297–299
Some systemic conditions that produce muscle pain are polymyalgia rheumatica, polymyositis, dermatomyositis, lupus erythematosus, and fibromyalgia. Fibromyalgia is of particular interest because it may easily be confused with a regional masticatory muscle disorder. When fibromyalgia is suspected, a referral to a rheumatologist is in order.
1. Muscle pain limited to the orofacial region
A. Myalgia (ICD-10 M79.1; ICD-9 729.1). Myalgia is defined as pain of muscle origin affected by jaw movement, function, or parafunction and replication of this pain with provocation testing of the temporalis or masseter muscles. Limitation of mandibular movements secondary to pain may be present. Although not required, a positive finding with the specified provocation tests when examining the other masticatory muscles can help to corroborate this diagnosis. There are three subtypes of myalgia: (1) local myalgia, (2) myofascial pain with spreading, and (3) myofascial pain with referral.
i. Local myalgia. Local myalgia is defined as pain of muscle origin plus a report of pain localized to the location of the palpating fingers. Limitation of mandibular movements secondary to pain may be present.
In order to diagnose local myalgia, the history must be positive for both of the following:
• Pain in the jaw, temple, in front of the ear, or in the ear in the last 30 days
• Pain changed with jaw movement, function, or parafunction
In addition, examination of the temporalis or masseter muscle must confirm both of the following:
• Confirmation of pain location in the area of the temporalis or masseter muscle
• Familiar muscle pain with palpation or maximum unassisted or assisted opening
Note: The pain is not better accounted for by another pain diagnosis, and other masticatory muscles may be examined as required.
ii. Myofascial pain with spreading. This type of pain is described as pain of muscle origin plus a report of pain spreading beyond the location of the palpating fingers but within the boundary of the masticatory muscle being examined. Limitation of mandibular movements secondary to pain may be present.
In order to diagnose myofascial pain with spreading, the patient must have local myalgia, and the examination of the temporalis or masseter muscle must confirm both of the following:
• Familiar muscle pain with palpation
• Pain with muscle palpation with spreading of the pain beyond the location of the palpating fingers
Note: Other masticatory muscles may be examined as required.
iii. Myofascial pain with referral. This type of pain is described as pain of muscle origin as defined for myalgia plus a referral of pain beyond the boundary of the masticatory muscles being palpated, such as to the ear, tooth, or eye. Limitation of mandibular movements secondary to pain may be present. Although not required for this diagnosis, taut bands (ie, contracture of muscle fibers) in the muscles may be present.
In order to diagnose myofascial pain with referral, the patient must have local myalgia, and the examination of the temporalis or masseter muscle must confirm all of the following:
• Confirmation of pain location in the area of the temporalis or masseter muscle
• Familiar muscle pain with palpation
• Pain with muscle palpation beyond the boundary of the muscle
Note: Other masticatory muscles may be examined as required.
B. Tendonitis (ICD-10 M67.90; ICD-9 727.9).Tendonitis is pain of tendon origin affected by jaw movement, function, or parafunction and replication of this pain with provocation testing of the masticatory tendon. Limitation of mandibular movements secondary to pain may be present. The temporalis tendon is a common site of tendonitis and referred pain to the teeth.
In order to diagnose tendonitis, the patient must have myalgia, and the examination must confirm the diagnosis of myalgia but restricted to the temporalis tendon.
Note: This condition could also apply to other masticatory muscle tendons.
C. Myositis (non-infective: ICD-10 M60.9; ICD-9 729.1; infective: ICD-10 M60.009; ICD-9 728.0). Myositis is pain of muscle origin with clinical characteristics of inflammation or infection: edema, erythema, and/or increased temperature. It generally arises either acutely following direct trauma of the muscle or infection or chronically with autoimmune disease. Limitation of unassisted mandibular movements secondary to pain is often present. Calcification of the muscle can occur (ie, myositis ossificans).
In order to diagnose myositis, the patient must have local myalgia, and the examination of the temporalis or masseter muscle must confirm both of the following:
• Local myalgia
• Presence of edema, erythema, and/or increased temperature over the muscle
In addition, serologic tests may reveal elevated enzyme levels (eg, creatine kinase), markers of inflammation, and the presence of autoimmune diseases.
Note: Other masticatory muscles may be examined as required.
D. Spasm (ICD-10 M62.838; ICD-9 728.85). A spasm is defined as a sudden, involuntary, reversible tonic contraction of a muscle.
In order to diagnose a spasm, the patient must report the following:
• Immediate onset of myalgia
• Immediate report of limited range of jaw motion
In addition, the examination must confirm both of the following:
• Myalgia in any of the masticatory muscles
• Limited range of jaw motion in the direction that elongates the affected muscle (eg, for jaw closers, opening will be limited to less than 40 mm; for the lateral pterygoid muscle, ipsilateral movement will be limited to less than 7 mm)
When this diagnosis needs to be confirmed, laboratory testing will confirm elevated electromyographic activity compared with the contralateral unaffected muscle.
Note: Acute malocclusion may be present.
2. Contracture (muscle: ICD-10 M62.40; ICD-9 728.85; tendon: ICD-9 727.81)
Contracture is defined as the shortening of a muscle due to fibrosis of tendons, ligaments, or muscle fibers. It is usually not painful unless the muscle is overextended. A history of radiation therapy, trauma, or infection is often present.
In order to diagnose contracture, the patient must have progressive loss of range of motion and the examination must confirm that unassisted and assisted jaw movements are limited.
3. Hypertrophy (ICD-10 M62.9; ICD-9 728.9)
Hypertrophy is the enlargement of one or more masticatory muscles. It is usually not associated with pain and can be secondary to overuse and/or chronic tensing of the muscles. Some cases are familial or genetic in origin.
In order to diagnose hypertrophy, the patient must have enlargement of one or more masti-catory muscles as evidenced from photographs or previous records, and the examination must confirm this englargement.
Note: Diagnosis is based on the clinician’s assessment of muscle size and requires consideration of craniofacial morphology and ethnicity.
4. Neoplasm
Neoplasms of the masticatory muscles result from tissue proliferation with histologic characteristics and may be benign (eg, myoma) or malignant (eg, rhabdomyosarcoma or metastasis). They are uncommon and may present with swelling, spasm, pain during function, limited mouth opening, and/or sensorimotor changes (eg, paresthesias). Diagnostic imaging and biopsy are essential when a neoplasm is suspected.
ICD coding of neoplasms includes the following:
• In the jaw:
º Malignant (ICD-10 C41.1; ICD-9 170.1)
º Benign (ICD-10 D16.5; ICD-9 213.1)
• In the soft tissues of the head, face, and neck:
º Malignant (ICD-10 C49.0; ICD-9 171.0)
º Benign (ICD-10 D21.0; ICD-9 215.0)
5. Movement disorders
A. Orofacial dyskinesia. Orofacial dyskinesia involves involuntary, mainly choreatic (dance-like) movements that may involve the face, lips, tongue, and/or jaw. The disorder may result in traumatic injury to the oral mucosa or the tongue. It is more common with advancing age and in patients with a history of using neuroleptic medications and/or associated with traumatic brain injury, psychiatric conditions, or other neurologic disorders (eg, Wilson disease). Reduction or discontinuation of the movement pattern could occur when the mouth or face receives sensory stimulation (“sensory trick”).
ICD coding of orofacial dyskinesia includes the following:
i. Abnormal involuntary movements (ICD-10 R25.1 [tremor unspecified], R25.2 [cramp and spasm], R25.3 [fasciculations]; ICD-9 781.0)
ii. Ataxia, unspecified (ICD-10 R27.0; ICD-9 781.3); muscular incoordination (ICD-10 R27.9; ICD-9 781.3)
iii. Subacute, due to drugs; oral tardive dyskinesia (ICD-10 G24.01; ICD-9 333.85)
B. Oromandibular dystonia. Oromandibular dystonia involves excessive, involuntary, and sustained muscle contractions that may involve the face, lips, tongue, and/or jaw. They could be components of a number of central nervous systems disorders, including Parkinson’s disease and Meige syndrome, and could be an adverse event related to medication usage, notably neuroleptics. Trauma to the brain, head, and neck can trigger the onset of transient or permanent dystonia of the masticatory muscles. The disorder can also be genetically determined. Normally, the dystonia disappears during sleep. The affected muscles are often painful. The condition can make opening and closing of the mouth difficult and impair speech, swallowing, and chewing.
ICD coding of oromandibular dystonia includes the following:
i. Acute, due to drugs (ICD-10 G24.02; ICD-9 333.72)
ii. Deformans, familial, idiopathic, and torsion dystonia (ICD-10 G24.1; ICD-9 333.6)
6. Masticatory muscle pain attributed to systemic/central disorders
A. Fibromyalgia (ICD-10 M79.7; ICD-9 729.1).Fibromyalgia is defined as widespread pain with concurrent masticatory muscle pain (see chapter 10).
In order to diagnose fibromyalgia, the patient must meet the Fibromyalgia Diagnostic Criteria,300 and examination must confirm either myalgia or myofascial pain.
B. Centrally mediated myalgia (ICD-10 M79.1; ICD-9 729.1). Centrally mediated myalgia is defined as chronic, continuous muscle pain that is aggravated by function. It is likely centrally mediated. Intermittent muscle pain conditions may not produce centrally mediated myalgia, while a prolonged and constant period of muscle pain is likely to lead to the condition.
In order to diagnose centrally mediated myalgia, the history must be positive for all of the following:
• Prolonged and continuous pain in the jaw, temple, in front of the ear, or in the ear in the last 30 days
• Regional dull, aching pain at rest
• Pain is aggravated by function of the affected muscles
• Presence of at least three nonspecific somatic symptoms, such as:
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


