Introduction
Patients with type 1 diabetes have shown decreased bone mineral density (BMD) values. The need for orthodontic treatment in diabetic patients is usually associated with occlusal problems and the occurrence of abnormalities in the development of the jaws. The aim of this study was to analyze bone response of insulin-treated and untreated diabetic rats after applying orthodontic forces.
Methods
Wistar rats were divided into 3 groups: experimental orthodontics, experimental diabetes and orthodontics, and experimental diabetes treated with insulin and experimental orthodontics. Orthodontic forces were applied the first day of the seventh week. Forty-eight hours after placement, all the animals were killed, and the maxillae were excised and processed using routine histologic techniques.
Results
Bone activity in the periodontal cortex of the dental alveolus showed a significant decrease in bone formation and erosive areas in diabetic animals as compared with controls. A recovery of these parameters could be observed in the group with experimental diabetes treated with insulin and experimental orthodontics. Bone volume in the interradicular bone showed no significant differences among groups.
Conclusions
People with diabetes should not receive orthodontic treatment until their metabolic status normalizes. Bone response to orthodontic forces in insulin-treated diabetic subjects does not differ significantly from that observed in healthy subjects.
Type 1 diabetes, previously known as insulin-dependent, type I or juvenile diabetes, accounts for 5% to 10% of diabetic patients and has a peak incidence at 10 to 14 years of age. It is caused by an absolute deficiency in insulin secretion, which results from an autoimmune destruction of pancreatic β cells. Type 2 diabetes, formerly referred to as insulin-independent, type II or adult-onset diabetes, accounts for 90% to 95% of diabetic patients, including many who are insulin resistant and often have relative insulin deficiency.
Long-term complications of diabetes include retinopathy, nephropathy, peripheral neuropathy, and autonomic neuropathy, causing gastrointestinal, genitourinary, and cardiovascular symptoms, as well as sexual dysfunction.
Alteration in bone metabolism is another common observation among long-term complications found in diabetic patients, even in patients treated with insulin. Clinical assessment of this alteration is usually performed based on bone mineral density (BMD) determinations. It is a fast, noninvasive, painless method to determine the amount of calcium in the spinal column, wrist, arm, or leg and therefore provides an estimate of fracture risk in individual patients.
Studies of type 2 diabetes report decreased, normal, and increased BMD values compared with nondiabetic patients. Most studies of type 1 diabetic patients show decreased BMD values, even when the onset of the disease is after peak bone mass has been achieved.
Insulin therapy reverses damage to organs associated with hyperglycemia, or at least it partially does. Nevertheless, there are reports that show significant changes in bone in diabetic patients, even in those receiving intensive insulin treatment, and in young patients.
The need for orthodontic treatment in adult diabetic patients is usually associated with a number of occlusal problems related to periodontal degradation and tooth loss, which in turn lead to nutritional deficiencies, psychosocial consequences and impaired quality of life. Another reason for orthodontic treatment in young diabetic patients is the occurrence of abnormalities in the development of the jaws. There are clinical and experimental studies reporting a decrease in cephalometric parameters and delayed skeletal development in individuals with type 1 diabetes, all of which are predisposing factors for dental malpositions.
Orthodontic treatment is based on the principle that a prolonged application of pressure to the tooth will result in movement, which is a consequence of remodeling of the surrounding bone. The success of orthodontic treatment largely depends on bone response to the application of forces; hence, bone health is fundamental in order to obtain tooth movements as they were planned.
According to Bensch et al, Burden et al, and Proffit and Fields, if diabetes is properly controlled, the periodontal and bone response to orthodontic forces is almost normal, and a satisfactory orthodontic result can be obtained. Conversely, if diabetes is poorly controlled, it is accepted that there is a real risk of accelerating periodontal degradation, therefore contraindicating orthodontic treatment until the metabolic disorder is compensated. However, there is no scientific evidence or experimental or clinical studies that support these principles used in the clinical practice of orthodontics, as there are scant reports in the literature combining experimental orthodontics with diabetes. Therefore, the aim of this study was to analyze bone response of insulin-treated and untreated diabetic rats after applying orthodontic forces.
Material and methods
Experimental animals
Twenty-four male Wistar rats weighing 100 to 130 g were used. They were divided into 3 groups of 8 animals each:
- I.
Experimental orthodontics (ORT)
- II.
Experimental diabetes and orthodontics (DBT + ORT)
- III.
Experimental diabetes treated with insulin and experimental orthodontics (DBT + INS + ORT)
The animals were kept in groups of 4 in each cage. They were given regular rat chow and water ad libitum . The animals were kept under 12-hour light-dark cycles (light 7:00 am to 7:00 pm ), at room temperature (18–22°C) and relative humidity (40%–60%). All procedures for treatment and maintenance of the animals were conducted in keeping with internationally accepted guidelines (“Guidelines for the care and use of laboratory animals”, NIH Publication, 1985) and were approved by the Ethics Committee of the School of Dentistry of the University of Buenos Aires.
Experimental diabetes induction
Diabetes was induced on day 1 of the experiment in groups II and III by administering a single intraperitoneal injection of streptozotocin (STZ) 60 mg/kg body weight (Sigma-Aldrich, Inc., St. Louis, Mo), dissolved in freshly prepared citrate buffer pH 4.0. STZ is a cytotoxic agent that is relatively selective against pancreatic β cells in animals, causing an irreversible diabetic state. Therefore, diabetes induced by this method is similar to type 1 diabetes, which is insulin-dependent or insulin-requiring diabetes. The animals in the nondiabetic group (group I) received an equivalent volume of citrate buffer. The orthodontic device was applied 6 weeks after diabetes was induced.
Sampling and determination of blood glucose
Twenty-four hours after administration of STZ in the diabetic groups and administration of the vehicle in the control group, blood samples were obtained from all animals for quantitative determination of blood glucose using the glucose oxidase method with Accu-Chek Sensor Comfort test strips (Roche Products Ltd, Santiago, Chile) in the Accu-Chek Sensor (Roche Diagnostics GmbH, Mannheim, Germany). Only animals with blood glucose levels higher than 250 mg/dL were included in the different groups of experimental diabetes, and only those with blood glucose levels less than 120 mg/dL were included in group I.
Determinations were repeated once a week in non–insulin treated groups, to ensure that glycemic levels were as expected, and every 48 hours in insulin-treated rats. All the animals included in the study met the inclusion criteria corresponding to their particular group.
Insulin treatment
Insulin therapy was initiated in the insulin-treated diabetic group the day after STZ injection by daily administration of human NPH insulin subcutaneously (Humulin N, Eli Lilly y Compañía de México S.A. de C.V., Mexico D.F., Mexico) according to the requirements of each animal.
Installation of the orthodontic appliance and sample collection
Orthodontic forces were applied to all animals the first day of the seventh week of the experiment. The orthodontic appliance was specifically designed for the rat and meets the basic standards applied in the clinical setting. It consists of 2 stainless steel molar bands with tubes welded to their palatal aspect. These bands are cemented to the upper first molars of rats with glass ionomer cement. A 0.014-in circular cross-section stainless steel wire was shaped into a helical spring to exert a force magnitude of 120 ± 15 g toward the vestibular plate ( Fig 1 ).
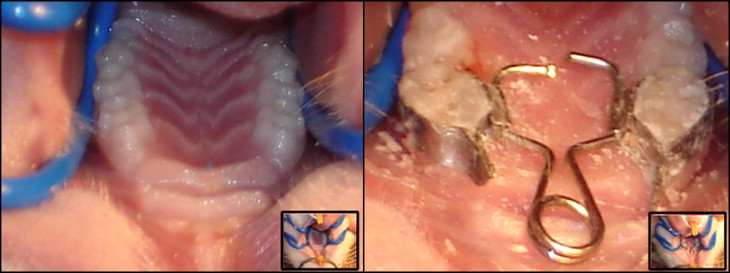
The animals were anesthetized with a xylazine and ketamine solution (Kensol, Laboratorios König SA, Buenos Aires, Argentina; Ketamine 50, Holliday-Scott SA, Buenos Aires, Argentina) in order to place the device. Forty-eight hours after placement of the orthodontic device and after measuring blood glucose levels, all the animals were killed (day 44 of the experiment) by an intraperitoneal overdose of sodium thiopental (150 mg/kg) (Pentovet, Richmond Laboratorios, Buenos Aires, Argentina) and acepromazine maleate (3 mg/kg) (Acedan, Holliday-Scott SA, Buenos Aires, Argentina).
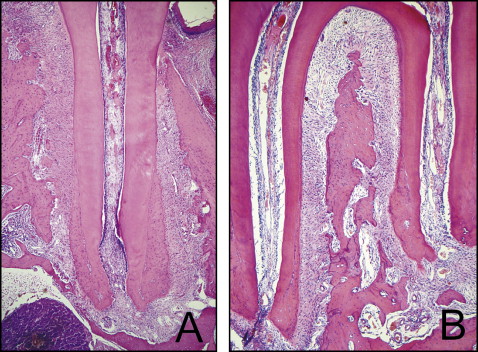
Immediately after death, body weight was recorded and the maxillae were excised and fixed in formalin buffer (pH 7.0) for 48 hours. After this time, the samples were carefully dissected and decalcified by immersion in a solution of 10% EDTA (ethylenediaminetetraacetic acid), pH 7, for 30 days. The samples were then processed using routine histologic techniques. Buccopalatal and longitudinally oriented sections of the mesial root of the upper first molar were obtained under stereoscopic microscope; the blocks were then reoriented under stereoscopic microscope, changing the angulation of the platen of the microtome to obtain sections of the distal roots, which were also buccopalatal and longitudinally oriented. All the sections were oriented in an apicocoronal direction, parallel to the long axis of the corresponding root. All the sections were stained with hematoxylin and eosin.
Digital photographs were taken of the histologic sections with a digital camera Canon Powershot A640 10.0 megapixel 4x optical zoom lens (Canon Inc., Tokyo, Japan) mounted on an optical microscope Carl Zeiss Axioskop 2 (Carl Zeiss Mikroskopie, Jena, Germany) with a 5x objective. The images obtained were used for the histomorphometric study using the Image-Pro Plus 4.0 image analysis program (Media Cybernetics, Inc, Bethesda, Md) ( Fig 2 ).
Body weight record
Body weight of each animal was recorded periodically with a digital laboratory balance Ohaus CS 2000 (Ohaus Corp, Pine Brook, NJ) to draw a curve of weight gain.
Histologic and histomorphometric studies
The following histomorphometric parameters were assessed on the dental alveolus according to stereologic principles and using current nomenclature as stated by Parfitt et al :
- •
Bone activity
-
On digital images of sections of the mesial root, static bone activity was evaluated by determining the percentage of the following parameters on the surface of the periodontal cortical of alveolar bone in the studied area:
-
Areas covered by osteoblasts (Obs/BS)
-
Total erosive surfaces (ES/BS)
-
Erosive surfaces covered with osteoclasts (ES(Oc+)/BS)
-
Erosive surfaces not covered with osteoclasts (ES(Oc−)/BS)
-
Resting surfaces (rest)
-
Surfaces facing hyalinized areas (nec): percentage of the surface of the periodontal cortex of alveolar bone in the studied area presenting surfaces facing necrotic periodontal ligament (hyalinized areas). These areas were defined as areas of necrosis.
-
-
- •
Number of osteoclasts
-
Total number of cells that are morphologically recognizable as osteoclasts found lining the periodontal cortex of alveolar bone measured at the mesial root.
-
- •
Bone volume of the interradicular bone (BV/TV)
-
Fraction of bone tissue in total volume. Total volume was taken as bone tissue plus bone marrow evaluated at the level of the distal roots.
-
Statistical analysis
The values are expressed as mean ± standard deviation. The statistical analysis of the results was performed using 1-way analysis of variance (ANOVA). Multiple comparisons were made using the Bonferroni test. The level of statistical significance was set at a value of P <0.05.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


