Introduction
The objective of this systematic review was to assess the short- and long-term release of components of orthodontic adhesives and polycarbonate brackets in the oral environment.
Methods
Electronic database searches of published and unpublished literature were performed. The following electronic databases with no language and publication date restrictions were searched: MEDLINE (via Ovid and PubMed), EMBASE (via Ovid), Cochrane Oral Health Group’s Trials Register, and CENTRAL. Unpublished literature was searched on ClinicalTrials.gov , the National Research Register, and Pro-Quest Dissertation Abstracts and Thesis database. The reference lists of all eligible studies were checked for additional studies. Two review authors performed data extraction independently and in duplicate using data collection forms. Disagreements were resolved by discussion or the involvement of an arbiter.
Results
No randomized controlled trial was identified. In the absence of randomized controlled trials, observational studies were included. Eleven studies met the inclusion criteria. All were observational studies conducted in vivo or in vitro. The bisphenol-A release from orthodontic bonding resins was found to be between 0.85 and 20.88 ng per milliliter in vivo, and from traces to 65.67 ppm in vitro. Polycarbonate brackets released amounts of 22.24 μg per gram in ethanol solution and 697 μg per gram after 40 months in water. Bis-GMA and TEGDMA leaching in vitro reached levels of 64 and 174 mg per 10 μL, respectively. Because of the heterogeneity in methodologies and reporting, only qualitative synthesis was performed.
Conclusions
The available evidence on this topic derived from observational in-vivo and in-vitro studies that represent a moderate level of evidence. The variety of setups and the different units allied to the diversity of reporting among studies did not allow calculation of pooled estimates.
Orthodontic polymers constitute a large class of materials, appliances, and auxiliaries, which entail a wide array of appliances including brackets and various auxiliaries: eg, polyamide-based wire sleeves, polyurethanes used for the manufacturing of elastomeric ligatures and chains, polypropylene for lip bumper appliances, and polycarbonates for esthetic bracket manufacturing. Furthermore, orthodontic composite resins have been used for the past 40 years for bracket bonding with success. These multifaceted applications of polymers in orthodontic material applications have introduced innovation, practicality, and esthetics into the specialty.
The compositions and configurations of these materials have notable variations; orthodontic resins, for example, along with monomer systems, contain many additives that are used to induce or inhibit the polymerization reaction. Bisphenol-A (BPA) is used in the manufacturing process of some of these monomers. It can be used either as a precursor of BPA glycidyl dimethacrylate (Bis-GMA) and BPA dimethacrylate, or in the production of the aromatic polycarbonate matrix of many plastic orthodontic brackets and other composite resins. Bis-GMA forms a bulky, stiff chain that offers less susceptibility to biodegradation and more rigidity and strength in comparison with aliphatic polycarbonates. Because of its high molecular weight, viscosity, and stereotactic structure, this monomer shows hindrance phenomena during polymerization as a result of its relatively high stiffness and decreased probability of flexibly moving into space. Thus, comonomers such as triethylene glycol dimethacrylate (TEGDMA) are used to dilute the thick consistency and provide a higher degree of cure because of their relatively lower molecular weight and flexibility.
Apart from residual monomers, which have not been polymerized, eluted components from resins can also include a wide variety of substances: methacrylic acid, benzoic acid as a degradation outcome of the benzoyl peroxide initiator, derivatives from enzymatic hydrolysis, and other substances from the polymerization process of accelerators and catalysts. Consequently, there has been a considerable body of research indicating adverse effects of residual monomer leaching from methacrylate-based restorative materials. Their biocompatibility has been investigated, showing allergic, cytotoxic, and mutagenic effects, both in vivo and in vitro.
On the other hand, BPA used for the manufacturing of some adhesive resins and polycarbonate brackets has been reported to induce hormone-related effects including altered peripubertal mammary gland development in mice, early puberty in females, and feminization in males. Research in humans has shown that the systemic intake of chemical components released from resin-based materials takes place via 3 main pathways: inhalation of volatile particles in the lungs, ingestion of compounds in the gastrointestinal tract, and diffusion into the pulp through the dentin.
In orthodontics, the most hazardous conditions leading to the systematic intake of chemical particles are brackets in the oral cavity for approximately 2 years and bonded lingual retainers for many years, as well as procedures such as debonding. Whereas the materials in the oral cavity are more relevant for patients, debonding procedures and associated production of particles are of concern to the orthodontist and the dental personnel.
Adhesives used to bond brackets on the tooth surface are exposed to the oral setting only through the brackets’ peripheral margins and have an average thickness of 150 to 250 μm. In this case, the functional stimuli are minimal, and the subsequent effect of aging is minor. The same material used to bond fixed lingual retainers, however, presents a drastically different situation. Much of the adhesive resin surface is exposed to the severe intraoral conditions, and the thickness of the material used might predispose it for an incomplete cure and, consequently, greater leaching. This applies especially to retainers that are bonded from canine to canine or even from premolar to premolar. Furthermore, the location of the bonded retainer encourages plaque accumulation and maximizes the effect of enzymatic activity, especially from the esterase in saliva.
The reports in the literature on the potential of leaching of various compounds are sporadic. Therefore, the objective of this systematic review was to review the literature and assess the short- and long-term release of components of orthodontic adhesives and polycarbonate brackets, such as BPA, Bis-GMA, TEGDMA, and other of their monomers, in the oral environment.
Material and methods
The selection criteria for the review included (1) study design: any study was considered including randomized clinical trials, observational studies, and in-vitro studies in the absence of the first; (2) interventions: use of resin-based materials such as orthodontic adhesives and orthodontic polycarbonate brackets; (3) quantified eluates under investigation: BPA, Bis-GMA, TEGDMA, and phosphoric acid ester; and (4) incubation medium: no restriction was applied regarding the incubation solution, its temperature, and the period of artificial aging.
The exclusion criteria were acrylic-based materials, materials used for removable orthodontic appliances, and autopolymerized adhesives. The exclusion criteria were selected because the focus of this review was the procedure of bonding brackets, particularly polycarbonate brackets. Many of the excluded materials are no longer used in orthodontics, or they contain particles irrelevant to the ones examined in this study.
The following electronic databases were searched: MEDLINE (via Ovid and PubMed; Appendices 1 and 2 [online only]) (1946 to week 2 of October 2012), EMBASE (via Ovid), the Cochrane Oral Health Group’s Trials Register, and CENTRAL.
Unpublished literature was searched on ClinicalTrials.gov , the National Research Register, and Pro-Quest Dissertation Abstracts and Thesis database.
The search attempted to identify all relevant studies irrespective of language. There were no restrictions on date of publication. The reference lists of all eligible studies were checked for additional studies.
Assessment of research for including studies in the review and extraction of data were performed independently and in duplicate by the first 2 authors, who were not blinded to the identity of the authors, their institution, or the results of the research. The full report of publications considered by either author to meet the inclusion criteria was obtained and assessed independently. Disagreements were resolved by discussion and consultation with the third author.
The first 2 authors performed the data extraction independently and in duplicate. Disagreements were resolved by discussion or the involvement of a collaborator (third author). Data collection forms were used to record the desired information. The following data were collected on a customized data collection form: type of orthodontic material; substance under investigation; amount of material used; time of polymerization or photocuring; control or reference group; solution of immersion or analysis or solution temperature; time of analyses after immersion or placement; and method of analysis.
Results
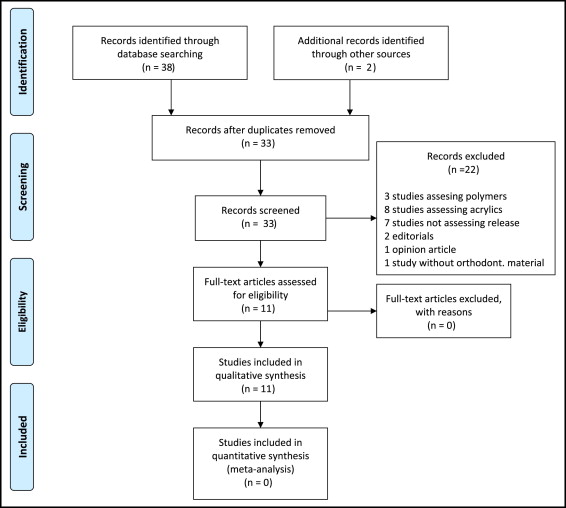
Thirty-three studies were initially deemed potentially relevant to the review ( Fig ). After we reviewed the abstracts and read the full texts, 11 studies satisfied the inclusion criteria. All 11 studies included in the qualitative analysis were observational studies conducted in vivo or in vitro ( Tables I and II ). Because of heterogeneity, only qualitative synthesis was attempted.
| Type of orthodontic material | Substance under investigation | Amount of material used | Time of polymerization or photocure | Control or reference group | Solution of immersion or analysis/solution temperature | Time of analyses after immersion or placement | Method of analysis | |
|---|---|---|---|---|---|---|---|---|
| Kang et al 2011 | Light-cured adhesive resins | BPA | As used in everyday practice | 6 s for each tooth | Samples before and after placement | Saliva and urine samples | Before placement, immediately after placement, 1 d, 1 wk, 1 mo | Liquid chromatography and mass spectroscopy |
| Sunitha et al 2011 | Light-cured adhesive resin | BPA | As used in everyday practice | 40 s for each tooth | None | Absolute alcohol (99% v/v) | 1, 7, 21, and 35 days | Liquid chromatography |
| Eliades et al 2011 | Light-cured adhesive resin | BPA | As used in everyday practice | 20 s with light- curing unit | Storage water of immersed teeth | Double distilled water | 10, 20, and 30 days | Gas chromatography and mass spectroscopy |
| Eliades et al 2007 | Chemically cured and light-cured adhesive resin | BPA | Standardized volume of adhesive | 20 s for each tooth for the light-cured adhesive | None | Absolute alcohol (99% v/v) | 1, 7, 21, and 35 days | Liquid chromatography |
| Gioka et al 2005 | Chemically cured and light-cured adhesive resin | Monomers Bis-GMA and TEGDMA | Standardized volume of adhesive | 20 s for each tooth for the light-cured adhesive | None | Saline solution, 37°C | 2 mo | Liquid chromatography |
| Watanabe 2004 | Polycarbonate brackets | BPA | 12 brackets retrieved from patients 2 pieces of polycarbonate bracket |
N/A | None | Chloroform and methanol Water at 37°C |
18, 25, 30, and 40 mo for patients’ brackets 12,16, 25 and 34 mo |
Liquid chromatography |
| Watanabe et al 2001 | Polycarbonate brackets | BPA | 5 brackets retrieved from patients New polycarbonate brackets |
N/A | None | Chloroform and methanol Water at 37°C and 60°C |
5, 7, 8, and 15 mo for brackets retrieved from patients 3, 6, 9, and 12 mo at 37°C 1, 2, 3, and 4 days and 1, 2, 4, 6, 10, and 14 weeks at 60°C |
Liquid chromatography |
| Suzuki et al 2000 | Polycarbonate brackets | BPA | 50-100 μg of specimens in blocks or powder | N/A | None | Unionized water and 95% ethanol | 1 and 5 h at 37°C | Liquid chromatography |
| Thompsonet al 1982 | Orthodontic bonding resin | Monomers EGDMA and Bis-GMA | 110-119 mg of material and standard doses for bonding brackets | According to the manufacturer’s instruction | None | Distilled water alone, with 50% EtOH, with 5% EtOH with 0.2 M citrate, all at 25°C | 48 h | Spectrometry |
| Eliades et al 1995 | Chemically cured and light-cured adhesive resin | Monomers Bis-GMA and TEGDMA | 10 mg of light-cured adhesive per bracket | 20 s direct through the bracket or 2 × 10 s indirect from the incisal and cervical edges | 5 brackets bonded to chemically cured adhesive | Nonagitated 75% ethanol/25% water solution at 37°C ± 1°C | 15 d | Liquid chromatography |
| Wendl et al 2009 | Self-etching primer | Phosphoric acid ester | Various, weighted specimens | Several times of light curing or no curing | None | Physiologic saline solution | 1, 2, 4, and 6 h | Liquid chromatography |
| Substance under investigation | Highest amount of substance traced | Amount of substance in control | |
|---|---|---|---|
| Kang et al 2011 | BPA | 5.042 ng/mL (saliva, immediately after retainer placement) | 0.461 ng/mL (saliva, before placement) |
| Sunitha et al 2011 | BPA | 65.67 ppm (day 7, curing distance, 10 mm) | None used |
| Eliades et al 2011 | BPA | 2.9 μg/L (30-d group) | 0.16 μg/L |
| Eliades et al 2007 | BPA | No trace of BPA | None used |
| Gioka et al 2005 | Bis-GMA and TEGDMA | No measurable quantities of Bis-GMA 13.2 ppm TEGDMA |
None used |
| Watanabe 2004 | BPA | 697 μg/g after 40 mo (brackets retrieved from patients) 472 μg/g after 34 mo (immersion experiment in water) |
None used |
| Watanabe et al 2001 | BPA | 102 μg/g after 7 months (bracket retrieved from patient) 132 μg/g after 12 months (immersion experiment at 37°C) 434 μg/g after 14 weeks (immersion experiment at 60°C) |
None used |
| Suzuki et al 2000 | BPA | 22.24 μg/g in ethanol solution for crushed brackets | None used |
| Thompson et al 1982 | Bis-GMA | “Significant amount leached” | None used |
| Eliades et al 1995 | Bis-GMA and TEGDMA | 174 mg/10 μL of TEGDMA (metallic bracket bonded to chemically cured adhesive) 61.6 mg/10 μL of Bis-GMA (metallic bracket bonded to light-cured adhesive) |
Results of chemically cured adhesive group |
| Wendl et al 2009 | Phosphoric acid ester | “Following bracket bonding with SEPs and standard light curing, half of the phosphoric acid ester remains active and is rapidly released into saliva” | None used |
In the category of orthodontic adhesives, in-vivo studies, and BPA release, only 1 study was identified. The in-vivo assessment of BPA released from orthodontic adhesives was assessed in the recent study by Kang et al. They evaluated the changes in BPA levels in saliva and urine before and after placing a lingual bonded retainer on the mandibular dentitions of 2 volunteers. Samples were obtained immediately before placement of the retainer and 30 minutes, 1 day, 1 week, and 1 month after placement. The only significant level of BPA was detected in the saliva collected immediately after lingual retainer placement. Age and sex of the volunteers did not seem to affect the BPA level in the saliva or urine. The salivary BPA level (mean, 5.04 ng/mL; range of levels, 0.85-20.88 ng/mL) observed in the immediately collected sample was, as implied by the authors, far lower than the reference daily intake dose. Nevertheless, they concluded that, since there was some evidence of “low-dose effect,” clinicians should reduce the uncured layer of the material, using pumice surface prophylaxis of the adhesive.
Three studies of orthodontic adhesives, in-vitro studies, and BPA release were identified. These studies are contradictory with respect to the qualitative and quantitative parameters of BPA release from adhesives, probably because of their varying methodologies. In 2 studies, the adhesive was used during bracket bonding; in the third one, it was used to bond a lingual retainer.
The first article investigated BPA release from orthodontic adhesives after artificial aging in vitro. A chemically cured, no-mix adhesive and a light-cured adhesive were used for bonding. Forty stainless steel brackets were equally divided into 2 groups. The first group was bonded with the no-mix adhesive, and the second group with the visible light-cured adhesive. The results showed no indication of BPA for either type of adhesive across all time intervals in the study: 1 day, and 1, 3, and 5 weeks. The authors noted that, although the lack of BPA release was demonstrated in a particularly severe environment and under artificial accelerated aging conditions, these results should not be unquestionably extrapolated to actual clinical conditions. The main reason is that intraoral aging involves complex mechanical and chemical effectors, such as esterases, that might induce degradation.
Similar protocols and techniques for assessing BPA release with the previous research were also used in a recent in-vitro study by Sunitha et al. The scope of this study was to assess the BPA released from an orthodontic adhesive by varying the light cure tip distance and by assessing the degree of conversion of the adhesives, since the degree of conversion has been found to significantly affect the physical, mechanical, and biologic properties of the material. The outcomes of the study indicated that an increase of the distance between the light-cure tip and the adhesive introduced a decrease in the degree of conversion of the substance, which led to a greater BPA release.
The release of BPA from an orthodontic adhesive used to bond lingual fixed retainers on the surface of teeth was studied by Eliades et al. Eighteen recently extracted teeth, divided into 3 groups of 6 teeth each, were used for this study. A light-cured adhesive was bonded to a twist-flex wire adjusted to the lingual surface of the teeth. The results showed measurable amounts of BPA for all groups, with the highest found in the immersion media of the 30-days groups: 2.9 mg per liter. The control group, which consisted of teeth maintained in immersion media, showed BPA in the range of 0.16 mg per liter. The authors concluded that the exposure of the adhesive (increased thickness, influence of oral conditions, location of retainer) might play a pivotal role in releasing BPA, in contrast to studies in which the adhesive was used to bond brackets (exposure through peripheral margins of the bracket).
No study was identified in the category of orthodontic adhesives, in-vivo studies, and leaching of monomers.
Four studies were identified in the category of orthodontic adhesives, in-vitro studies, leaching of monomers. The first, by Thompson et al, assessed the release of Bis-GMA and ethyleneglycol dimethacrylate from an orthodontic bonding resin. The authors reported that significant amounts of residual unpolymerized material in cured orthodontic bonding resins from remaining in set orthodontic bonding resins were leached out in various solutions, such as saliva, water, soda water, and ethanol.
The second study, by Eliades et al, assessed the release of Bis-GMA and TEGDMA from adhesives used in bonding different types of brackets and concluded that both polymers leach in the incubation medium. An interesting finding of this study was that the polycarbonate base ceramic bracket released significantly higher amounts of monomers compared with ceramic brackets; evidence of polycarbonate base degradation was also found. The authors also concluded that the chemically cured resin might have an inherent disadvantage that can lead to relatively high residual TEGDMA and Bis-GMA concentrations eluted. It was recommended to apply indirect irradiation (around the bracket edges) instead of direct irradiation (through the bracket).
The third study, by Gioka et al, assessed the release of Bis-GMA and TEGDMA from a light-cured and a chemically cured adhesive. No measurable quantities of Bis-GMA were released, and the higher amount of TEGDMA traced was in the solution of the chemically cured adhesive.
The fourth study, from Wendl et al, assessed the release of phosphoric acid ester from the self-etching primer. The results indicated that conventional curing led to the leaching of almost 50% of the phosphoric acid ester in the self-etching primer. The authors suggested that light curing before and after application of the adhesive, longer curing times, and mouth-rinsing during the first hour after bracket bonding could help prevent patients’ exposure to this potential hazard.
In the category of polycarbonate brackets, in-vitro studies, and BPA release, Suzuki et al described BPA release from 4 orthodontic polycarbonate brackets. Analysis of the total and released amounts of BPA indicated that, during the synthesis of polycarbonates, nonreacting BPA probably remains inside the materials and is released when they are immersed in water or organic solvents.
Watanabe et al investigated the change in the BPA content in a polycarbonate orthodontic bracket and its leaching characteristics during incubation in water and found that the rate of BPA release increased with time and temperature.
A consequent study by Watanabe validated the previous findings. This study was conducted mainly in vitro but also included brackets retrieved from 3 patients’ oral cavities. The results showed a linear relationship between the cumulative amount of BPA eluted into water and the time of intraoral service of polycarbonate brackets; BPA eluted increased linearly from 12 to 34 months.
In the category of polycarbonate brackets, in-vivo studies, and BPA release, Watanabe et al investigated the BPA in the polycarbonate brackets retrieved from patients and attempted to clarify whether the BPA content might change in the oral cavity. The findings suggested that polycarbonate brackets degrade in the oral cavity and release BPA. Subsequent research by Watanabe demonstrated that BPA release from the brackets in the oral cavity was higher relative to the expected amount based on in-vitro investigations.
No study was identified in the category of polycarbonate brackets, in-vivo studies, and leaching of monomers.
No study was identified in the category of polycarbonate brackets, in-vitro studies, and leaching of monomers.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


