Peripheral nerve injury and regeneration in the maxillofacial region remain significant clinical problems. Peripheral nerves, including the inferior alveolar and lingual nerves, possess a limited ability to regenerate after traumatic injury. The quality of this regeneration depends on several factors. These include the size and type of injury, location, and the age of the patient. Healing after nerve trauma is complicated, partly because mature neurons do not replicate. Under the right conditions, however, axons can regenerate across relatively long nerve gaps generated due to injury or resection of tumors, reconnecting with distal stump and eventually reestablishing functional contacts. Those nerves that do not spontaneously restore their function require microsurgical end-to-end coaptation of proximal and distal nerve segments to produce a tension free repair. When surgical repair is required for a transected nerve, or a neuroma requiring excision, higher success rates have been seen with direct repair rather than through the use of grafts. Better results are obtained when the nerves are purely motor or purely sensory and when the amount of intraneural connective tissue is relatively small. For optimal nerve regeneration after repair, nerve stumps must be properly prepared, aligned without tension, and repaired atraumatically with minimal tissue damage and minimal number of sutures. When primary repair cannot be performed without undue tension, nerve grafting or tubulization techniques are required.
The current gold standard for bridging nerve gaps is nerve autografting. Autologous nerve grafts fulfill the criteria for an ideal nerve conduit, because they provide a permissive and stimulating scaffold, including Schwann cell basal laminae, neurotrophic factors, and adhesion molecules. The disadvantages of this technique include donor site morbidity, size mismatch between injured nerve and graft nerve diameter, neuroma formation at the donor site, and even in the best-case scenarios, incomplete recovery. Increasing evidence suggests that the modality of the donor nerve may influence regeneration, with mixed nerves or purely motor donor nerves having better outcomes than commonly used sensory nerves such as sural nerves. In addition, peripheral nerves might also express inhibitory proteoglycans such as chondroitin sulfate proteoglycans.
These limitations of autografting have encouraged the search for alternative means of nerve gap reconstruction. These have included nerve allografts and biologic and artificial conduits. Achieving better clinical outcomes in the future depends greatly upon simultaneous advancements in microsurgical technique, but perhaps more importantly, translation of molecular biology and bioengineering discoveries into clinical practice. The field of peripheral nerve research is a dynamically developing arena, and recent concentration has focused on sophisticated approaches tested at the basic science level. To date, much of the research effort has focused on nerve guidance channels (NGCs) to enhance regeneration across nerve gaps. The purpose of this article is to illustrate recent advances in artificial NGCs and discuss the variables that may be manipulated to enhance the efficacy of scaffolds designed for peripheral nerve regeneration, which may have a dramatic clinical impact on peripheral trigeminal nerve injury in the future.
Artificial conduits
Over the past 25 years the concept of an NGC has evolved from a tool to investigate nerve regeneration to a device that is being used clinically in patients as an alternative to autografts. Their use has been mostly limited to the repair of small defects (<3 cm) in small-caliber digital nerves. There have been reports of use in the maxillofacial region, but few outcome studies at present. Initial clinical trigeminal nerve case series showed poor outcomes with the use of Gore-Tex conduits for inferior alveolar and lingual nerve repair. Possible improvement upon this nonresorbable material with the use of a polyglycolic acid conduit filled with heparin has been proposed. Presently there are several tubes being marketed including Neurotube (Synovis), Neurolac (Ascension), SaluBridge (SaluMedica), and NeuraGen (Integra). While these NGCs have been shown to enhance regeneration across nerve gaps when compared with no intervention, guidance channels rarely approach the performance of autografts when the gaps are 10 mm or longer in a rat model. Although approved for human use, the efficacy for each is limited to the repair of short defects (<3 cm) of small-caliber nerves. The basic design of these tubes has been similar, namely a hollow tube in which the two ends of the nerve are inserted at either end. They differ, however, in the composition and properties of the biomaterial from which they are made. Currently, there is little information as to which tube provides better clinical outcomes in the repair of small nerve gaps.
In the last decade, artificial NGCs have been produced using various natural (collagen) and synthetic, nonbiodegradable polymers (silicone) and biodegradable polymers, including poly lactic acid and polycaprolactone (PGA, PCL, hydrogel). Multiple modification techniques have been developed to obtain porous and nonporous NGCs, incorporate electrically active channels into the channels, or utilize bioactive fillers within the lumen of the NGCs. The current trend in peripheral nerve research and tissue engineering is the realization of biomimetic NGCs providing topological, haptotactic, and chemotactic signaling to cells, respectively by surface functionalization with cell binding domains, the use of internal oriented fibers and the sustained release of neurotrophic factors.
Generally, NGCs can be categorized by whether they are degradable or nondegradable, and by the nature of additional cues that are presented within the lumens of NGCs to promote axonal regeneration.
Nonbiodegradable Materials as NGCs
Silicone NGCs have been used historically for peripheral nerve repair. They have been widely employed to study NGC fillers, as they are nonbiodegradable and not permeable to large molecules. Disadvantages of the use of nondegradable NGCs include chronic foreign body reaction, inflexibility, and lack of stability. Particularly, the inflammatory response of silicone NGCs may lead to fibrotic capsule formation around the guide and consequent nerve compression. Alterations in the blood–nerve barrier occur, followed by demyelination of the nerve fibers. Silicone tubes used as NGCs clinically must be removed for a successful outcome.
Biodegradable Synthetic Materials as NGCs
Recent research has been focused on biodegradable NGCs. Biodegradable NGCs should ideally not be toxic or elicit an immunologic response and be able to bear the stresses of the surgical procedure (handling and suturing) and implantation time (due to patient movements). Various different synthetic biomaterials have been used in the fabrication of NGCs, mostly polymers of lactic and glycolic acid and capralactone through various fabrication techniques. Both the material composition and the fabrication technique have great influence on the physical properties of the NGC for entubulation repair; these properties include permeability, swelling, and degradation behavior.
Permeability of a NGC is an important property, as nutrients and oxygen need to diffuse to the site of regeneration before revascularization and may influence the formation of fibrin matrix in the initial stage of regeneration. It should allow for the exchange of fluids between the regenerating environment and avoid the build-up of pressure due to fluid retention. At the same time, the pore dimension has to be small enough to prevent infiltration of scar-forming fibroblasts that could alter the regeneration process. Different fabrication techniques can make NGCs permeable including cutting holes in the wall of the tube, rolling of meshes, adding salt or sugar crystals, which are leached after fabrication, or by injection molding solvent evaporation. The permeability of an NGC can be closely related to the crystallinity of the material. As the crystalline phase is inaccessible to water and other permeable molecules, both biodegradation and permeability decrease with increasing crystallinity degree. Permeability also depends upon the hydrophilic properties of the material, which can be measured from the contact angle of a drop of water on the material. It is not known whether the extended regeneration with the use of permeable chambers is due to:
-
Metabolic exchange across the tube wall (diffusion of nutrients such as glucose, oxygen, and elimination of waste products)
-
Diffusion into the guide lumen of growth or trophic factors generated in the external environment (wound healing factors)
-
Retention of growth or trophic factors secreted by the nerve stumps
-
A combination of the previously mentioned factors.
Swelling and degradation characteristics are important inter-related physical properties of a NGC also. Materials selected for the production of NGCs should be slowly degradable into biocompatible products and have a low degree of swelling during degradation. Swelling of the NGC may occlude the lumen for regeneration or cause compression of the regenerated axons. The rate of degradation may contribute to the swelling of the NGC through the formation of breakdown products that increase the osmotic pressure within the conduit. The NGC should remain intact for the time it takes axons to regenerate across the gap and then degrade gradually with minimal swelling and foreign body reaction. This degradation rate is optimized by the copolymer ratio, in an analogous fashion to resorbable fixation plates.
Intraluminal Modifications of Single-Lumen Tubes to Enhance Regeneration
Several modifications to the single lumen hallow NGC have been investigated. These modifications are necessary when large nerve gaps exist which will exceed the inherent regenerative capacity of the peripheral nerve. When nerve gaps are short, and inherent regeneration is possible, a fibrin cable forms across the nerve gap, allowing for Schwann cell infiltration and the formation of the Bands of Bungner, which are arrays of Schwann cells and their interdigitating processes within a space circumscribed by the basement membrane (Schwann tube). Regenerating fibers then enter the gap and follow these Bands of Bungner, reach the distal end of the severed nerve, enter it, and go on to re-innervate the original target ( Fig. 1 ). During the embryonic development of the nervous system, the developing axons are stimulated by various haptotactic (contact mediated) and chemotactic (diffusible) cues that guide growth toward their targets. Some of these signals are naturally present in a gradient concentration. Similar cues have been identified during the spontaneously occurring axon regeneration response after axonotomesis injuries. When nerve gaps are large, however, the formation of the fibrin cable and the Bands of Bungner is compromised, necessitating exogenous support to enable the regenerating fibers to cross the large nerve gap (>10 mm in rat animal models).
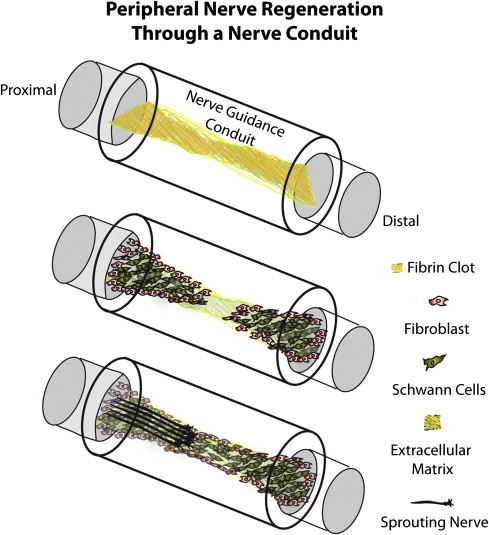
In attempts to maximize the regenerative capacity over larger peripheral nerve gaps, several research groups have implanted natural or synthetic materials, cells, microfibers, nanofibers, or Schwann cells seeded in matrigel to enhance the regeneration across in vitro peripheral nerve gaps. There are four essential components that are typically introduced intraluminally in NGCs to enhance NGC’s outcomes.
These components are:
-
Growth-permissive substrates (hydrogels or nano/micro fibers)
-
Neurostimulatory extracellular matrix (ECM) proteins or peptides (laminin [LN-1] or LN-1 fragments)
-
Neurotrophic factors (bFGF, NGF, or BDNF)
-
Schwann cells/glial cells/stem cells.
The introduction of these factors in NGCs can provide haptotactic and chemotactic cues that enhance axonal regeneration.
Growth-permissive substrates
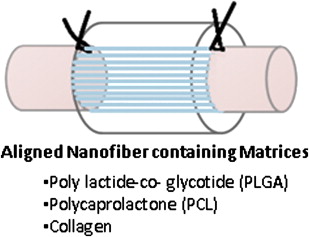
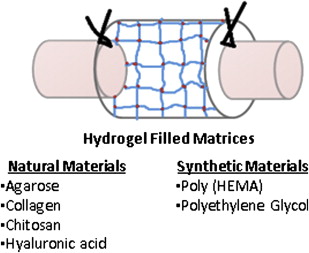
Growth-permissive substrates for NGCs may include intrinsic scaffolds ( Fig. 2 ) or extracellular matrix containing gels ( Fig. 3 ). Intrinsic scaffolds such as filaments, sponges, and multichannel nerve tubes enhance regeneration by stabilizing the fibrin matrix that is formed inside the NGC through contact guidance. The internal structure increases the intraluminal surface area and potentially concentrates growth-promoting physical and biochemical cues by facilitating endogenous Schwann cell function or facilitating local release of incorporated growth factors. However, the additional intraluminal structures may negatively influence the NGC’s properties either by affecting its physical properties such as permeability or flexibility or by reducing the total open intraluminal cross-sectional area available for nerve regeneration. Therefore, the number of filaments and channels that can be put into a NGC is limited by the size (how small) at which they can be produced. Longitudinally oriented structures, such as filaments, may be used to mimic the fascicular pattern of the nerve, which consists of an intraneural plexus. This has led many researchers to pursue development of three-dimensional gels and scaffolds (collagen/laminin-containing gels).

Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


