Chapter 9 Implant Body Size: A Biomechanical and Esthetic Rationale
The initial treatment plan for implant dentistry should include the ideal implant size, based primarily on biomechanic and esthetic considerations. In traditional prosthetics, when a tooth is replaced, the abutment teeth are already provided by nature with wide posterior abutments for posterior teeth. When teeth are replaced with dental implants, the implant team should preselect the ideal abutment size, based on the ideal size for an esthetic restoration within biomechanical guidelines.
The size of an implant used to be determined primarily by the existing bone volume in height, width, and length. The surgeon would select longer implants in the anterior regions of the mouth and shorter ones in the posterior areas because of the limits of the mandibular canal and maxillary sinus. The width of the implant, also determined at surgery, would relate to the existing width of available bone, and one diameter implant (4 mm) would be used in most all situations.1
Over the years, dental implant treatment plans incorporating biomechanics have been advocated by Misch to decrease the most common complications–those related to stress.2 The prosthesis first is planned, including whether the restoration is fixed or removable, how many teeth are replaced, and the esthetic demands. The patient force factors are then considered to evaluate the magnitude and type of force applied to the restoration. The bone density is evaluated in the regions of the potential implant placement. The key implant positions and the implant number are then selected in relation to the patient force factors and the bone density in the implant sites. For example, when the patient has parafunction or the bone is less dense or when a cantilever is present, the greater force exerted on the implant abutments will transmit greater stresses to the implant-bone interface. The next consideration in this ideal treatment plan sequence is the implant size.1
Dental implants function to transfer loads to surrounding biological tissues.3,4 Biomechanical load management is dependent on two factors: the character of the applied force and the functional surface area over which the load is dissipated.5 The implant size directly affects the functional surface area that distributes a load transferred through the prosthesis. A comprehensive approach to the overall dental implant size begins with the identification of clinical problems to be addressed. Fundamental scientific principles related to force and surface area are then combined with engineering principles to pursue the desired clinical goals. When relevant, esthetic considerations in regards to implant size are part of the evaluation. This chapter will build on and apply basic biomechanics and demonstrate how these principles also relate to the ideal dental implant size to support a prosthetic load. The esthetic guidelines related to implant size will also be addressed.
CHARACTER OF FORCES APPLIED TO DENTAL IMPLANTS
Stress and Strain
The presence of fibrous tissue has long been known to decrease the long-term survival of a root form implant.1 Excessive loads on an osteointegrated implant may result in mobility of the supporting device, even after a favorable bone-implant interface has been obtained.6 Although several conditions may cause crestal bone loss, one of these may be prosthetic overload.7 Excessive loads on the bone result in increased strain conditions in the bone.8 These microstrains on the bone may affect the bone remodeling rate and cause pathologic overload, which results in the loss of bone. The amount of bone strain is directly related to the amount of stress applied to the implant-bone interface. The greater the stresses throughout the implant-bone interface, the greater the risk factor for crestal bone loss and subsequent implant failure.9 Therefore the stress and strain relationship has been shown to be an important parameter for crestal bone maintenance and implant survival.
Forces applied to dental implants may be characterized in terms of five distinct (although related) factors: magnitude, duration, type, direction, and magnification.5 Each factor should be considered within the physiologic constraints on implant size. In addition, there are surgical and prosthetic considerations related to implant size. For example, the increase in stress to an implant body also increases the risk of abutment screw loosening or implant body fracture. As a result, a relative risk factor for different implant sizes may be established.
Force Magnitude
The physiology of the stomatognathic system imposes a range on the magnitude of forces that may be applied to an implant in the oral environment. The magnitude of bite force varies as a function of anatomical region and state of the dentition.10 Average bite forces can range from 10 to 350 lb. The magnitude of force is greater in the molar region (200 lb), less in the canine area (100 lb), and least in the anterior incisor region (25 to 35 lb).11 These average bite forces increase with parafunction to magnitudes that may approach 1000 lb in the posterior regions.12
After sustained periods of edentulism, the bone foundation often becomes less dense. Studies on dentate and edentulous jaws illustrate greater trabecular bone density in the anterior regions compared with the premolar or molar regions.13 The bone’s ultimate strength is highly dependent on its density.14 As such, less dense bone may no longer be able to support normal physiologic bite forces on a dental implant. Careful treatment planning, including appropriate implant size selection, is imperative to lower the magnitude of loads imposed on the vulnerable implant-bone interface under these less ideal conditions. Thus the posterior regions with higher bite forces and lower bone densities should use a different parameter for implant size compared with the anterior regions. In addition, the anterior regions are more often in the esthetic zone and may influence the size of the implant as a consequence.
Force Duration
The duration of bite forces on the dentition has a wide range. Under ideal conditions, the teeth come together during swallowing and eating for only brief contacts. The total time of those brief episodes is less than 30 minutes per day. Patients who exhibit bruxism, clenching, or other parafunctional habits, however, may have their teeth in contact several hours each day.12 Fatigue fractures increase in direct relationship to the amount of the force and the number of cycles of load. Therefore an increase in force duration directly increases the risk of fatigue load to the implant body when the force is higher than the endurance limit of these entities.15 The implant body width is directly related to the strength of an implant, and wider diameter implants reduce the risk of fatigue fracture.
The duration of a force may also alter the implant-bone interface. Fatigue damage to cortical bone has been reported under relatively high-frequency loading rates (e.g., shin splints in runners).16 Although fatigue damage to alveolar bone has not yet been reported in the literature, it is unlikely the alveolar bone reacts differently to parafunctional loads. Roberts et al. report the bone around an implant may be remodeled at a rate of 500% each year after loading, compared with normal trabecular physiologic remodeling around a tooth of 20% to 40% per year.17 The dramatic increase in remodeling rates may eventually lead to fatigue damage and resultant bone loss.18
Force Type
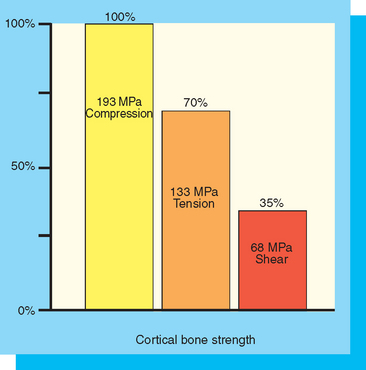
Three types of forces may be imposed on dental implants within the oral environment: compression, tension, and shear. Bone is strongest when loaded in compression, 30% weaker when subjected to tensile forces, and 65% weaker when loaded in shear (Figure 9-1).19 Therefore an attempt should be made to limit shear forces on bone, because it is least resistant to fracture under these loading conditions. This is most important in regions of decreased bone density, because the strength of bone is also directly related to its density.14 An increased width of an implant may decrease offset loads and increase the amount of the implant-bone interface placed under compressive loads.
Force Direction
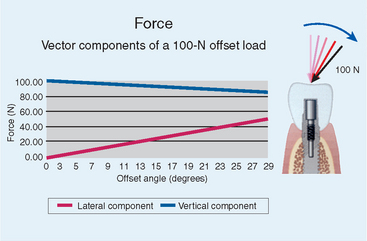
The forces to an implant body are typically greatest at the crestal bone interface.20 Angled loads to the implant prosthesis produce angled loads to the crest module of the implant. The implant angulation is important to consider. For example, given an occlusal load with a 25-lb magnitude, the direction of the load has a significant effect on the magnitude of compressive and lateral load components. By increasing the angle of the load by only 6 degrees, the lateral component of that load is increased by 233%. In addition, every degree of angled load greatly increases the damaging shear load component experienced by the implants, which is the most damaging component of the load (Figure 9-2).
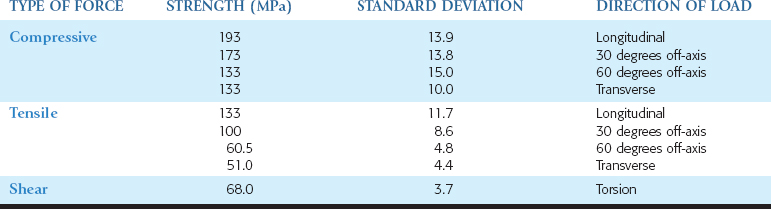
Angled loads increase the amount of shear loads to the bone, and the bone is weakest to shear-type loads21 (Table 9-1). Therefore, under ideal conditions, the implant body should be oriented to provide long axis compressive loads to the implant and to decrease shear loads to the crestal bone region. Occlusal loads in centric occlusion occur where the occlusal forces are usually the greatest. As such, the implant should be inserted perpendicular to the curve of Wilson and curve of Spee. Additionally, axial alignment places less stress on the overall implant system (i.e., abutment and abutment screw components) and decreases the risk of screw loosening and fatigue fractures of the implant or its components.
The anatomy of the mandible and maxilla places significant constraints on the ability to surgically place root form implants suitable for loading along their long axis. Bone undercuts further constrain implant placement and thus load direction imposed on the implant. Most all undercuts occur on the facial aspects of the bone, with the exception of the submandibular fossa in the posterior mandible.22 Therefore implant bodies are often angled to avoid perforation of the lingual/facial bony undercut during insertion. Resorptive patterns after prolonged edentulism also exacerbates the normally occurring angulation challenges.23
The maxillary anterior region does not permit an ideal implant position, even under ideal conditions. The natural maxillary anterior teeth are 12 to 15 degrees off the long axis of load, and the bone of the premaxilla is in a similar relationship after tooth loss. Therefore implants in this region are often positioned with a greater relative angle to occlusal loads than any other region.21 To decrease the effect of an angled load on the implant, the implant body may be increased in diameter.
Force Magnification
Force magnification increases the stress beyond the usual conditions of load; for example, a cantilevered prosthesis, a crown height greater than normal, or parafunction.5 Multiple force magnifiers, such as a patient with parafunctional habits and an excessive crown height, may exceed the capability of any dental implant to withstand occlusal loads. Careful treatment planning with special attention to the implant position, implant number, occlusal loading, and an increase in implant size to increase functional surface area is indicated when a clinical case presents the challenge of force magnifiers.
A magnifier of force around an individual implant is also affected by the density of bone. Four distinct bone density categories within the maxilla and mandible exhibit a broad range of biomechanical strengths (i.e., ability to withstand physiologic loads).13,14 Significantly increased clinical failure rates in poor-quality, porous bone compared with denser bone have been documented worldwide by multiple independent clinical investigators for more than a decade, with failure rates as high as 35% with implants in D4 (Type IV) bone.24–28 Bone density is directly related to bone strength, and D4 bone may be more than 10 times weaker than D1 bone and 70% weaker than D2 bone.14 Most implant failures in soft bone are from occlusal overload from a decrease in bone strength, a decreased bone-implant contact percentage, and the type of load transfer to the implant-bone interface on functional loading.14,29 Therefore the effect of a resultant force is magnified as to its clinical result when placed in softer bone types.
SURFACE AREA
Rationale for Longer Implant Length
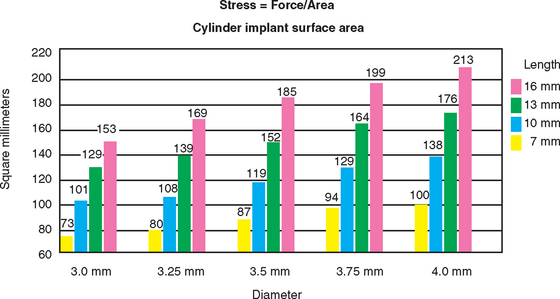
The length of the implant is directly related to the overall implant surface area, when all other variables are constant. A 10-mm–long cylinder implant increases surface area by approximately 30% over a 7-mm-long implant and has about 20% less surface area than 13-mm–long implants (Figure 9-3).30,31 As a result, a common axiom has been to place an implant as long as possible. The length of the implant used for prosthetic support often corresponds to the height of available bone in the edentulous site.
Conventional thinking suggests that longer implants provide maximum functional surface area in a healed implant interface. However, when the length axiom is reevaluated, several challenges ensue. The available bone height is greater in the anterior regions of the mouth, especially the anterior mandible. Yet the bite forces are lower and the bone density is greater in these regions, especially in the mandible. The posterior regions have less bone height and the implant cannot engage the dense opposing cortical plate, because it is either nonexistent (maxilla) or beyond anatomical limits (neurovascular canal in the mandible). The posterior maxilla often has less height than the mandible, and the bone is less dense. As a result, poorer survival rates are reported for posterior implants, with the posterior maxilla associated with the highest failure rate (Figure 9-4).
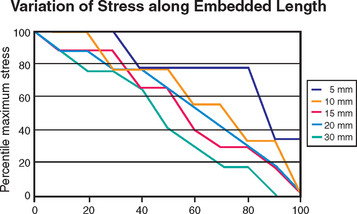
Implants longer than 15 mm have been suggested to provide greater stability under lateral loading conditions. However, reports suggest that increasing the length beyond a certain dimension may not reduce force transfer proportionately.20,32 Finite element analysis provides an analytical means to investigate the influence of implant length relative to functional surface area under such extreme loading conditions. Misch and Bidez placed cylindrical implants of different lengths in a computer bone model with ideal bone density and volume (see Chapter 11). The embedded implant body lengths were 5, 10, 15, 20, and 30 mm. A 10-mm crown height was designed above the level of bone, and a lateral force of 50 N was applied to the top of the 10-mm crown. For these loading conditions, the percentage of maximum stress was plotted against the percentage of embedded length (Figure 9-5). Peak stresses were not completely dissipated in the 5-mm-long implant model, with approximately 30% of the maximum stress still present at the apex of the implant. The 5-mm implant model, therefore, did not provide sufficient length for lateral force dissipation in spite of good bone density and volume. In the 10-mm implant model, 80% of maximum stress was dissipated in approximately 95% of the embedded length. For the 15- and 20-mm lengths, 80% of the maximum stress was dissipated in approximately 90% of the embedded length. For the 30-mm length, 80% of the maximum stress was dissipated in approximately 70% of the embedded length. Therefore the length of the implant in favorable bone quality and crown height may range from 10 to 15 mm, and 12 mm is usually ideal. All implant lengths exhibited 80% to 100% of the stress in the crestal 40% of the implant length. The results of this analysis point to the fact that the majority of the maximum stress generated by a lateral load can be dissipated as well by implants in the range of 10 to 15 mm in length, compared with implants in the range of 20 to 30 mm in length. In addition, the highest stresses were observed in the crestal bone regions, regardless of the implant length. This biomechanical analysis supports the opinion that longer implants are not necessarily better in good bone volume and density, such as that found in the anterior regions of the mandible. An implant length of 12 mm is usually ideal under most patient force and bone density conditions and up to 15 mm is suggested in softer bone types.
Bicortical stabilization, a rationale often cited for longer implants, is not needed in D1 bone, which is primarily homogenous cortical bone.13 Because bone overheating is a primary cause of surgical failure, attempting to engage the opposing cortical plate and preparing a longer osteotomy may result in overheating the bone when it is D1 or D2.33 A threaded implant may not readily engage the denser bone of the apical cortical plate, and the implant threads may strip along the rest of the osteotomy, especially if D3 or D4 trabecular bone types. In addition, after the implant-bone interface is formed, excessively long implants do not transfer stress to the apical region, because most of the stresses are transmitted within the crestal 7 to 9 mm of bone (except in the softest bone types) and therefore are often not needed.20 A further analysis relative to implant length is suggested, especially in good-quality bone.
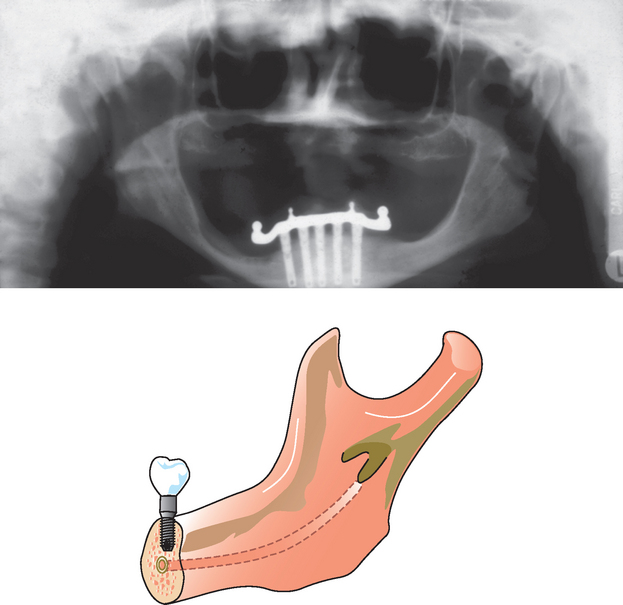
D3 and D4 bone are primarily present in the posterior regions of the jaw, where less available bone height is observed compared with the anterior regions. Nerve repositioning is cited as an acceptable clinical treatment to facilitate placement of longer implants in the posterior mandible. However, this advanced surgical procedure represents an increased risk for paresthesia and is often not indicated, especially when other implant options are available.34 To place the longest implants in the maxillary posterior regions, a sinus graft is often required. Sinus grafts have been shown to grow bone into the graft from the surrounding walls of bone, and bone does not form under the sinus mucosa. The apical end of longer implants will not benefit from the sinus bone graft procedure for some time, and the crest of the bone below the graft will remain at greater risk. Therefore increasing surface area primarily by length in the posterior regions of the jaws requires advanced grafting or nerve repositioning surgery and, in spite of this, does not benefit the primary regions of increased stress (i.e., the crestal bone region). However, implants from 12 to 15 mm in length are usually considered ideal for most situations.
Disadvantages of Short Implants
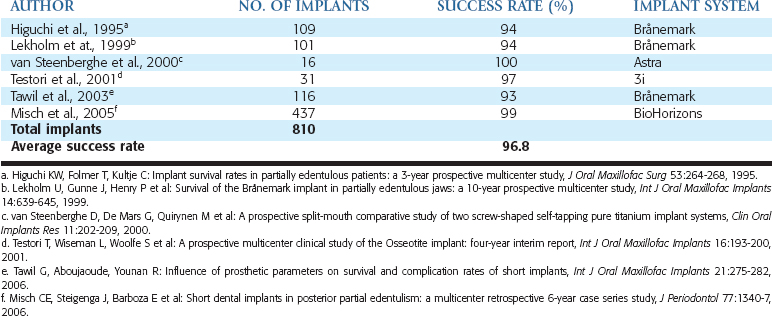
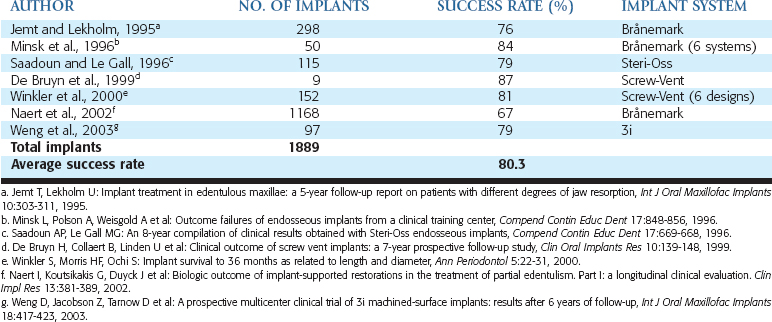
Different risk factors for implant longevity have emerged over the years. A review of the literature related to implant failure and implant length was published by Goodacre et al. in 2003.35 In the majority of articles addressing implant length, implants 10 mm or smaller have increased failure rates. In the reports summarized, the failure rates of short implants were 10%, compared with a 3% failure rate of longer implants. The failure is even more apparent when the literature reviews implants smaller than 10 mm in the posterior regions of partially edentulous patients. Less than half the clinical reports had survival rates higher than 90%, and more than half of the reports had implant failure higher than 19.7% (Tables 9-2 and 9-3).36–46
A review of several of the larger, multicenter reports of short implants is noteworthy. Minsk et al. reported the results of a training center in 1996, with 80 different operators using 6 different systems over a 6-year period.27 Implants 7 to 9 mm in length were reported to have a 16% failure rate. The overall survival rate of all longer lengths was 95%. Winkler et al. published a multicenter report over a 3-year period in 2000.37 The implant survival was directly related to the length of the implant. The 7-mm-long implants had a 26.4% failure rate, whereas 16-mm implants demonstrated only a 2.8% rate of failure. Implants of 8 mm had a 13% failure rate, whereas 10-mm implants in the report failed at a rate of 10.9%, and the 13-mm implants failed 5.7% of the time.
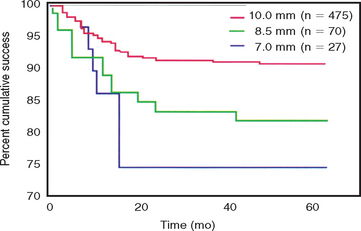
A multicenter report by Weng et al. in 2003 found 60% of all failed implants were 10 mm or less in length.38 The overall failure rate of all implants in the study was 9%, yet the 7-mm implant failed 26% of the time and the 8.5-mm implant had a 19% failure rate39 (Figure 9-6). Naert et al. reported in the literature on clinical outcomes of short dental implants.6 Implants shorter than 10 mm had a survival rate average of 81.5%, whereas longer implants had a survival rate higher than 95%.
It should be noted the failure rates in most all these reports are not surgical failures or failures to osseointegrate. The failures associated with short implants often occurred after prosthetic loading. In other words, the surgical success was not affected by implant length. However, after the prostheses were in function, an increase in early loading failure was observed, especially within the first 12 to 18 months.
When an implant of 12 mm or more is unavailable without advanced surgical procedures to gain height, a shorter implant may be considered. However, four reasons linked to biomechanics may explain why the posterior short implant may have a higher failure rate after loading compared with longer implants48:
IDEAL IMPLANT SIZE
To obtain predictable success in situations with most patient force factors or bone densities, there is a minimum implant length, depending on the implant body width and the implant design49 (Figure 9-7). The softer the bone, the greater the implant body length and diameter suggested50. The literature observes a wide range of implant studies reporting higher implant survival rates when the implant is at least 12 mm in length. Shorter implants usually increase the risk of failure and therefore are not initially treatment planned as a first option. Rather than establish an ideal treatment plan that may be deficient in surface area, the minimum length of an implant for an unloaded healing protocol is usually 12 mm or more for most implant designs. Therefore, after the ideal treatment plan determines the key implant positions and implant number, the implant length selected for most treatment plan options is at least 12 mm long.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses