Chapter 7
Sterilization and disinfection of dental instruments
DECONTAMINATION CYCLE
Decontamination is carried out for two important reasons:
- To make a reusable device safe for staff to handle
- To minimise (disinfection) or eliminate (sterilization) the risk of crossinfection from person to person by direct contact
The ultimate goal is to produce a sterile instrument that is completely free of all microorganisms. Sterilization is not sufficient on its own to achieve this aim. Instruments cannot be sterilised unless they are first cleaned because debris (cement, blood, lubricant oil etc.) adhering to the surface of an instrument can inhibit or interfere with the sterilization process. Therefore, decontamination is an incremental process with each stage (see Figure 7.1) contributing to the killing and removal of microorganisms.
Definitions:
- Cleaning: physical removal (including prions) but not necessarily killing of microbes
- Disinfection: reduction of the microbial load to a level that makes the disinfected object safe to handle
- Sterilization: killing and removal of all microorganisms including bacterial spores
Vegetative (non-spore-forming) bacteria, fungi, viruses and protozoa are readily killed at temperatures above 60–65°C. Spore-forming bacteria and Mycobacteria spp. have protective cell walls that make them relatively resistant to thermal killing. To destroy the organisms, the higher temperatures generated during sterilization are necessary, whereas prions are highly resistant to thermal killing and are mainly removed during cleaning and disinfection. Complete removal of bacteria is an essential component of the process, as endotoxin a breakdown product derived from the cell wall of dead Gram-negative bacteria, though not infectious, can trigger an inflammatory reaction that ranges from mild to sometimes fatal toxic shock. In addition to removal of endotoxin, a decontaminated instrument whether it has been disinfected or sterilised should be free from:
Figure 7.1 Decontamination cycle of reusable instruments (courtesy of Paul Morris).
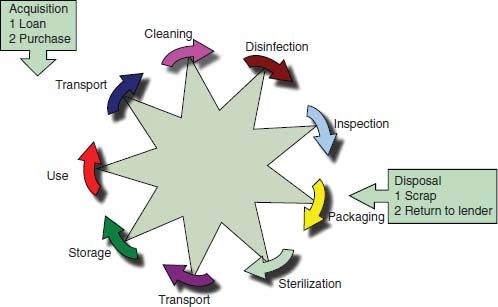
- Residues of previous clinical treatment, e.g. cement, microbes, blood and human tissue
- Residues from the decontamination process, e.g. disinfectants and limescale
- Contamination by dust, dirt and environmental microbial contamination
WHY HAS CLEANING BECOME SO IMPORTANT?
Cleaning has always been an important component of the decontamination process, but has increased in significance as a result of the prion disease vCJD (variant Creutzfeldt–Jakob disease) appearing in the humans in the mid-1990s (see Chapter 2). Prion diseases are rare, infectious, fatal, neurodegenerative disorders, which present as sporadic, familial or acquired disease. The infectious agents are prions. Unlike other pathogens, they contain no genetic material in the form of DNA or RNA, but instead are composed of a protein with an abnormal conformation. Proteins are considerably more resistant to the thermal denaturation than DNA or RNA. Sterilization reduces infectivity (i.e. deactivation of prion agents) only by a 100- to 1000-fold. There is evidence to suggest that prions are modified by adsorption onto a metal surface making them more resistant to decontamination. In most infections, the organism has to be released from the surface of a surgical instrument into the patient’s bloodstream or tissues to initiate an infection. This sequence of events is unnecessary for transmission of prion disease, as infection can occur even when prion agent remains adherent to the instrument. Once present on an instrument, the prion can continue to infect patients until it is physically removed.
At present, there are no modifications to the sterilization processes available for routine use, which can inactivate prion protein without having a detrimental impact on instrument integrity. Radiofrequency-generated gas plasma and enzymic disinfectants, which are undergoing experimental evaluation, look promising and may in the future be employed to deactivate prions. Currently, cleaning which physically removes protein has become of prime importance in the decontamination cycle. Previously, the benchmark standard for cleaning was ‘the reduction of bacterial load’, and now it has been raised to the ‘removal of prion proteins’. On a practical level, this change is translated into the replacement of manual cleaning by automated cleaning, preferably with a thermal washer disinfector. However dedicated the person cleaning the dental instruments, all manual procedures are subject to operator variation as a result of time constraints or tiredness. Automated cleaning has the major advantage of being reproducible and open to validation.
LEGAL REQUIREMENTS AND TECHNICAL STANDARDS FOR DECONTAMINATION
Complying with the law
Sterilisers and thermal washer disinfectors are classed under the law as medical devices. Both the equipment and the site where decontamination takes place are required to operate according to EEC Medical Devices Directive (MDD) 93/42/EEC. In the UK, the directive is translated into the Medical Devices Regulations (MDR) 2002 under the Consumer Protection Act. The regulations apply specifically to large sterile services departments supplying hospitals with sterile reusable instruments. But under the Health Act 2006 (Hygiene Code – see Chapter 1), the basic principles affecting control of the decontamination process and the working environment apply to all health care facilities including dental practices (see Table 7.1).
Table 7.1 Hygiene code
Summary of regulations affecting reusable instrument decontamination:
|
Technical standards for decontamination
European technical standards for sterilisers and washer disinfectors were recently revised and upgraded. In the UK, to reflect theses changes and to bring them into line with the new British/European standards, BS EN ISO 15883 (washer disinfectors) and BS EN ISO 13060 (benchtop sterilisers), all the technical and operating guidance was united into a single document called Decontamination Series HTM 01. Part 05 of the series ‘Decontamination in Dentistry’ governs activities in general dental practice (available from www.decontamination.nhs.uk), which is monitored by the Healthcare Commission, UK. They take the Hygiene Code as their benchmark for audit purposes (see Table 7.1). As dental practices like other health care organisations are required to take into account all relevant national guidelines, dental practice staff are obliged to be familiar with the content of these documents. In addition, they should follow the guidance on setting up a decontamination room and operating equipment such as sterilisers.
WHERE SHOULD INSTRUMENT DECONTAMINATION TAKE PLACE?
Currently, sterilization of equipment for use in dentistry takes place within the dental practice, but this is not the only option available.
Options:
Table 7.2 Key points for organising decontamination in the dental practice
|
Compiled from HTM 01-01 and HTM 01-05.
Most, but not all, dental practices will probably opt for option 4, i.e. to continue local decontamination combined with selective use of disposable instruments. As we have seen in the previous section, this puts the onus on the dental practice to comply with the national standards which are outlined in brief in Table 7.2. Note that validation of the decontamination cycle is considered as important as the design of the decontamination room and the type of equipment used within it. The next part of this chapter will outline how this can be achieved.
DESIGN OF DEDICATED DECONTAMINATION UNITS
Ideally, the treatment room should be used exclusively for clinical treatment and should not be used for any part of the decontamination cycle including storage of instruments. Why is this? Primarily, to ensure both patient safety and quality assurance of the sterilization process. To ensure that each step of the decontamination process reduces the load of microbes on the instruments, it is crucial that clean and dirty instruments are segregated and are not recontaminated after cleaning or sterilization by direct contact, splashes and aerosols. Moving sterilization out of the treatment area protects the patient from contaminated aerosols generated during cleaning instruments used on previous patients. A steriliser is a pressurised item of equipment and the pressure that builds up behind the door of the steriliser is equivalent to 750 kg or three-quarters of a tonne. If the door fails, which occasionally happens, then the steriliser effectively becomes a bomb, doing considerable damage to anything in its path. If the steriliser is housed on the work surface in a small surgery, the patient or members of the dental team could be seriously hurt.
Figure 7.2 Basic design for a two-room decontamination facility.
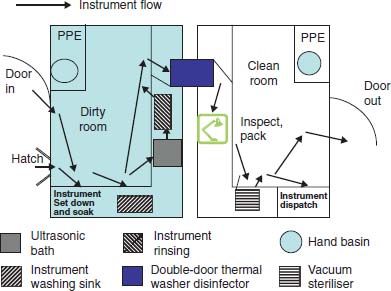
All theses requirements can be met with a dedicated decontamination facility. Segregation between those instruments awaiting decontamination and disinfected and sterile instruments can be achieved in several ways:
- A two-room decontamination facility with one room for dirty instruments and the other room for cleaned instruments for sterilization. A double-door washer disinfector is placed in the barrier between the dirty and clean rooms (see Figure 7.2). The design includes controlled access to the rooms for authorised trained staff with a hand-through hatch (for the delivery of dirty sets of instruments and equipment). This is the preferred option as crosscontamination is very unlikely to occur. The design most closely mimics the tried-and-tested layout used in hospital sterile services departments.
- A single-room decontamination facility (see Figure 7.3) with a one-way flow of instruments and pattern of working from dirty to clean. Thermal washer disinfector is placed in between dirty and clean areas. To prevent aerosol cross-contamination, the room will need to be ventilated by a controlled airflow in the direction opposite to that of the instruments, i.e. from clean to dirty. This is not foolproof as airflows can be disrupted by opening the doors or windows, so this is not the preferred option. Figure 7.3 shows movement patterns of staff within the room to maintain the zoning. This requires a high level of staff discipline and training to achieve this work flow and therefore more prone to human error than a two-room facility.
Figure 7.3 Basic design for a one-room decontamination facility.
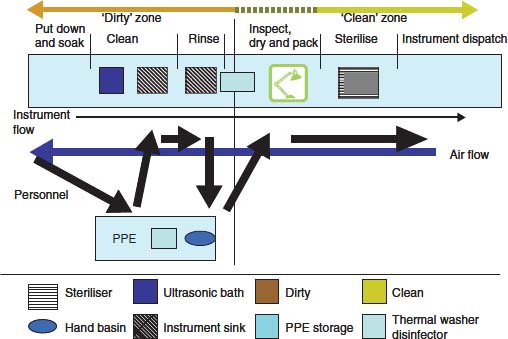
In both options, instruments are transported to and from the treatment room in a clean box with a secure lid. Staff are advised to clean hands and put on personal protective equipment (PPE) on entering the decontamination facility and remove PPE and clean hands on exiting. They are advised to change gloves and aprons when shifting from working in the dirty to the clean zone to prevent cross-contamination of the two processes. Decontamination rooms should contain the following features.
Basic requirements
- Controlled access doors
- Preferably an instrument pass-through hatch
- Uncluttered, single run of sealed, easily cleaned worktop which makes working and surface disinfection easier
- Hand basin and storage for PPE (see Chapter 6)
- Filtered clean air flow to prevent contamination (this includes both microbes and dust particles)
Dirty room/zone (instrument cleaning)
- A setting down area for dirty instruments and precleaning soaking of instruments
- Ultrasonic bath
- A deep instrument-washing sink with detergent dispenser
- A rinsing sink
- Thermal washer disinfector
Clean room/zone (inspection, sterilization and storage)
- A setting down area for machine-washed instruments, with suitable lighting for instrument inspection (e.g. checking for visible contamination and instrument functionality/damage)
- Instrument wrapping (if using a vacuum steriliser)
- A steam steriliser (vacuum and/or non-vacuum)
- An area for setting down (and wrapping if instruments sterilised in nonvacuum steriliser)
- Controlled access to the clean room as repeated door opening hinders environmental control
Sterile instrument storage
Storage of instruments must be away from the patient treatment area and it has been recommended that they should also be stored outside of the decontamination room (Scottish Health Planning Note 13, Part 2, Decontamination Facilities: Local Decontamination Units, NHS National Services Scotland, June 2008; www.hfs.scot.nhs/uk/publications).
- Cleanable shelving or racking (covered) for storing sterilised instruments in procedure-based trays, wrapped cassettes or pouches, with orderly storage to facilitate stock control
- Separate sterile wrapped instruments from instruments that are packaged post-sterilization, which are non-sterile and have a shorter storage life
The actual design will vary with the space available in the dental practice.
Temporal separation
The illustrated designs are for ‘ideal’ decontamination facilities and the interpretation of the Medical Devices Directive is not uniform across the European Union. But the same best practice principles of segregation of dirty and clean procedures apply even when the decontamination is performed in the treatment room, as patient safety is paramount. In the latter situation, a degree of separation can be achieved by introducing some simple measures. Temporal separation (separating activities in time) can be used to conduct different activities in the same space. Patient safety can be enhanced by not decontaminating instrument whilst the patient is in the treatment room. Most surgeries have two sinks: one for the nurse and the other for the dentist, and one of these can be designated as the dedicated sink for instrument cleaning. Recontamination of instruments can be reduced by thorough surface disinfection or worktops between dirty and clean stages. Adequate ventilation and exhaust air extraction with 8–10 air changes per hour in the treatment room will help to reduce air contamination to a low level.
However, such an approach would not be considered acceptable in the UK except as an interim measure before a planned move into a dedicated decontamination facility. The size of the dental practice is not considered as an excuse for omission of any of the fundamental principles of segregation and separation of clean and dirty instruments. Practices which have limited space may consider outsourcing their instrument decontamination to a sterile services provider.
Purchasing of dental equipment
Dental devices marketed in the European Union carry a ‘CE’ mark, which indicates that the equipment is ‘fit for the intended purpose’. Manufacturers of medical/dental devices must by law provide decontamination instructions including methods for cleaning, disinfection, sterilization and state any limit to the number of times an item can be sterilised. Before purchasing a new item, it is prudent to check the manufacturer’s decontamination instructions to ensure that the instruments can be decontaminated with the facilities, that you have, available in the practice. Give preference when choosing instruments to those that can be cleaned by using automated methods, e.g. thermal washer disinfector (see Figure 7.4); alternatively, consider a single-use instrument for difficult-to-clean instruments.
Figure 7.4 Thermal washer disinfector safe symbol on a dental handpiece.
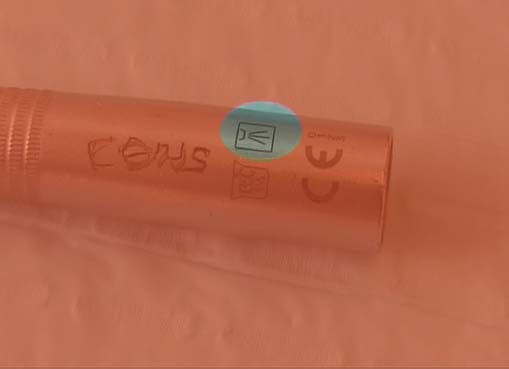
PRESTERILIZATION CLEANING OF DENTAL INSTRUMENTS
The effectiveness of the sterilization process depends upon direct contact between the steam and all surfaces of the load. Residual human tissues, oil or other deposits will prevent contact between the steam and the surfaces of the load. Unremoved contaminants can become fixed to the instruments during sterilization making subsequent removal more difficult. Therefore, all items that you intend to sterilise must be clean and dry before you place them in the steriliser chamber.
Automated versus manual cleaning
Instruments can be cleaned manually or by machine using an ultrasonic bath and/or a thermal washer disinfector. Manual cleaning of instruments should be restricted to those items incompatible with automated washing. Studies have demonstrated that washer disinfectors are more efficient at reducing the bacterial load during pre-sterilization cleaning than either ultrasonic cleaning or manual cleaning of dental instruments. Staff safety is enhanced as instruments do not need to be scrubbed, so there is less exposure to pathogenic microorganisms, sharps injuries or noxious chemicals. Overall productivity is increased in the dental practice as more time becomes available for clinical activities. There are some disadvantages to thermal disinfectors. Manual cleaning is faster than using a thermal washer disinfector. Downtime for instruments cleaned and dried in a thermal washer disinfector and sterilised in a vacuum steriliser is approximately 2 hours, necessitating the purchase of additional instruments if the same number of patients are to be seen.
Preventing corrosion of instruments
Proteins in blood and saliva left on the instrument after treatment are absorbed and then fixed onto the surface of the instrument. Fixation is a chemical process that takes about 40 minutes at room temperature, but is faster at higher temperatures. Eventually, blood and saline will corrode stainless steel instruments, leading to pitting and rusting of the surface. Damaged and pitted instruments are more difficult to clean effectively and corrosion may reduce the life of the instrument. Ideally, instruments should be cleaned as soon as possible after use. If it is not practical to clean the instruments immediately then soak them in a non-ionic or enzymatic detergent solution, or precleaning foam.
Manual cleaning of instruments
Use a non-foaming, neutral (pH 5–9) detergent dispensed in a measured dose in a measured volume of water for manual cleaning or ultrasonic baths. Always take great care with sharp instruments. Before cleaning the instruments the member of staff should put on the following protective clothing:
- Disposable apron/gown
- Face shield or face mask
- Household gloves (surgical gloves should not be worn)
- When moving from working in a designated dirty area to a clean area, change gloves and plastic apron and clean hands
Best practice guide: manual instruments cleaning
- Dedicated deep sinks (separate washing and rinsing sinks) located in a designated dirty zone
- Use a non-foaming detergent (do not use washing-up liquid, which leaves a residue and the surface foam impedes visibility of sharp instruments)
- Use nylon brushes, not green pads or wire brushes. Cleaning equipment (e.g. brushes) should be cleaned and sterilised (or alternatively use disposables) and stored dry between use. Brushes should not be stored in disinfectant solutions
- Use non-shedding and disposable cloths for cleaning and drying equipment
- Fill basin with lukewarm water (water hotter than 35°C will coagulate proteins and inhibit their removal)
- Fully immerse instruments and clean below the waterline to reduce aerosol formation. Avoid scrubbing under running water
- Disassemble multi-part instruments; pay attention to crevices and joints
- Final rinse under hot water to aid instrument drying
- Dry instruments to prevent carryover of contaminated wash water into the steriliser
- Visually inspect instruments for residual debris or blood after cleaning and repeat if necessary
CLEANING OF DENTAL HANDPIECES
Automated dental handpiece cleaning
Dental handpiece can be cleaned manually or preferably by automated cleaning either using a pre-sterilization dental handpiece cleaning machine or using a thermal washer disinfector. Ma/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


