Maintenance of Dental Implants
Jon B. Suzuki, Lynn D. Terracciano-Mortilla, Carl E. Misch
Implant dentistry has evolved into an evidence-based, clinical science with well-documented research to validate previously unsupported clinical practice procedures. Significant efforts that focus on the biology and biomechanics of implant dentistry helped develop and refine clinical techniques based on peer-reviewed findings. The evolution of research and understanding of biological concepts in implant dentistry has caused many areas of conflict and controversy. Innovative theories developed and techniques changed. In addition, sciences have spurred implant dentistry to new pinnacles of success.
The tremendous expansion of knowledge in this field has created new ideas and terminology that is redefined based on new principles.1 In many instances, new research may contradict established paradigms. It may be confusing for clinicians to select correct protocols, procedures, instruments, and techniques. As materials and technique are further investigated, dogma may undergo criticism and controversy. Seasoned clinicians consistently update and modify techniques and instruments to maintain clinical excellence as technology and research advance.
One area of expansion of knowledge and conflict of views relates to the maintenance of dental implants. Early research explored techniques and instruments that were current for the methods and materials of that time. Although many of those implants still exist and are functional in patients, research and advances in technology have given us newer materials and advances in implant design and structure, minimizing previous challenges from a maintenance perspective.
An understanding of the mucoepithelial implant attachment is essential before commencing maintenance procedures. Controversies and parameters for probing and crestal bone loss are important for clinicians to recognize. There are anatomical and histologic differences between the attachment apparatus of teeth compared with implant osseointegration. The bacterial plaque biofilm challenges on these implant–tissue attachments may be significant to clinical success. This chapter explores the success and failure of implants and discusses the biological and engineering parameters that need to be considered when evaluating implants and periimplant tissues.
When the clinician understands the parameters of implants and teeth, specific maintenance plans may be established for the patient. Clinicians should inform patients of expectations and outcomes during treatment and demonstrate oral hygiene options appropriate during each stage. Patients need to recognize the importance of maintenance before the decision is made to commence implant therapy.2 The ability of the patient to understand financial, time, and maintenance obligations is critical and should be made clear to the patient initially and during subsequent appointments. In addition to educating patients, clinicians should assess compliance to home care routines. Patients also should be competent to perform home maintenance procedures.3 As the acceptance and demand for dental implants increase, the need to understand the importance of maintenance as it relates to long-term implant success also increases.3 The role of the dental hygienist in implant maintenance and care is also increasing and becoming more defined3 (Box 34-1).
Implants and associated prostheses are different from natural teeth and may require adjunctive procedures and instruments for professional and patient care.4 Instruments must be effective at removing biofilms and accretions, and procedures performed by patients and clinicians should avoid damage to all components of the implant, abutment, restoration, and tissues.5 Establishment and maintenance of the soft tissue seal around the transmucosal portion of the implant enhances the success of an implant. This barrier is fundamentally a result of appropriate wound healing and connection of epithelial attachments. The maintenance of healthy periimplant tissues may contribute to implant success. In addition, tissues free of inflammation and a biofilm-free implant sulcus will support the patient’s general and oral health.
Plaque Biofilm and Dental Implants
The differences between teeth and implants suggest that dental implants may increase susceptibility to inflammation and bone loss in the presence of bacterial plaque biofilm accumulation.6 Plaque biofilm and inflammation are established etiologic factors of periodontal disease. Sticky masses of bacteria with a polysaccharide matrix colonize on hard and soft surfaces in the oral cavity but can be disrupted and removed with mechanical debridement or chemical inactivation. If undisturbed, plaque biofilm matures. Current chemotherapeutics do not penetrate thick biofilm.
Rough implant surfaces have been reported to retain more biofilm than smooth surfaces,7 yet the rough surfaces result in greater bone–implant contact and are therefore favorable below the bone. Biofilm migrates and transitions from teeth to implants and from implant to implant.8 Similar to teeth, clinical findings of failing implants include inflammation, pockets, and progressive bone loss.9 Similarities of bacteria responsible for periodontitis, periimplant mucositis, and periimplantitis are now recognized.
When evaluating the periimplant microbiota, Lee et al. compared microbial changes between patients with a history of periodontal or periimplant infections and implants that have been in function for a period of time.10 This study reported that patients with a history of periodontitis had a greater impact on enhancing periimplant microbiota than implant loading time. One of the major influences on periimplant microbiota may be the presence of specific microbiota on remaining teeth. Although all implants were successfully osseointegrated, Porphyromonas gingivalis and Tannerella forsythia, red complex periodontal pathogens may colonize contiguous implants. Thus, it is important to emphasize to patients about their responsibility to control plaque biofilm consistently and effectively, especially if the patient has a history of periodontal disease.
Plaque biofilm development and maturation have similarities for natural teeth and dental implants. The gingival sulcus in periodontal health and the permucosal attachment of a successful dental implant are similar.7 In a study of biofilm from 18 edentulous patients with successful dental implants, facultative anaerobic cocci (52.8%) and facultative anaerobic rods (17.4%) were reported.11 However, the pathogens P. gingivalis and spirochetes were absent, and minimal gram-negative rods (7.3%) were present.
Generally, pellicle, a naturally occurring glycoprotein in the saliva, first adheres to intraoral structures, including both teeth and implants. Gram-positive cocci bacteria are the first, “early colonizers” beginning with single cocci and progressing to streptococci forms (Box 34-2). Without adequate oral hygiene measures (i.e., brushing, flossing, and interdental cleaning), additional bacteria colonies, including gram-negative, rod-shaped bacteria, synergistically grow with the established gram-positive bacteria. The gram-negative bacteria are frequently facultative or strict anaerobic bacteria and are considered “late colonizers.” Many, if not the majority, of these gram-negative bacteria are black pigmented and are classified under a number of genera (e.g., Bacteroides, Prevotella, Porphyromonas, Fusobacterium spp.).
Periimplant mucositis is an inflammatory change of the soft tissue surrounding an implant. Periimplant mucositis around an implant is similar to gingivitis around a tooth. There is no loss of attachment for teeth with gingivitis, and there is no loss of bone for implants with periimplant mucositis. The primary etiology is plaque biofilm. Similar to gingivitis, periimplant mucositis is reversible when the plaque biofilm is removed.10 If allowed to progress, periimplantitis may result, which includes loss of bone and loss of osseointegration, similar to loss of attachment and bone with periodontitis.
Periimplantitis exhibits similar microbial flora as adult periodontitis.12 Changes involve both the hard and soft tissues surrounding an implant. The implant may exhibit all the signs of periimplant mucositis as well as exudate, increased pocket depth, and bone loss. If left untreated, significant bone loss, infection, and mobility could result, leading to the failure of an initially integrated implant.
Plaque biofilm reported to be associated with failing dental implants consists largely of gram-negative rods.13 Clinically, failing dental implants are characterized by soft tissue inflammation, increased probing depths, increased mobility, and periimplant radiolucency. Specific pathogens in implant pockets greater than 6 mm include Aggregatibacter (Actinobacillus) actinomycetemcomitans, Prevotella intermedia, and P. gingivalis in more than one third of the sites, as confirmed by DNA analysis.14
More specific studies on plaque biofilm around dental implants suggest similarities between periodontal diseases and failing implants,15 but differences have also been reported.16,17 Spirochetes were not detected in plaque samples from well-maintained and clinically healthy implants.15 Higher proportions of staphylococci (15.1%) were reported than generally found in gingivitis (0.06%) and periodontitis (1.2%) sites.18 This finding suggests that staphylococci may be more significant in developing periimplantitis lesions than previously recognized.
Comparisons of plaque biofilms have been reported in a limited study of Brånemark and ITI (Straumann Institute) implants and are remarkably similar in controlled studies. Ten patients with Brånemark implants and 10 patients with ITI implants were evaluated, and the deepest pockets around the implants were sampled.17,18 After 3 and 6 months, several periodontal pathogens were cultured and isolated, including P. gingivalis, P. intermedia, Fusobacterium nucleatum, and various spirochetes. None of the implants were colonized by A. actinomycetemcomitans. Longer investigations extended microflora reports on dental implants in 19 patients.18 At 3 years, the osseointegrated implants were colonized predominantly by P. gingivalis, P. intermedia, and A. actinomycetemcomitans.
Natural dentitions with dental implants appear to increase the risk for implant infections compared with completely edentulous patients. This suggests that natural teeth may serve as a reservoir for periodontal pathogens, which may promote their colonization and growth to contiguous implants in the same oral cavity.19 Quirynen and Listgarten reported that proportions of coccoid forms (65.8%), motile rods (2.3%), and spirochetes (2.1%) in implant pocket areas were similar to the microorganisms in natural teeth (55.6%, 4.9%, and 3.6%, respectively).20 On the other hand, fully edentulous patients exhibited more coccoid forms (71.3%), fewer motile rods (0.4%), and no spirochetes. They concluded that microflora in partially edentulous implant patients were potentially more pathogenic than in fully edentulous patients. Implant longevity for greater than 3 and 4 years appears to have greater numbers of bacteria colonized than implants in place for 1 or 2 years.21
Probing Depths
Probing depths around teeth are an excellent approach to assess the past and present health of natural teeth. The increasing sulcus depth around natural teeth is related to disease and bone loss.22 However, probing depth indices used to evaluate dental implants are more controversial because relating implant sulcus depth to health is not always directly related.
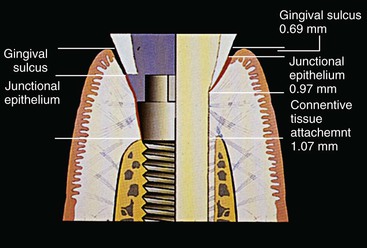
For natural teeth, the surrounding soft tissue has an average biological width of 2.04 mm between the depth of the sulcus and the crest of the alveolar bone.23 It should be noted the biological “width” is actually a height dimension with a greater range in the posterior region compared with the anterior and may be greater than 4 mm in height.24 In teeth, it is composed of a connective tissue (CT) attachment (average, 1.07 mm) above the bone and a junctional epithelial attachment (JEA) (average, 0.97 mm) at the sulcus base, with the most consistent value between individuals being the CT attachment (Figure 34-1).
The sulcular regions around an implant and around a tooth are similar in many respects. The rete peg formation within the attached gingiva and the histologic lining of the gingiva within the sulcus are similar in implants and teeth.25 A free gingival margin forms around a tooth or implant with nonkeratinized sulcular epithelium, and the epithelial cells at its base are also similar in teeth and implants, with junctional epithelial cells for both. However, a fundamental difference characterizes the base of the gingival complex around teeth. A tooth has two primary regions that make up of the biological width compared with one region in an implant (Figure 34-1).
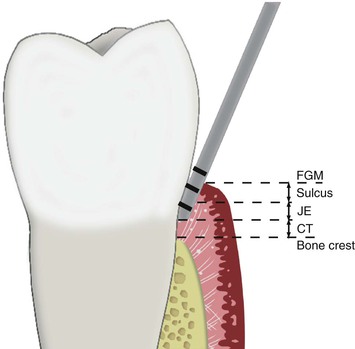
When probing a tooth, the probe measures the sulcus depth, and may penetrate and measure the JEA.26 The junctional epithelial “attachment” of a tooth is not a true attachment. A periodontal probe easily separates the hemidesmosomal close approximation of the epithelial cells. High-volume air from a syringe may dislodge the attachment. Plaque destroys this attachment. The placement of impression string in the sulcus displaces it. The mucopolysaccharide approximation of the hemidesmosome found in the junctional epithelium is not an attachment (Figure 34-2).
The CT attachment zone of the “biological width” around a tooth prevents the probe from penetrating deeper into the sulcus and allows gingival fibers of the CT attachment zone to establish direct connection with the cementum of the natural tooth. This attachment acts as a physical barrier to the bacteria in the sulcus to the underlining periodontal tissues. Eleven different gingival fiber groups comprise the CT attachment zone observed around natural teeth and gingival tissues: dentogingival (coronal, horizontal, and apical), alveologingival, intercapillary, transgingival, circular, semicircular, dentoperiosteal, transseptal, periosteogingival, intercircular, and intergingival.22 At least six of these gingival fiber groups insert into the cementum of the natural tooth: the dentogingival (coronal, horizontal, and apical), dentoperiosteal, transseptal, circular, semicircular, and transgingival fibers. In addition, some crestal fibers from the periodontal fiber bundles insert into the cementum above the alveolar bone. These Sharpey fibers form a true attachment to the tooth, preventing a periodontal probe from invading the periodontal ligament (PDL) space and preventing or delaying the apical movement of plaque biofilm.
A systematic study has investigated the biological seal phenomenon of the soft tissue around dental implants.25 Hemidesmosomes from the JEA region help contribute to a basal lamina–like structure on the implant, which can act as a biological seal.27 However, collagenous components of the linear body cannot physiologically adhere to or become embedded into the implant body.28 The hemidesmosomal seal has a circumferential band of gingival tissue to provide mechanical support against tearing.29 However, the mucopolysaccharide layer is less adherent to an implant surface than natural teeth. The hemidesmosome of natural teeth has a lamina lucida and a lamina densa. The hemidesmosome next to an implant has a lumina lucia, lamina densa, and sublamina lucida (which is less adherent).30
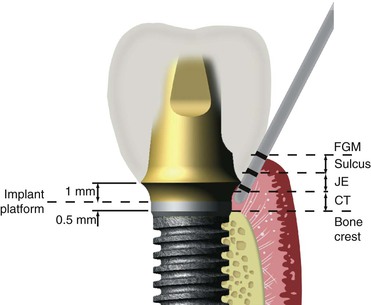
According to Cochran et al., the biological width for implants is 3.3 mm,31 but unlike the biological width dimension for teeth, sulcus depth was included. In an implant gingival region, two of the gingival fiber groups are located around a tooth (circular and periosteogingival fibers), and no periodontal fibers are present.32 These fibers do not insert into the implant body below the abutment margin as they do into the cementum of natural teeth. Instead, the collagen fibers around an implant run parallel to the implant surface, not perpendicular as with natural teeth. Hence, the implant only has a junctional epithelial “attachment” system.
The gingival and periosteal fiber groups are responsible for the CT attachment component of the biological width around teeth. These fiber groups are not present around the transosteal region of an implant. The “biological width” around the abutment–implant connection is not analogous to the CT attachment of teeth. The biological seal around dental implants can prevent or minimize the colonization of bacteria and endotoxins into the underlying bone. However, the seal does not constitute an attachment component of the biological width similar to the one found with natural teeth.
A dental probe introduced into an implant sulcus may proceed past the junctional epithelial close approximation of the tissue, and the probe may proceed to the crestal bone (Figure 34-3). The CT zone for an implant has two fiber groups, and neither inserts into the implant. As a result, with an implant, the probe goes beyond the sulcus, through the JEA, and through the type III collagen CTs and extends closer to bone.26 Because the probe penetrates deeper next to an implant compared with a tooth, dental clinicians have the potential to cross-contaminate the implant sulcus with bacteria from a diseased periodontal site during probing or scaling.
The benefit of probing the implant sulcus has been challenged in the literature because sound scientific criteria for the rationale are lacking. The location of the probe tip subgingivally for a tooth depends on the pressure used, the presence of inflammation, and the angle at which the probe is introduced in the sulcus depth between the junctional epithelium and the root surface. The correct pressure recommended for probing is 20 g, yet conventional probing often exerts a force more than five times this level, and this varies greatly. Pressure-sensitive probes have been made available to address these issues but are rarely used in a clinical practice.33 However, these pressures on probing issues are less important for probing next to an implant because the probe depth is limited by the bone, not a CT attachment.
The potential for damage to the fragile hemidesmosome attachment to the implant or marring of the implant surface exists during probing. In addition, reports in the literature suggest that the reproducibility of attachment level measurements may be questionable and independent from the instrument used to perform the measurements.34,35 Many of these variables are similar for dental implants. In addition, unlike natural teeth, fixed implant prostheses with subgingival margins of crowns often have wide emergence profiles, which make probe positioning difficult around selected implant bodies.
Unlike natural teeth, the implant sulcus depth may be a reflection of the original soft tissue thickness (i.e., biotype) of the area before implant placement. The posterior maxillary tissue can be thicker than 4 mm after tooth extraction and subsequent bone volume loss before implant placement. As a result, the tissue above the bone before implant insertion may be 4 mm thick or more. As a result of greater tissue thickness before surgery and greater probing depth compared with teeth, the probing depth next to a healthy implant may be greater than that of healthy natural teeth.
When the tissues have a thick biotype, gingivoplasty to reduce the flap thickness and pocket depth may be performed at the initial surgery. The advantage of the reduction in tissue thickness at this time is that the tissue heals and matures as the bone–implant interface develops. However, thinning the flap during the initial surgery may cause greater loading of the implant body during healing from an overlying soft tissue–borne temporary prosthesis. After initial bone healing, the stage II uncovery surgery also may correct tissue thickness.
The presence of deep pockets is not always accompanied by accelerated marginal bone loss.36 Stable, rigid, fixed implants were reported with pocket depths ranging from 2 to 6 mm. Healthy, partially edentulous implant patients consistently exhibit greater probing depths around implants than around teeth. An increasing probing depth next to an implant is a more significant sign than a probing depth unrelated to a time interval because it generally reflects bone loss except in cases of gingival hyperplasia or hypertrophy. Probing using fixed reference points on the abutment or crown margin allows evaluation of crestal bone loss versus tissue hypertrophy.
Despite the limitations, charting the attachment level in implant permucosal areas does aid the dentist in monitoring these regions. As the sulcus depth increases, the oxygen tension decreases. The bacteria in an implant sulcus are similar to those of natural teeth.37 A toothbrush and daily hygiene procedures cannot clean a sulcus greater than 2 mm.38 Sulcus depths greater than 5 to 6 mm have a greater incidence of anaerobic bacteria.37,39 As a consequence, this sulcus depth often requires gingivectomy or bone revision surgery. As a general rule, to enable the patient to perform effective daily hygiene, the ideal implant sulcus should be maintained at less than 5 mm.
The monitoring of early crestal bone loss is most important during the first year of stress accommodation of the bone. Minor bone changes are clinically easier to observe with a periodontal probe than with radiographs. Early bone loss may occur on the facial aspect of the implant; radiographs only demonstrate the mesial and distal regions. Changes in crestal bone levels warrant close monitoring and early intervention. Patient education to reduce parafunctional stress on the implant system, the use of parafunctional appliances, and other stress-reducing methods are required when early crestal bone loss beyond the first thread is detected.
Despite the uncertain clinical implications of pocket depth increases, probing is an appropriate method to assess potential deleterious changes in the periimplant environment and should be performed every 3 to 4 months for 1 year after prosthesis delivery. After this time, if crestal bone levels are stable, probing is still relevant. Probing also reveals tissue consistency, bleeding, and exudate. Therefore, probing is important not only to measure increasing sulcus depths but also to allow the dentist to evaluate several periimplant parameters at the same time and at the same sites.
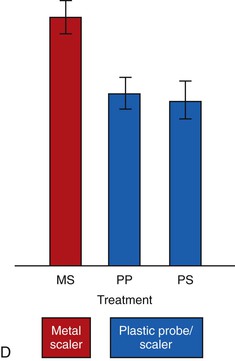
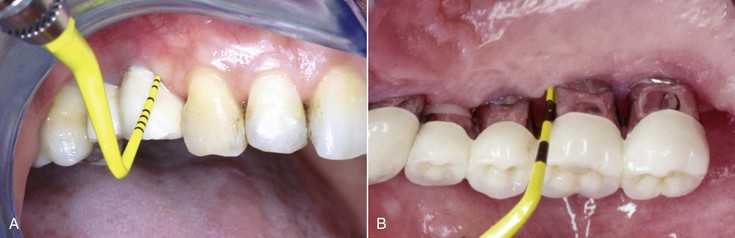
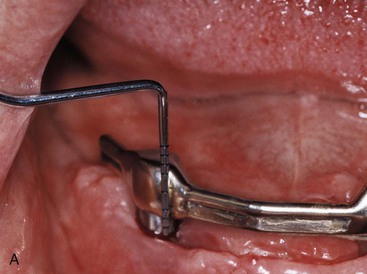
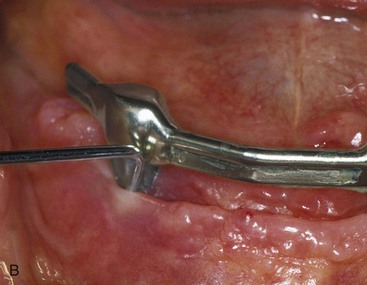
Clinical concern has been raised regarding selection of probes for implant evaluation. It has been argued that different metal types (e.g., stainless steel, titanium) should not come into contact because of a risk of metallic contamination of the two metals and the resulting galvanic corrosion that may develop and cause crestal bone loss. As a result of this concern, the suggestion has been made that only titanium surgical instruments be used to contact the implant and that only titanium or plastic instruments be used to probe or scale the implant (Figure 34-4).
Touching the surface of the abutment subgingivally with a stainless steel instrument is not a clinical concern (Figure 34-5). However, scratching the surface may contribute to plaque biofilm colonization following the direction of the scratch (Figure 34-6). Plaque biofilm follows the direction of scratches on a titanium plate even though right angles and a maze pattern may be scratched onto the surface. Therefore, when probing almost to the bone level around the implant, clinicians should avoid scratching the surface because plaque that forms at the surface may follow the scratch subgingivally to the bone level. This is particularly important during scaling procedures or during the removal of cement below a crown margin. When possible, one should use semicircular strokes, parallel to the sulcus or crown margin, to scale the implant above the bone. If a scratch on the implant body occurs, plaque will not have a direct “highway” below the tissue.
Bleeding Index
Gingival bleeding when probing around teeth correlates with sulcular inflammation and the plaque index. Easily ulcerated sulcular epithelium represents inflammation from plaque biofilm and is the primary cause of bleeding upon probing. A bleeding index is an indicator of sulcus health. Bleeding also can be provoked by undue pressure on the probe.
Controversy surrounds the issue of using bleeding and gingival health as an implant health indicator.27 Unlike a natural tooth, implant success in the first few years is related more often to biomechanical equilibrium than to gingival health. Compared with a natural tooth, soft tissue inflammation from bacteria may be more restricted to supracrestal bone because of the lack of a periodontal membrane or fibrous tissue between the implant and the bone interface. As a result, the bleeding index may not be an important factor when evaluating the early implant quality of health.
The correlation between gingival health and implant success appears in part to be related to the cervical surface condition of the implant. It has been stated with dental implants, there is no evidence that gingivitis is a precursor of progressive bone loss.40 Gingivitis and deep sulcular pockets were not accompanied by accelerated bone loss.36 Both of these reports evaluated a machined-surface titanium screw design (e.g., Nobel Biocare).
In contrast to the previous reports with machined-surface implants, correlations were reported between gingival sulcus depth and implant failure.41 The implant design evaluated in this report had an intramobile element with a larger implant body abutment crevice and a roughened, titanium plasma spray body (IMZ, Germany). A similar correlation between implant failure and gingival health status was observed when a porous titanium alloy microball surface was exposed above the bone (Endopore, Canada).42,43
In addition to the surface condition of the implant, other studies show a correlation to gingival health and implant health.44 One study identified elevated levels of proteolytic enzymes in an implant sulcus with inflammation and bleeding on probing as predictors of implant disease.44 Other studies reported that plaque and gingivitis around implants were correlated.36,45 Another study concluded that the gingival bleeding index is correlated highly with plaque index and the crevicular fluid index.46
The dentist already is encouraged to probe the sulcular region to evaluate crestal bone loss around the implant. Periodontal probing is less demanding and more commonly used than the determination of gingival sulcular fluid volume index. One may observe the bleeding index while probing for sulcus depth and therefore readily record it to evaluate gingival health (Figure 34-7).
Regardless of whether gingival health is relative to implant success, all dentists agree that the ideal soft tissue condition around an implant includes an absence of inflammation. Radiographic bone loss and increased pocket depth have been correlated with sulcular bleeding.46 Therefore, the gingival status around an implant should be recorded and used to monitor the patient’s daily oral hygiene. However, surrounding soft tissues around implants have fewer blood vessels than around teeth. Therefore, inflammation is typically less often observed around implants than around teeth.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses