Chapter 11
Salivary Glands, Salivary Secretion, and Saliva
Arthur R. Hand
Department of Craniofacial Sciences and Cell Biology, School of Dental Medicine, University of Connecticut
Salivary glands are exocrine glands located in and around the oral cavity. There are three major paired glands located extraorally, the parotid, submandibular, and sublingual glands. Additionally, numerous small minor glands are present in the submucosal tissues throughout the oral cavity, except in the gingivae and parts of the hard palate. The salivary glands produce saliva, an aqueous fluid containing electrolytes, other small molecules, proteins, glycoproteins, and mucins. Saliva coats and protects the oral tissues, initiates digestion, and facilitates speaking, chewing, and swallowing. The structure of the salivary glands, the mechanisms underlying saliva secretion, and the composition and functions of saliva will be described in this chapter.
Structure
Histological organization
Salivary glands consist of epithelial cells, collectively termed the glandular parenchyma, that are specialized for the synthesis of various organic molecules, the secretion of these molecules along with water and electrolytes, and their transport to the oral cavity. The gland cells are organized into acini (s. acinus) or secretory endpieces (the terminology used in this textbook) and ducts of different caliber and structure (Fig. 11.1). The secretory endpieces may be spherical or tubular in shape and are the sites of fluid and protein/glycoprotein secretion. The ducts are tubular in shape, and begin as small ducts connected to the endpieces that increase in size as they merge and eventually form a large main duct that empties into the oral cavity. Also present within the gland are connective tissue cells and extracellular matrix, called the interstitial tissue or stroma, that surrounds and supports the parenchyma. Fibroblasts and their products (collagen, elastic and reticular fibers, proteoglycans, and glycoproteins), mast cells and other immune system cells (lymphocytes, macrophages, dendritic cells, plasma cells) are regularly found in the stroma. The connective tissue forms a capsule around the gland, and partitions or septa of connective tissue extend into the gland and organize it into lobes and lobules (Figs. 11.1 and 11.2). Blood vessels and nerves travel in the connective tissue septa and are distributed throughout the stroma, supplying the gland cells with nutrients and oxygen and removing waste products and stimulating saliva secretion, respectively.
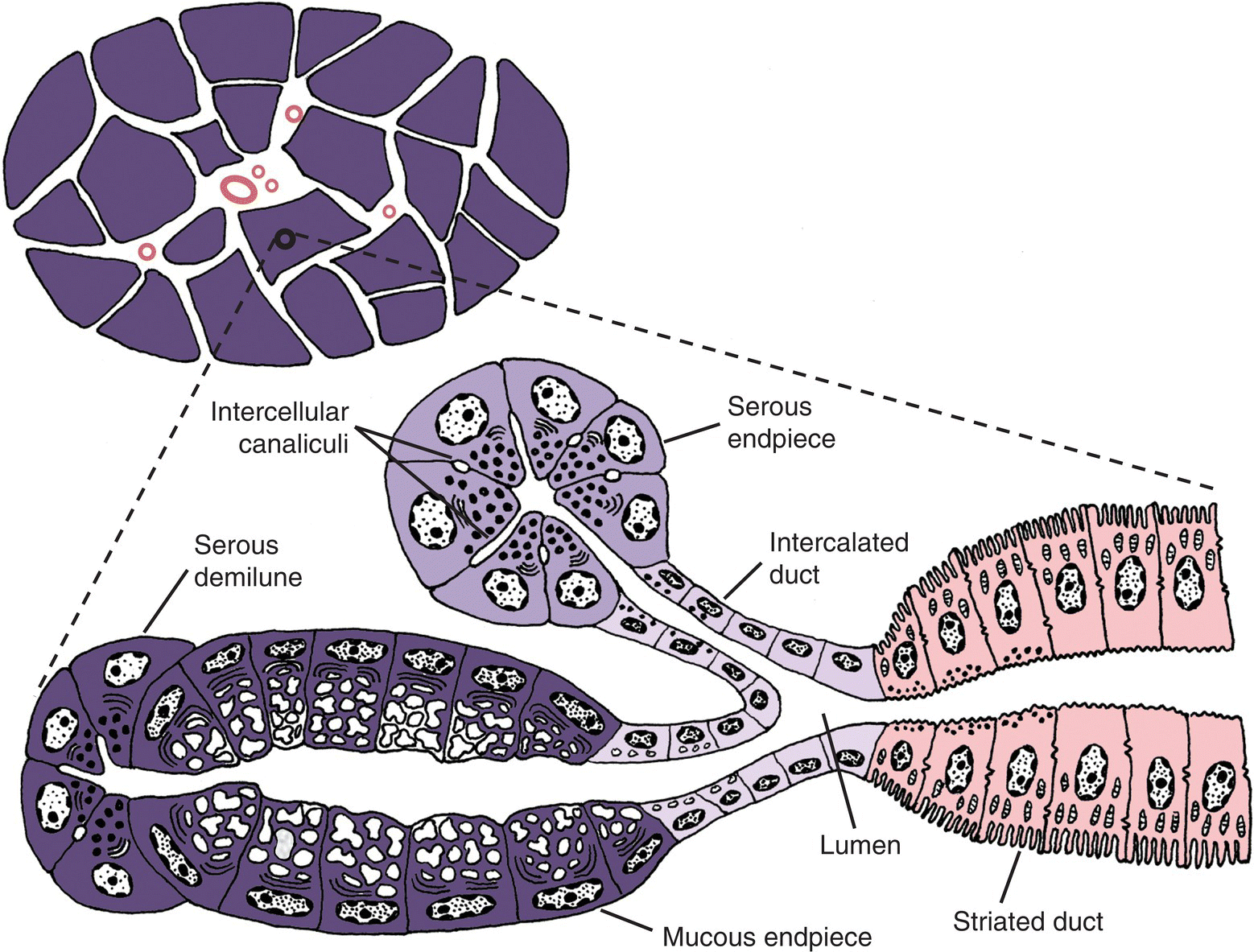
Figure 11.1 Diagram of serous and mucous endpieces, intercalated and striated ducts.
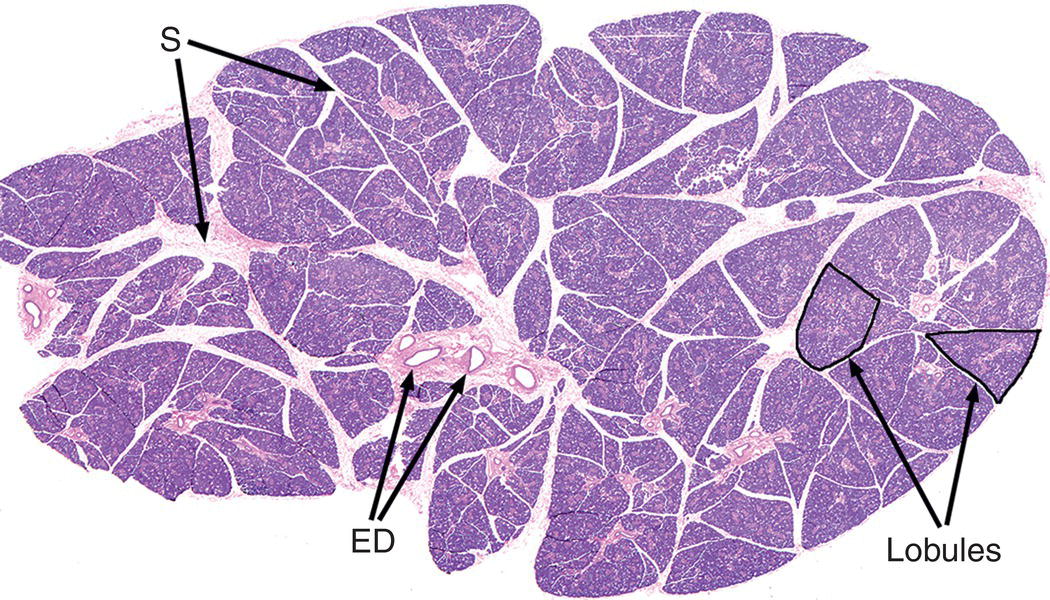
Figure 11.2 Submandibular gland illustrating lobular organization of the gland. Individual lobules (two examples are outlined) are separated by connective tissue septa (S) containing excretory (interlobular) ducts (ED), blood vessels, and nerves.
Two types of secretory cells are found in the endpieces: serous cells and mucous cells. The endpieces may be composed of all serous cells, all mucous cells, or may be mixed, with both serous and mucous components.
Serous cells
In a serous endpiece, the cells are roughly pyramidal in shape, with a broad basal surface adjacent to the basal lamina and connective tissue stroma, and a narrow apex that, along with the apices of adjacent serous cells, forms a lumen in the center of a spherical endpiece (Fig. 11.3). The cells are joined by junctional complexes that include tight and adhering junctions and desmosomes. The luminal surface area is increased by extensions of finger-like intercellular canaliculi down the lateral sides of the cells. Serous cells have a spherical, basally located nucleus, abundant basal rough endoplasmic reticulum, a prominent Golgi complex, and numerous secretory granules, about 1 μm in diameter, that are stored in the apical cytoplasm (Figs. 11.4 and 11.5). Serous cells synthesize proteins and glycoproteins, which are transported from the rough endoplasmic reticulum to the Golgi complex, where they are modified, condensed, and packaged into the secretory granules.
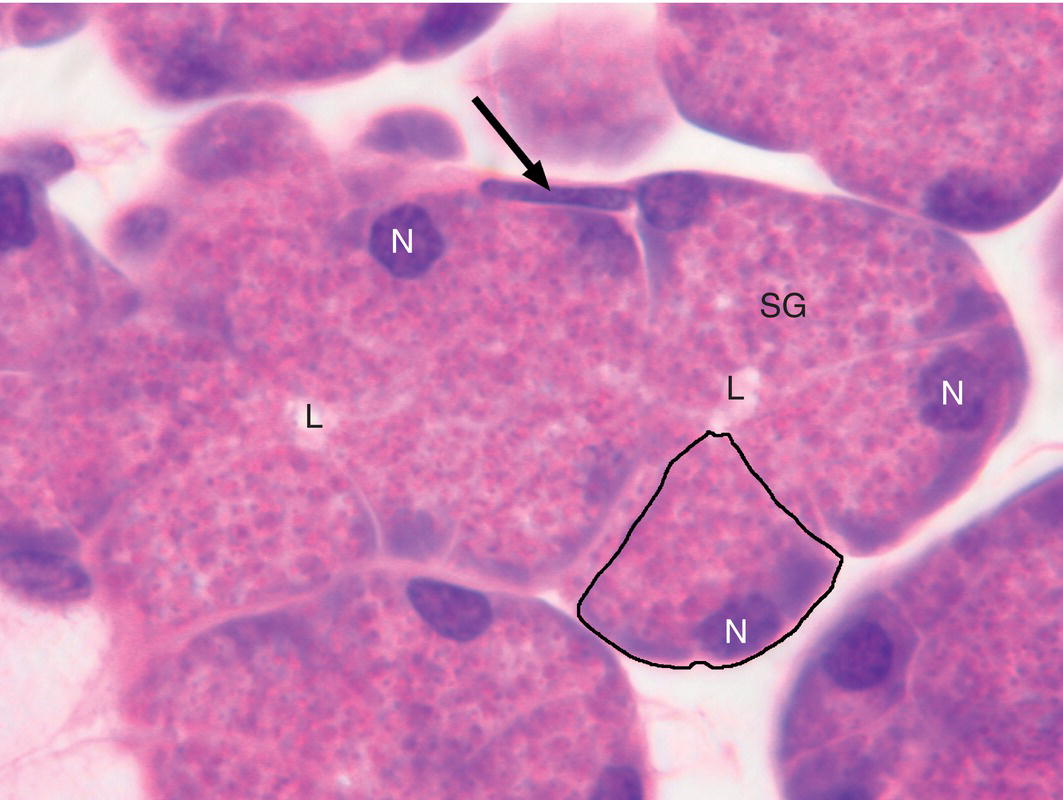
Figure 11.3 Serous endpiece. Serous cells are pyramidal in shape (example outlined), with a broad base adjacent to the connective tissue stroma and a narrow apex abutting a central lumen (L). The nuclei (N) are located in the basal cytoplasm, and the supranuclear cytoplasm is filled with secretory granules (SG). The elongated nucleus of a myoepithelial cell (arrow) is located along the basal surface of the endpiece.
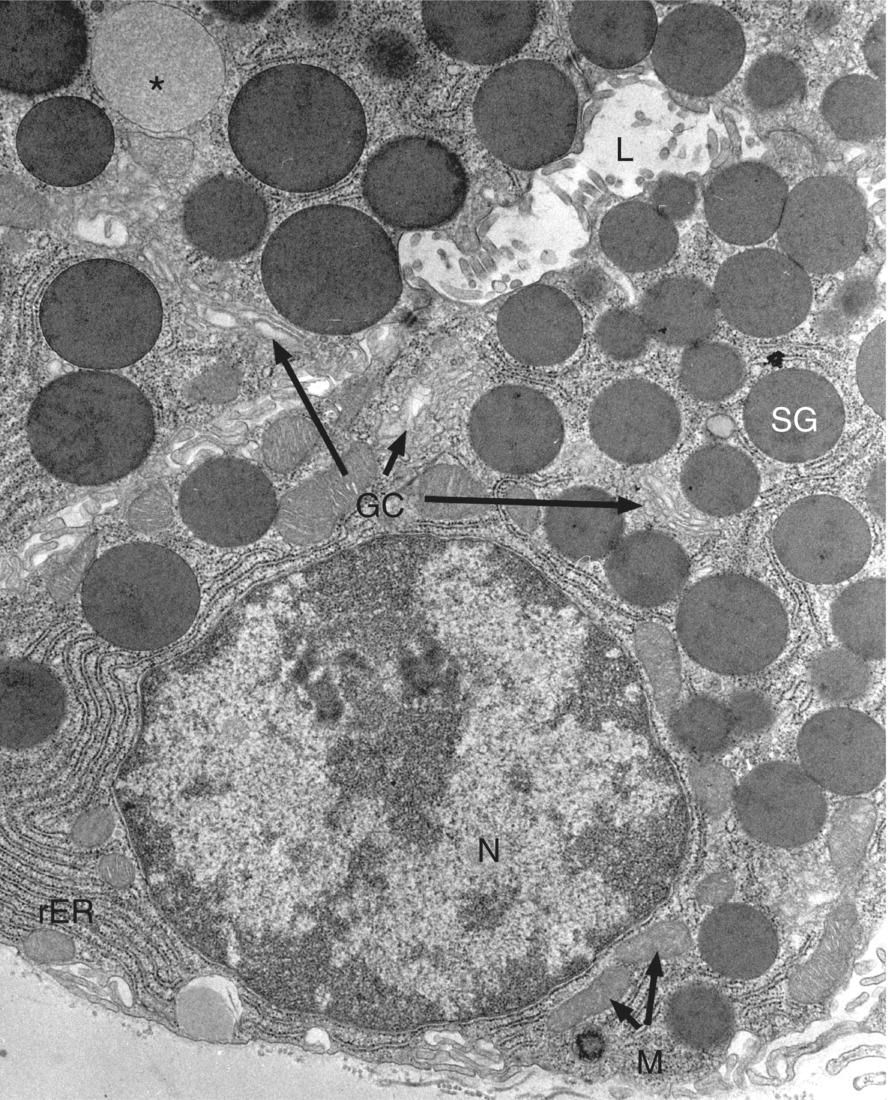
Figure 11.4 Serous cell. Electron micrograph showing portions of three serous cells. A spherical nucleus (N), cisternae of rough endoplasmic reticulum (rER), and several mitochondria (M) are located in the basal cytoplasm. The Golgi complex (GC) is located near the nucleus, and an immature (forming) secretory granule (*) is seen at the upper left. Dense secretory granules (SG) fill the apical cytoplasm adjacent to the lumen (L).
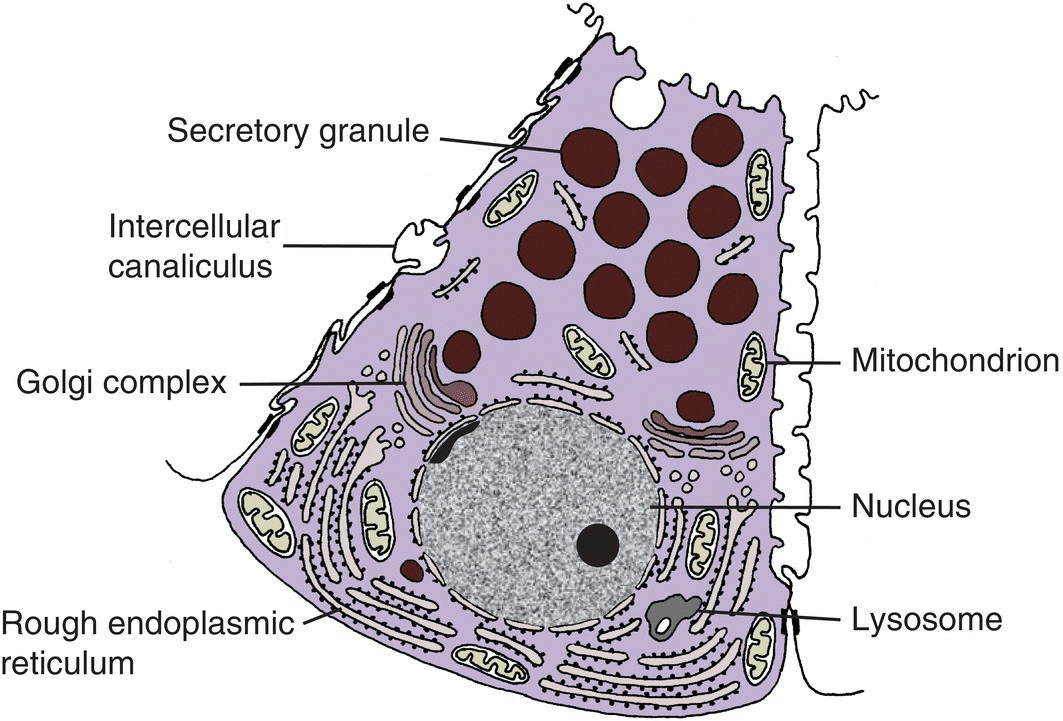
Figure 11.5 Diagram of a serous cell.
Mucous cells
In a mucous endpiece, the cells may have a pyramidal or cuboidal shape, and the endpieces are typically tubular in shape with a relatively wide lumen (Fig. 11.6). The apical ends of adjacent mucous cells are joined by junctional complexes to form a lumen, but mucous endpieces lack intercellular canaliculi. The nuclei of mucous cells usually are somewhat flattened and compressed against the base of the cell, and have denser chromatin than the nuclei of serous cells. The mucous cells have basally located rough endoplasmic reticulum, an extensive Golgi complex, and large, pale, poorly staining secretory granules that fill the apical cytoplasm (Figs. 11.7 and 11.8). The main products of mucous cells are mucins, which have a polypeptide core that is highly glycosylated during transit through the Golgi complex.
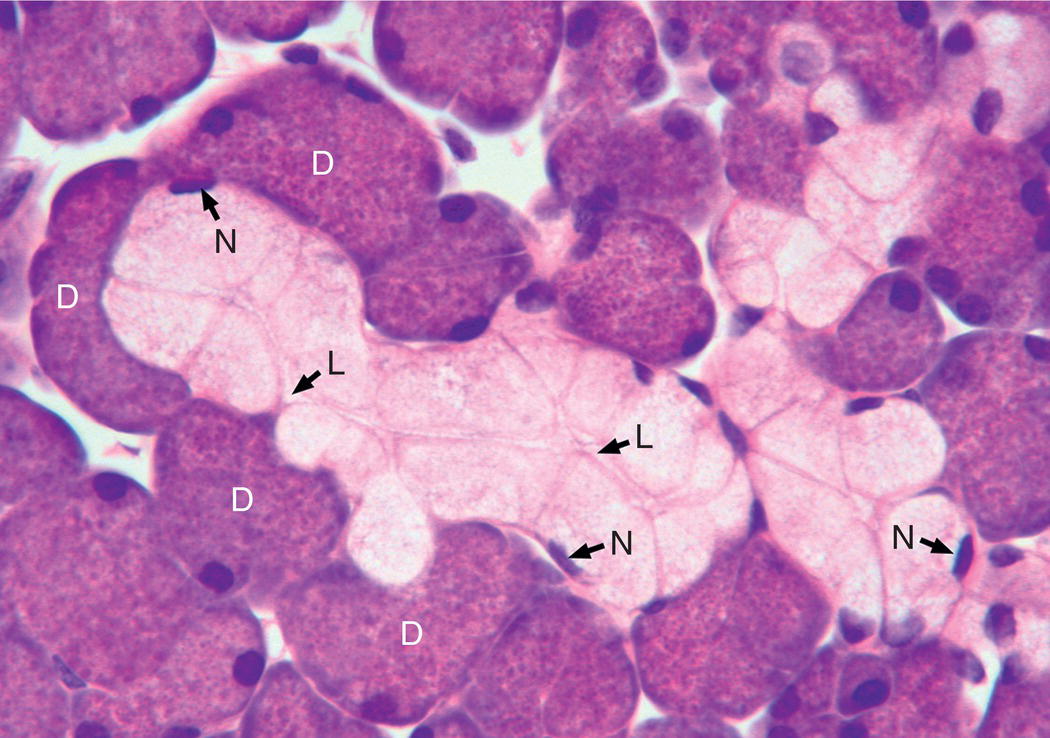
Figure 11.6 Mucous endpiece. Several mucous cells filled with pale-staining mucous granules form a tubular endpiece. Only a few flattened mucous cell nuclei (N) are visible in this section. The lumen (L) extends the length of the tubule. Several serous demilunes (D) surround the mucous tubule.
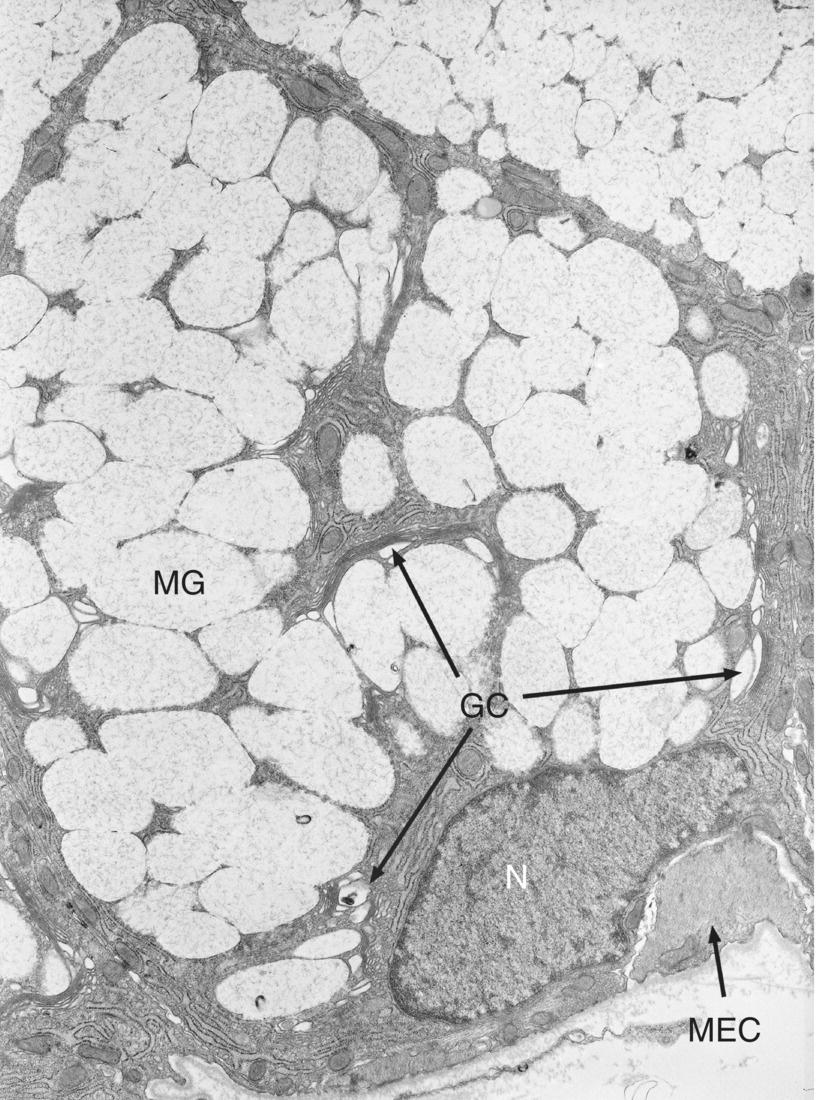
Figure 11.7 Mucous cell. Electron micrograph of a mucous cell. The flattened nucleus (N) is located basally, and rough endoplasmic reticulum is present mainly in the basal cytoplasm. The Golgi complex (GC) is extensive, and the supranuclear cytoplasm is packed with pale mucous secretory granules (MG), many of which are fused with adjacent granules. A myoepithelial cell process (MEC) is present at the base of the mucous cell.
(From: Hand, A.R. (1986) In Oral histology: inheritance and development, 2nd edn. (eds. D.V. Provenza & W. Seibel). Lea and Febiger, Philadelphia. Reprinted by permission of Lippincott Williams & Wilkins.)
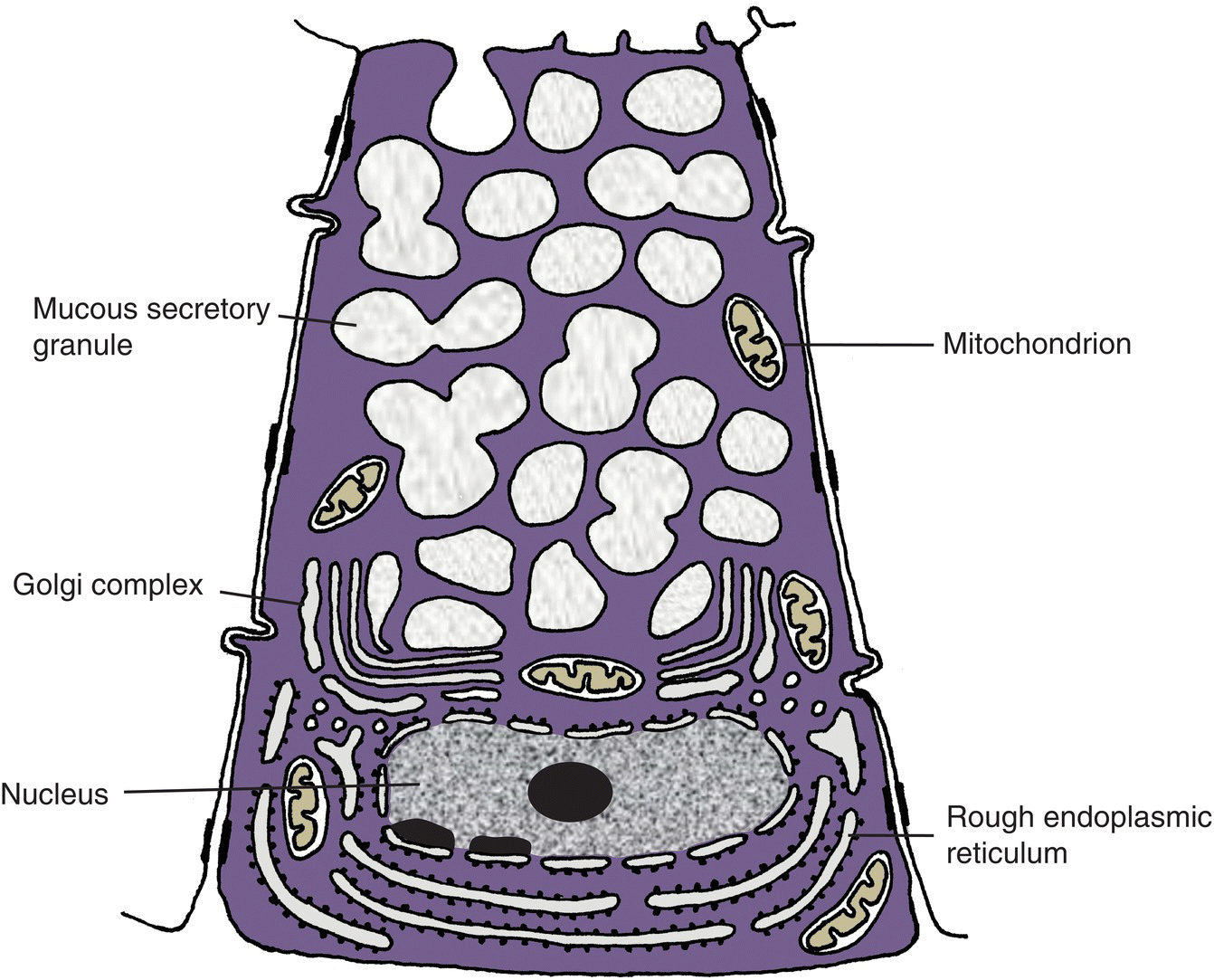
Figure 11.8 Diagram of a mucous cell.
In glands containing mucous endpieces, it is common to find a few serous cells in a characteristic relationship to the mucous cells. In these mixed endpieces, mucous cells are arranged in a typical tubular endpiece structure, and serous cells form a crescent at the blind end of the tubule, called a serous demilune (Fig. 11.6). The serous cells have a narrow apical extension and intercellular canaliculi that reach the lumen of the mucous tubule.
The above description of serous and mucous cells implies a clear distinction between these two cell types. In reality the differences are less distinct. The oligosaccharide component of serous glycoproteins can range from neutral to acidic in nature, with histochemical characteristics similar to those of mucous glycoproteins; hence some authors use the term seromucous to describe these cells. Moreover, some serous cells are known to produce some of the same mucins that mucous cells make, and some mucous cells produce non-glycosylated proteins. The histological appearance of fixed mucous cells described above, common to mucous cells throughout the body, is believed to be caused by swelling of the mucous granules during the preservation of the cells by chemical fixatives. When mucous cells are preserved by rapid freezing, which is believed to more closely reflect the native state, their structure is remarkably similar to that of serous cells.
Myoepithelial cells
A third cell type found in secretory endpieces is the myoepithelial cell. Although epithelial in origin, myoepithelial cells are similar in structure and function to smooth muscle cells. They are located between the basal membrane of the secretory cells and the basal lamina and have a stellate shape with numerous branching processes that are filled with contractile filaments (actin and myosin) and wrap around the endpieces (Figs. 11.9, 11.10, and 11.11). Contraction of myoepithelial cells squeezes the endpiece and forces the primary saliva in the lumen into and along the duct system. Myoepithelial cells also have an important role in maintaining the integrity of the secretory endpieces. They help the secretory cells maintain their polarity, and they secrete protease inhibitors and antiangiogenesis factors. Myoepithelial cells also are found in some parts of the duct system, where they tend to have a fusiform shape with fewer processes.
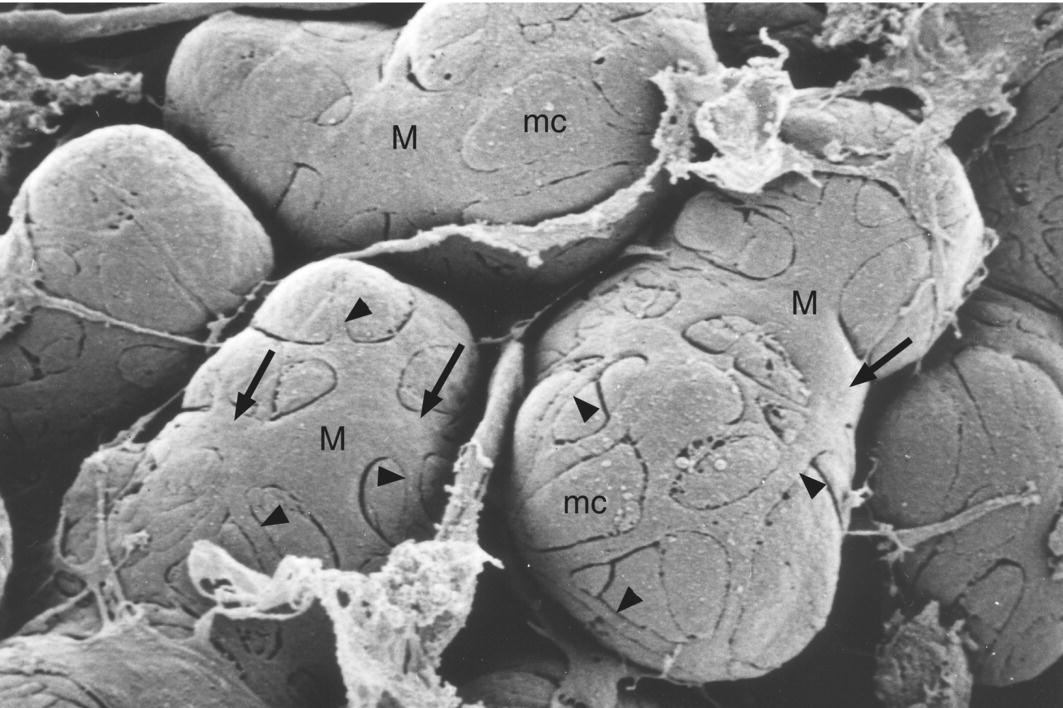
Figure 11.9 Myoepithelial cells. Scanning electron micrograph showing myoepithelial cells (M) covering the basal surface of mucous endpiece cells (mc) in the rat sublingual gland. Broad processes (arrows) extend from the cell bodies, and give rise to smaller processes (arrowheads).
(From: Nagato, T. et al. (1980) A scanning electron microscope study of myoepithelial cells in exocrine glands. Cell and Tissue Research, 209(1), 1–10. Reprinted by permission of Springer Science + Business Media.)
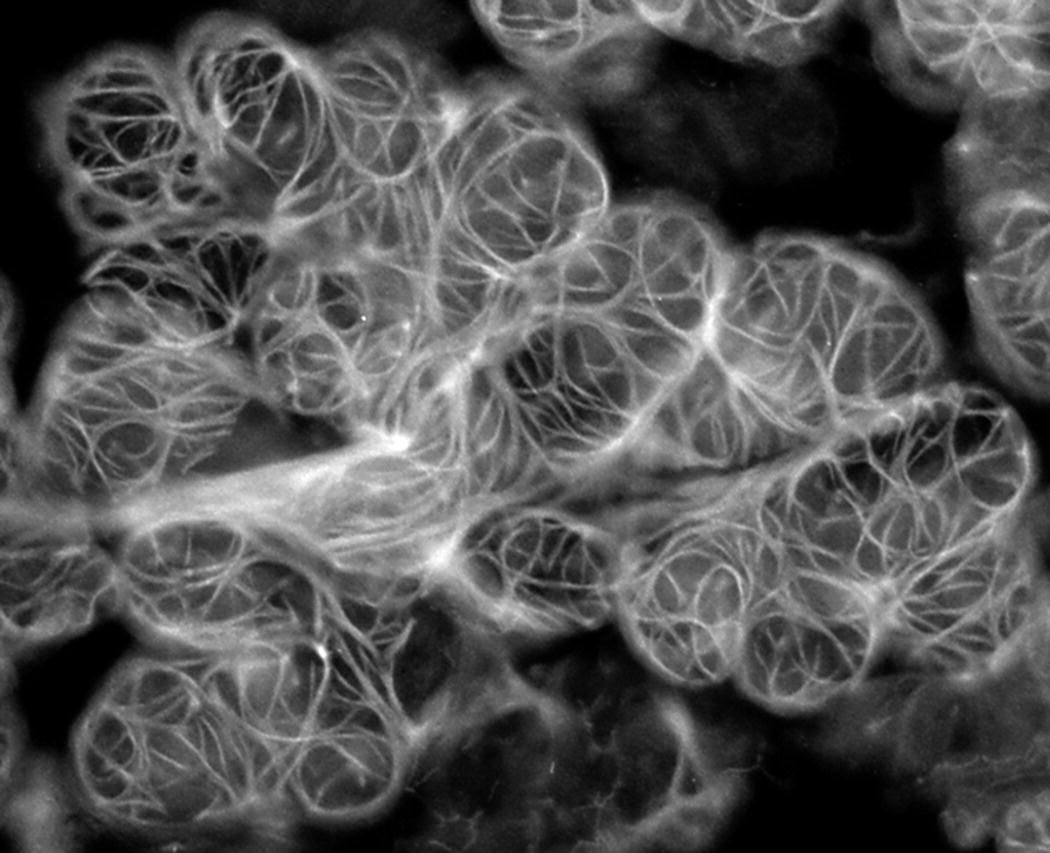
Figure 11.10 Myoepithelial cells. Fluorescence micrograph of myoepithelial cells surrounding the secretory endpieces of the rat sublingual gland. Actin filaments in the processes are labeled with fluorescent phallacidin, an actin-binding compound from the mushroom genus Amanita.
(From: Murakami, M. et al. (1991) Effect of parasympathectomy on the histochemical maturation of myoepithelial cells of the rat sublingual gland. Archives of Oral Biology, 36(7), 511–517. Reprinted by permission of Elsevier.)
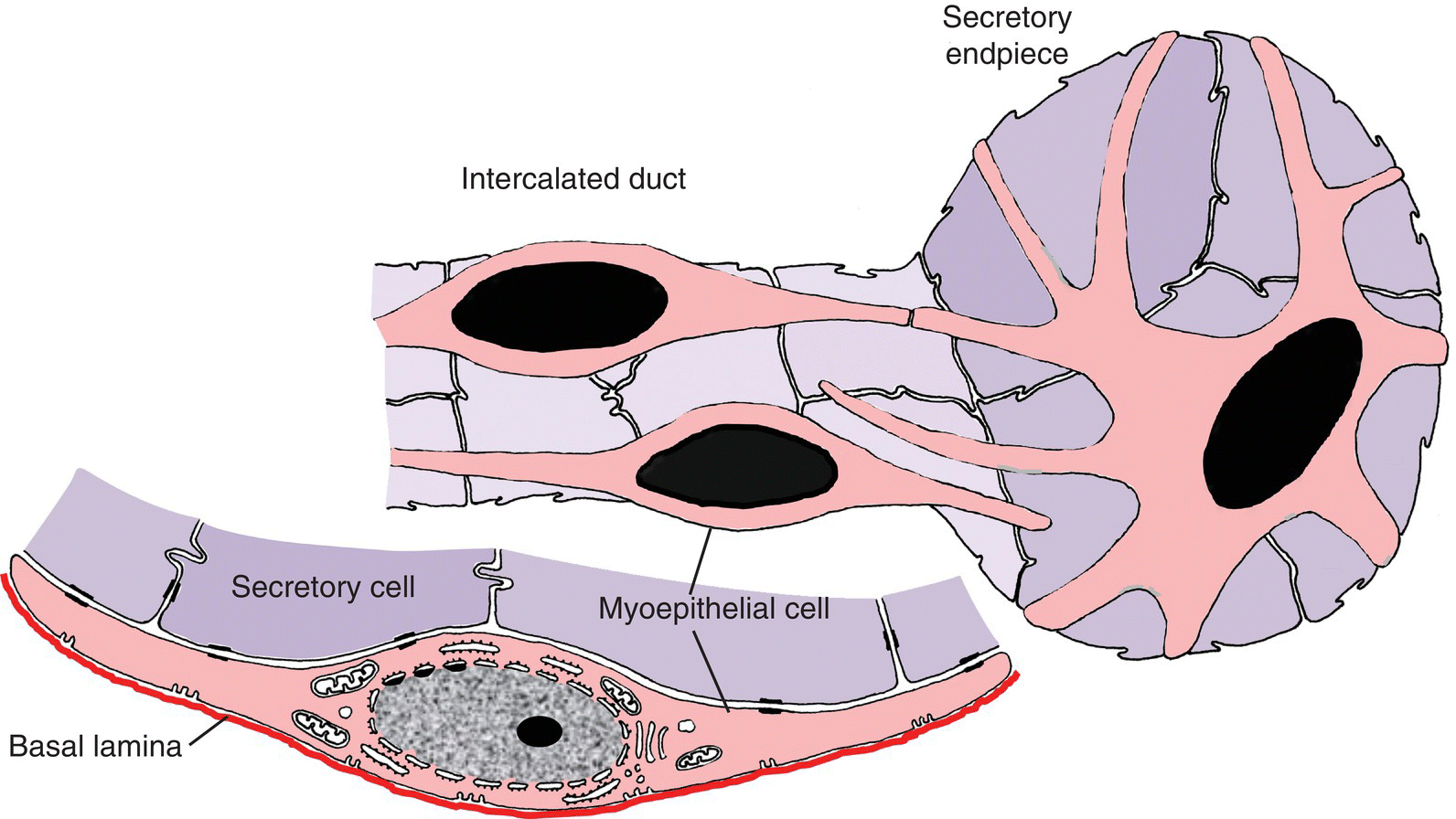
Figure 11.11 Diagram of myoepithelial cells in section and surface views.
Ducts
The duct system of salivary glands consists of three distinct structural components: intercalated ducts, the first duct leading from the secretory endpiece; striated ducts, the main intralobular component of the duct system; and excretory ducts, located in the interlobular connective tissue.
Intercalated ducts
Intercalated ducts have a small diameter, less than that of the endpieces, and are composed of cuboidal-shaped cells with a relatively undifferentiated appearance (Figs. 11.12 and 11.13). The first cells of the duct, adjacent to the endpiece, may have a few apical secretory granules that contain small amounts of some secretory proteins or mucins. These small ducts merge with the ducts leading from adjacent endpieces to form a larger intercalated duct, and these may again merge prior to emptying into a striated duct. As noted above, fusiform-shaped myoepithelial cells are present in intercalated ducts, oriented longitudinally with respect to the duct.
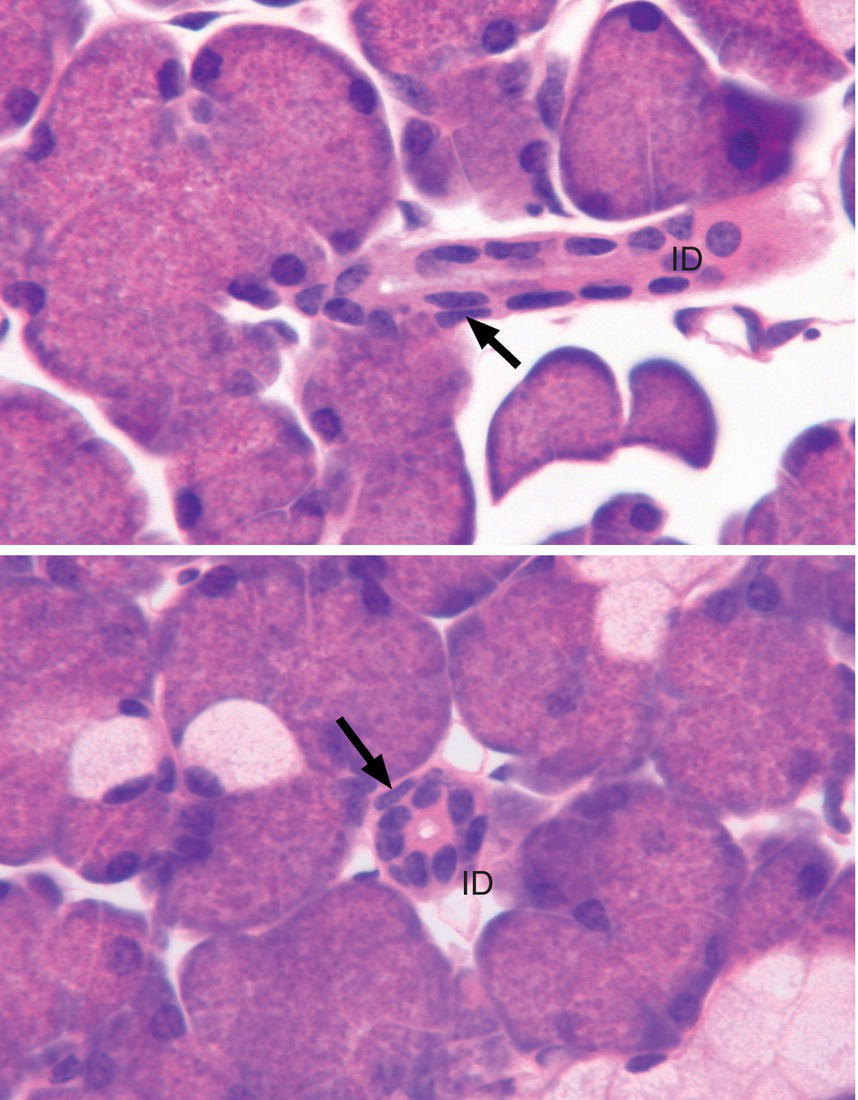
Figure 11.12 Intercalated ducts. Intercalated ducts (ID) are shown in longitudinal (upper panel) and cross section (lower panel). The diameter of the intercalated duct is considerably smaller than that of the endpiece. The duct cells are low cuboidal to cuboidal in shape; nuclei of myoepithelial cells (arrows) are present along the basal surface of the ducts.
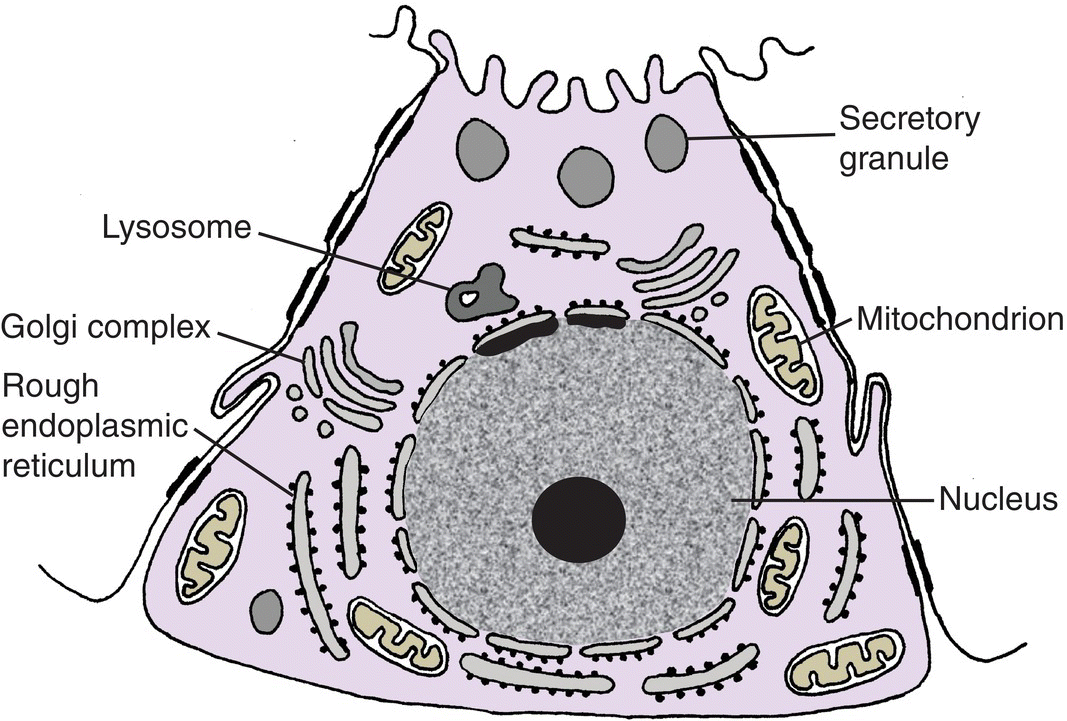
Figure 11.13 Diagram of an intercalated duct cell.
Striated ducts
Striated ducts are present in abundance in the parotid and submandibular glands, less frequently in the sublingual gland, and rarely in minor salivary glands. They are not found in the pancreas or other exocrine glands, thus they serve to distinguish the major salivary glands histologically from other glands. These ducts have a simple columnar epithelium, a diameter greater than that of the endpieces, and larger lumina than those of the endpieces and intercalated ducts (Fig. 11.14). The nuclei are spherical to oval in shape and located more or less centrally in the columnar duct cells. The cytoplasm contains abundant mitochondria, a few cisternae and tubules of rough and smooth endoplasmic reticulum, lysosomes, and peroxisomes. Some cells have small secretory granules and/or endocytic vesicles in the apical cytoplasm. The basal cell membranes as well as the lateral membranes are highly infolded and interdigitate with similar membrane folds of adjacent cells (Figs. 11.15 and 11.16). The cytoplasm in between these membranous partitions is filled with elongated mitochondria. In the light microscope, the infolded membranes and mitochondria create an impression of lines or “striations” extending from the base of the cell toward the nucleus, thus giving these ducts their name. Numerous capillaries and small venules are present in the connective tissue around the striated ducts. Within the lobule, these ducts merge into larger striated ducts, which may have occasional small basal cells that do not extend to the ductal lumen.
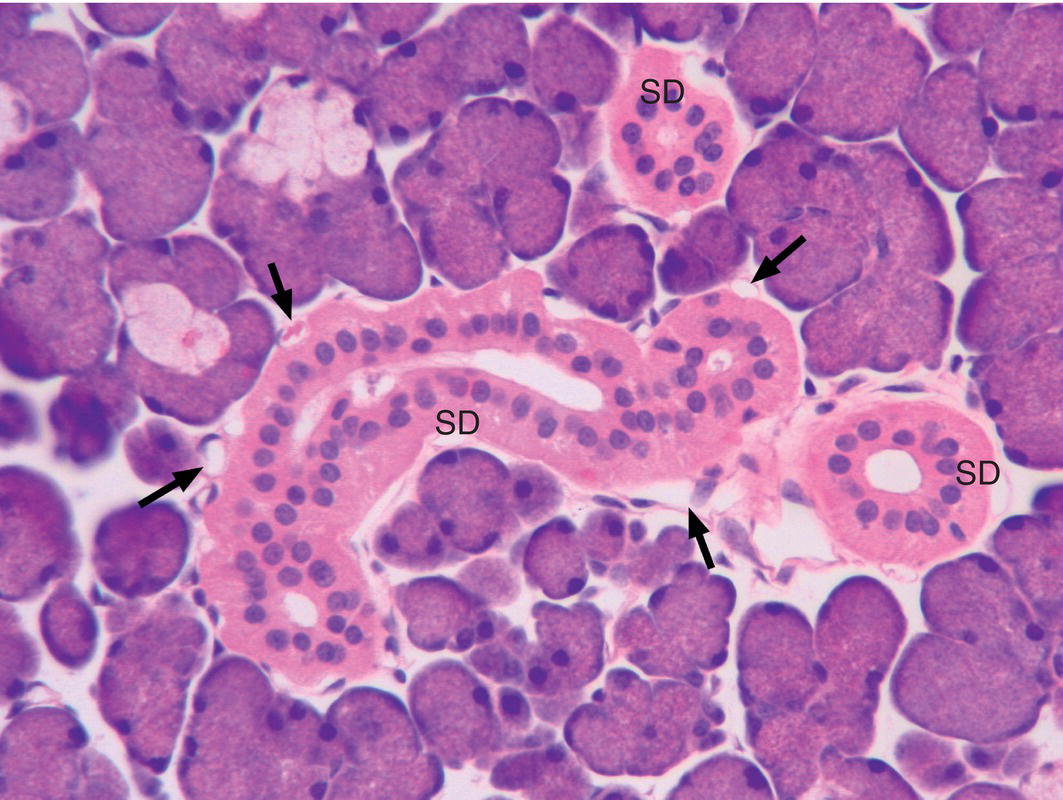
Figure 11.14 Striated ducts. Cross and longitudinal sections of striated ducts (SD). The ducts are lined by a simple columnar epithelium, and their diameter is usually greater than that of the endpieces. Numerous small blood vessels (arrows) are present adjacent to the striated ducts.
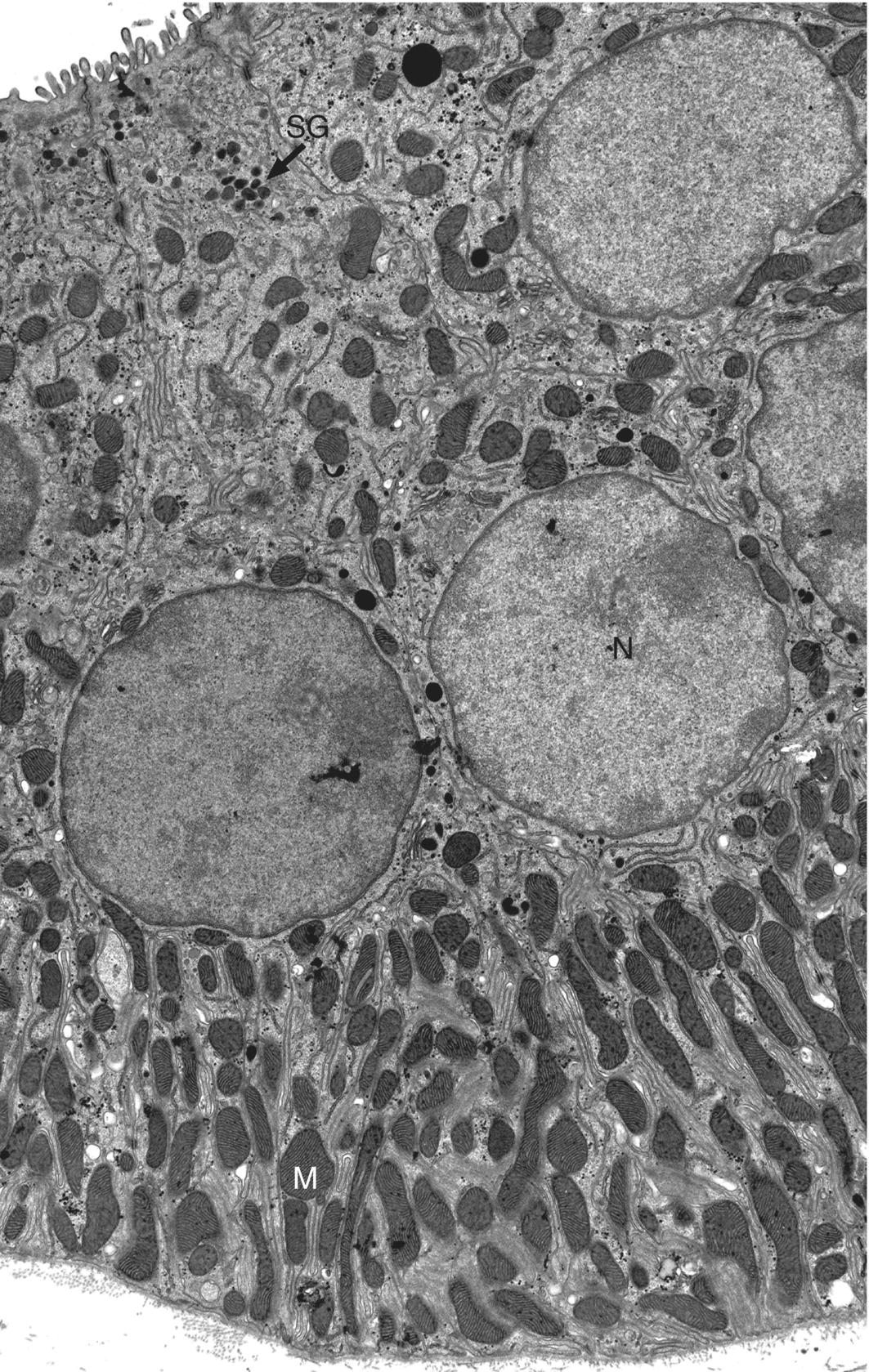
Figure 11.15 Striated duct. Electron micrograph showing portions of several striated duct cells. The basal and lateral cell membranes are highly infolded and interdigitate with similar infolded membranes from adjacent cells. The cells contain numerous mitochondria (M), especially in the cytoplasmic partitions between the infolded basal membranes. Small secretory granules (SG) are present in the apical cytoplasm.
(This micrograph was originally published in the Journal of Biological Chemistry. Evans, R.L. et al. (2000) Severe impairment of salivation in Na+/K+/2Cl− cotransporter (NKCC1)-deficient mice. Journal of Biological Chemistry, 275(35), 26720–26726. © The American Society for Biochemistry and Molecular Biology.)
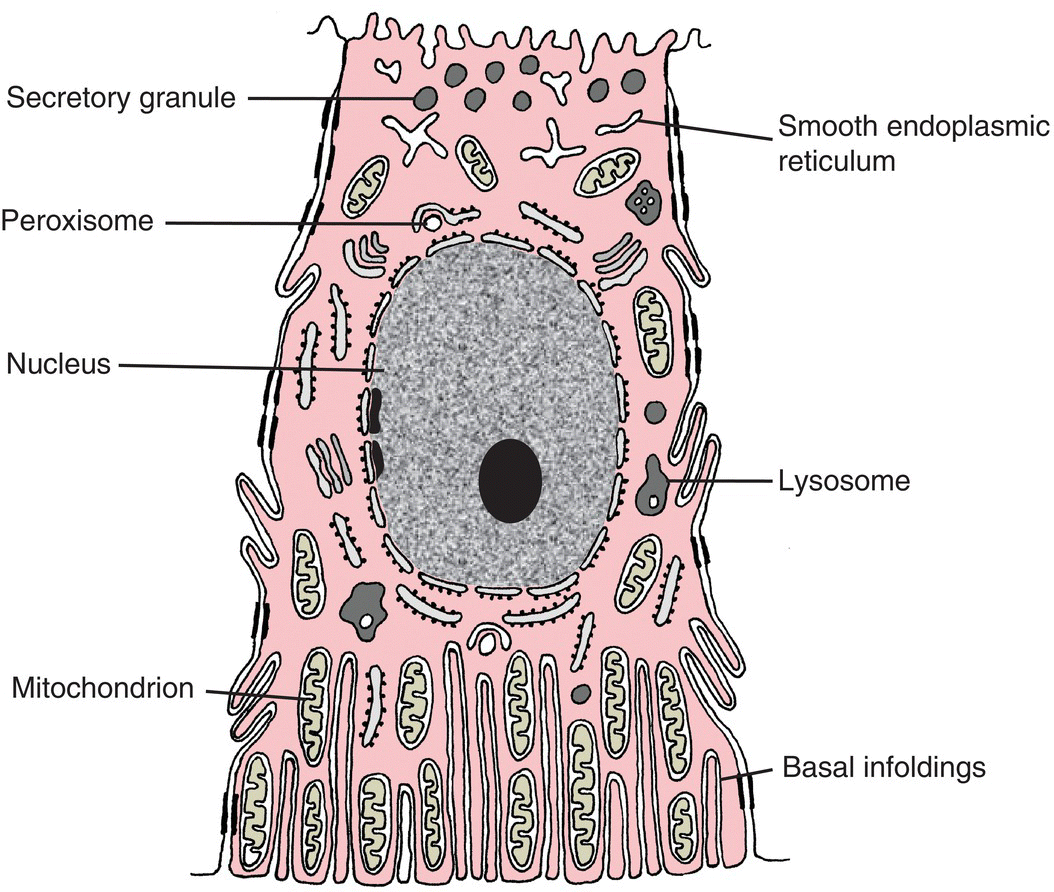
Figure 11.16 Diagram of a striated duct cell.
Excretory ducts
As the large striated ducts leave the lobule and enter the connective tissue septa, they become excretory (interlobular) ducts. Excretory ducts are larger in diameter and have wider lumina than striated ducts, and are surrounded by relatively dense connective tissue. The epithelium of the ducts is pseudostratified, with columnar cells that extend from the basal lamina to the lumen and basal cells that contact the basal lamina but do not reach the lumen (Fig. 11.17). The columnar cells are somewhat similar in appearance to the striated duct cells, but have fewer basolateral membrane infoldings and fewer mitochondria. The excretory ducts continue to merge, increasing in size until a single main duct emerges from the gland. Occasional goblet cells are present in the larger ducts, and the epithelium may change to stratified columnar or even stratified squamous near the oral opening.
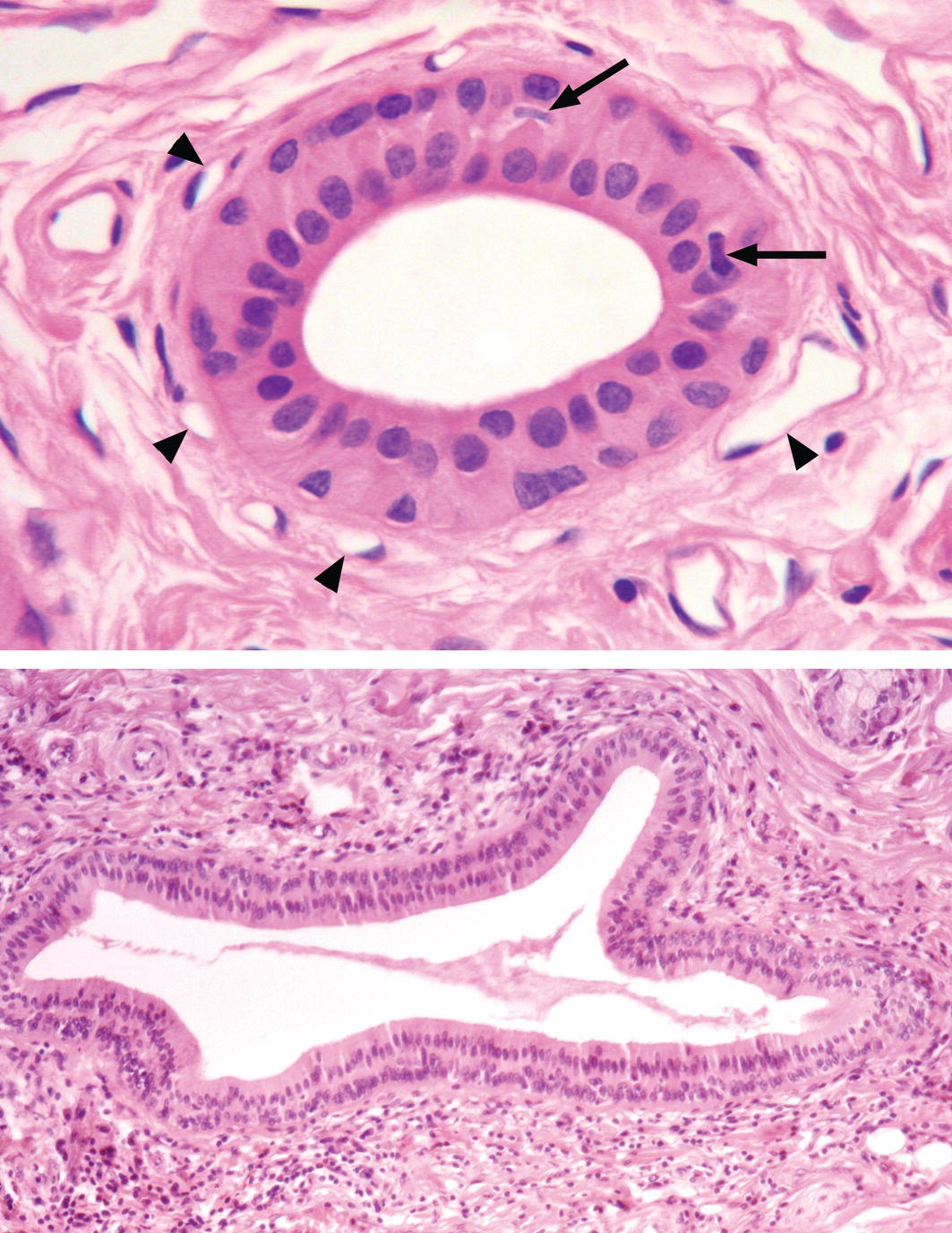
Figure 11.17 Excretory duct. Excretory ducts are located in the interlobular connective tissue septa. Smaller excretory ducts (upper panel) are lined by a pseudostratified epithelium and have a larger lumen than the intralobular striated ducts. Numerous small blood vessels (arrowheads) are located adjacent to the duct. Nuclei of two dendritic cells are indicated by the arrows. Larger excretory ducts may have a stratified cuboidal or stratified columnar epithelium (lower panel) or a stratified squamous epithelium close to the oral opening.
Other cell types occasionally are found in the ducts. Myoepithelial cells may be present in the striated and excretory ducts, and brush (tuft) cells, presumed chemosensory cells with numerous long microvilli and apical microvesicles, are found in excretory ducts. Migratory cells such as lymphocytes and macrophages may be present in the epithelium throughout the duct system, and dendritic cells (antigen-presenting cells) also are commonly found within the ductal epithelium (Fig. 11.17). Finally, stem cells capable of proliferation and differentiation into other gland cell types are thought to reside in the intercalated and excretory ducts.
Development
Development of the different salivary glands is initiated at different times during embryonic life. The parotid gland begins its development at four to six weeks, the submandibular gland at six weeks, and the sublingual and minor glands at eight to twelve weeks. The glands begin their development as a proliferation of epithelial cells at specific sites in the primitive oral mucosa. Continued proliferation and growth of the epithelial cells into the underlying mesenchyme results in a solid cord of cells that begins to undergo repeated dichotomous branching, eventually producing a “bush-” or “tree-like” structure (Fig. 11.18). The proliferation and branching is regulated by epithelial-mesenchymal interactions, and occurs by the developmental process of branching morphogenesis that also forms the pancreas, lungs, kidneys, and other organs. A variety of growth factors and their receptors and specific transcription factors are necessary for proper development of the glands. As development proceeds, lumina are formed in the ducts and then in the terminal buds, and the epithelial cells begin the process of cytodifferentiation. The inner layer of cells differentiate into secretory cells, whereas cells of the outer layer become myoepithelial cells. The secretory cells initiate secretory protein synthesis, secretory granules accumulate in the cytoplasm, autonomic innervation is established, and with the development of functional neurotransmitter receptors the cells attain the ability to secrete saliva. Maturation of the secretory cells and ducts occurs during the last two months of gestation, and the glands continue to increase in size postnatally.
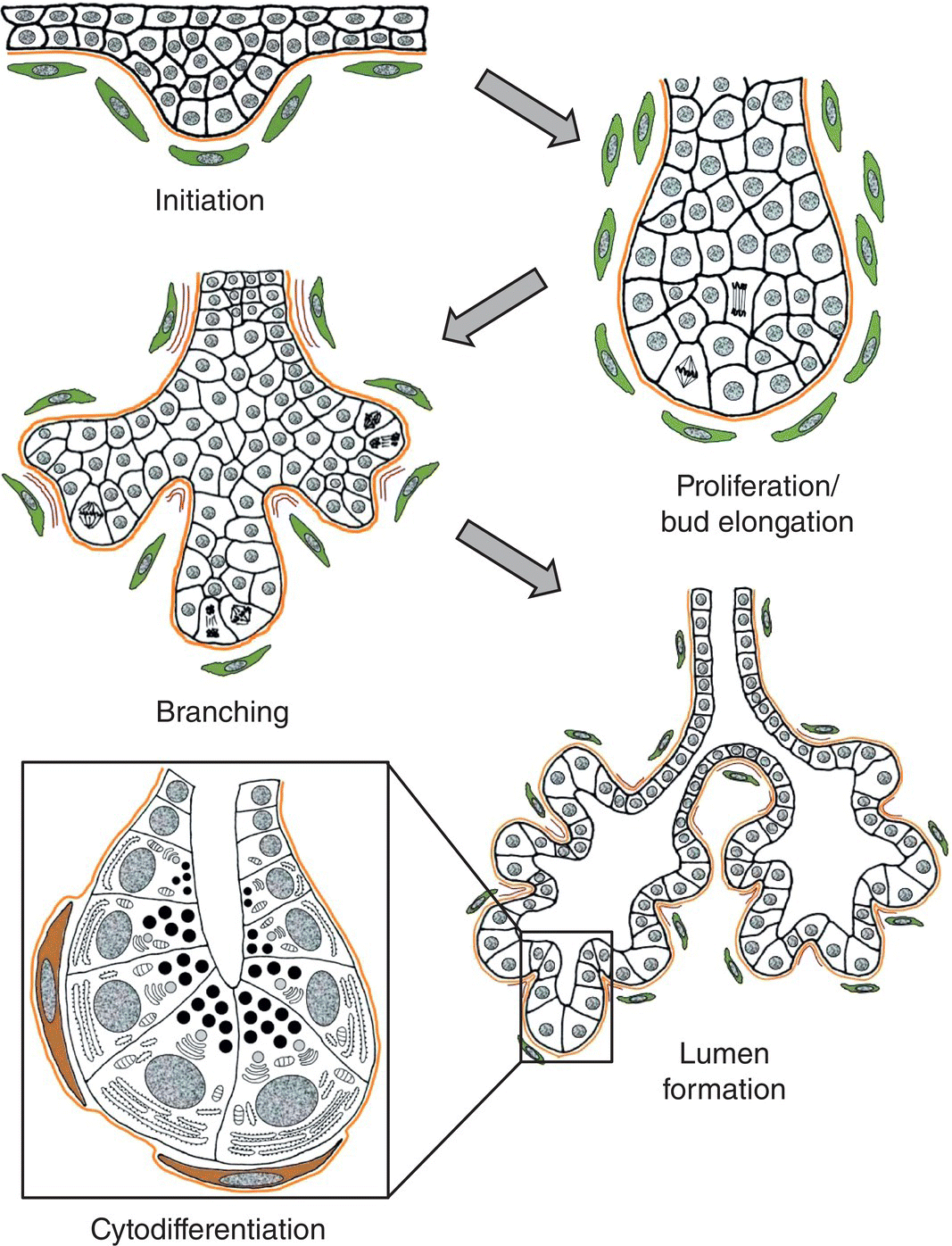
Figure 11.18 Salivary gland development occurs by the process of branching morphogenesis. The gland is initiated as a proliferation of the oral epithelium, forming a bud that grows into the underlying mesenchyme as a solid cord of epithelial cells. Reciprocal interactions between the epithelium and mesenchyme, involving specific growth factors, receptors, and transcription factors, regulate the developmental process. Continued cell proliferation and branching of the epithelial cord results in terminal buds connected by developing ducts. Deposition of extracellular matrix components stabilizes the clefts formed between adjacent branches. Lumina form in the ducts and terminal buds, and cells of the terminal buds undergo cytodifferentiation to become secretory cells of the endpieces. The basal lamina separating epithelium from mesenchyme is shown as an orange line, mesenchymal cells are green, and differentiated myoepithelial cells are brown.
(Modified from Cutler, L.S. (1989) Functional differentiation of salivary glands. In: Handbook of Physiology: The Gastrointestinal System, Vol. 3, pp. 93–105. American Physiological Society, Bethesda, MD.)
Major glands
Parotid gland
The parotid is the largest salivary gland. It is located in front of the ear, superficial to the masseter muscle, and the branches of the facial nerve (cranial nerve VII) pass through the gland. Its main duct, Stensen’s duct, passes forward across the masseter muscle, enters the oral cavity through the buccinator muscle, and opens at the parotid papilla on the buccal mucosa opposite the maxillary second molar. Branches of the carotid artery provide the blood supply to the gland. The preganglionic sympathetic nerves innervating the gland originate in the upper thoracic spinal cord and synapse in the superior cervical ganglion, and the postganglionic fibers accompany the blood vessels supplying the gland. The parasympathetic innervation of the parotid gland arises from the inferior salivatory nucleus of the glossopharyngeal nerve (cranial nerve IX) located in the medulla and reaches the otic ganglion via the lesser petrosal nerve. The postganglionic fibers travel to the gland via the auriculotemporal nerve.
The parotid gland is a pure serous gland: all of the secretory endpieces are serous in nature (Fig. 11.19). The serous cells produce a variety of proteins and glycoproteins that have significant roles in protection of the oral tissues and digestion. The intercalated ducts are relatively long, and the striated ducts are prominent. Scattered adipocytes are frequently found in the parenchyma. In older individuals substantial amounts of the parenchyma may be replaced by fat.
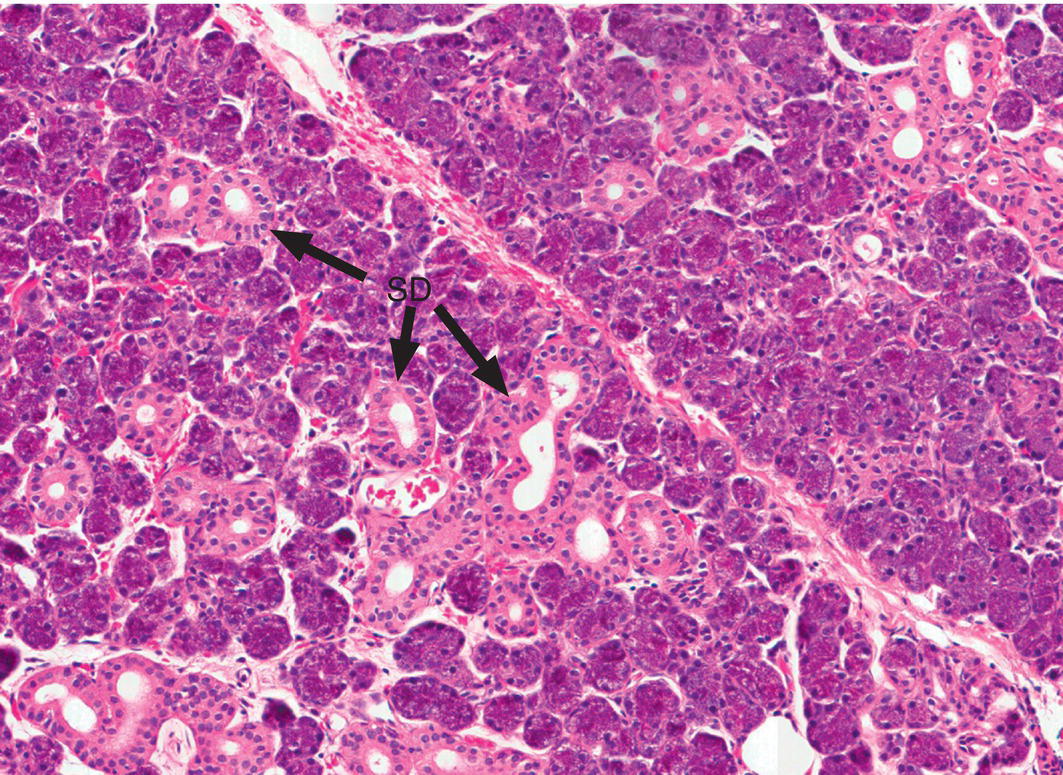
Figure 11.19 Parotid gland. The parotid gland contains serous endpieces and has numerous striated ducts (SD).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


